Abstract
Objectives
To evaluate the effect of obstructive sleep apnoea syndrome (OSAS) and continuous positive airway pressure (CPAP) therapy on choroidal structural changes and choroidal vascularity index (CVI) in patients with OSAS.
Methods
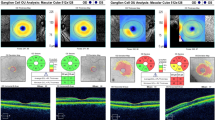
Choroidal structural changes in patients with OSAS immediately after diagnosis and 12 months after CPAP treatment were evaluated and compared with healthy controls. The choroidal images on enhanced depth imaging optical coherence tomography (EDI-OCT) were binarized into luminal area (LA) and stromal area (SA) using the ImageJ software. CVI was calculated as the ratio of LA to total choroid area (TCA). The correlations between the results of polysomnography (PSG) and choroidal parameters were evaluated.
Results
A total of 48 eyes of 48 patients (22 patients with OSAS, and 26 controls) were included. The mean age of the patients was 47.21 ± 8.82 (range, 30–63) years. The mean CVI values were 68.10 ± 1.80% in the OSAS group before CPAP therapy, and 69.22 ± 1.40% in the control group (p < 0.05). After 12 months of regular CPAP therapy, the mean CVI value increased significantly to 69.15 ± 1.77%, and SA decreased significantly from 0.51 ± 0.07 mm2 to 0.48 ± 0.07 mm2 in the OSAS group (p < 0.05). No statistically significant correlation was found between the results of PSG and choroidal structural parameters.
Conclusion
According to our results, OSAS was associated with increased stromal oedema in the choroid, which improved after 12 months of regular CPAP therapy. CVI can be an important parameter for the follow-up of patients with OSAS.
Similar content being viewed by others
Introduction
Obstructive sleep apnoea syndrome (OSAS) is characterized by recurrent episodes of upper airway collapse during sleep, which results in recurrent nocturnal asphyxia, fragmented sleep, fluctuations in systemic blood pressure, and increased sympathetic nervous system activity [1]. The estimated prevalence of OSAS was reported to be around 34% in men and 17% in women aged 30–70 years [2]. The first-line treatment for patients with OSAS is continuous positive airway pressure (CPAP) therapy [3]. In clinical trials, elimination of apnoeic events during sleep by CPAP in patients with OSAS was shown to reduce blood pressure and autonomic function abnormalities and improve vascular functions [4, 5].
Patients with untreated OSAS have an increased risk of systemic hypertension (HT), stroke, heart failure, diabetes mellitus (DM), and depression [6]. Also, some ocular disorders have been reported to be associated with OSAS including floppy eyelid syndrome, non-arteritic anterior ischaemic optic neuropathy, central serous retinopathy, retinal vein occlusion, and glaucoma [7]. In addition, OSAS and CPAP therapy in patients with OSAS may cause structural changes in the choroid. The choroidal tissue has the highest blood flow per unit weight of all tissue [8]. It is the densest vascular structure of the eye and responsible for the vascular supply to the outer retina, retinal pigment epithelium (RPE), and some portions of the optic nerve [9]. Choroidal changes are associated with retinal diseases, such as senile macular degeneration (SMD) and diabetic retinopathy [10, 11].
The choroidal tissue is localized between the light-absorbing RPE and the optically opaque sclera, thus it is more difficult to visualize it than the optically accessible retina. Accordingly, its role in the pathogenesis and progression of retinal diseases remains unclear. Enhanced depth imaging optical coherence tomography (EDI-OCT) is an imaging modality that has helped to gain insight into the choroid’s role in the eye because it provides better visualization of the choroid [12]. In the literature, several reports evaluated choroidal thickness (CT) changes using EDI-OCT in OSAS [13].
Although CT measurement is useful, it is an indirect assumption of choroidal blood flow, it cannot give information about the choroidal structure. Agrawal et al. described a new quantitative parameter, the choroidal vascularity index (CVI), as a novel OCT parameter for measuring structural parameters of the choroid in healthy eyes, and in eyes with panuveitis [14, 15]. CVI was calculated as the ratio of the luminal area (LA) over the total choroidal area (TCA). Since then, CVI has been evaluated in several conditions that potentially affect the choroidal circulation.
In our study, our aim was to evaluate the effect of OSAS and CPAP therapy on choroidal structural changes in patients with OSAS. We compared the mean CVI, LA, SA, and TCA values of patients with OSAS before and 12 months after CPAP treatment with those of healthy individuals.
Methods
Patient selection
The eyes of patients diagnosed as having OSAS between January 2015 and December 2020 and healthy controls were included in the study, retrospectively. All procedures were performed according to the tenets of the Declaration of Helsinki. Written informed consent was obtained before all procedures. The study was approved by the Scientific Research Commission of Hisar Hospital.
The eyes of the patients with best corrected visual acuity (BCVA) of at least 8/10 Snellen lines, refractive error with spherical equivalent (SE) within ±3 dioptres, IOP < 21 mm Hg, without any ocular or retinal disease, and no history of any ocular surgery or intravitreal injection were included. Patients with accompanying systemic diseases such as cardiovascular diseases, systemic hypertension (HT), metabolic syndrome, diabetes mellitus (DM), central apnoea, and history of any cerebrovascular disease were excluded.
Ocular examinations
The medical records of the patients were obtained retrospectively from the patients’ files. All eyes underwent ophthalmic examinations with BCVA using Snellen charts, slit-lamp biomicroscopy, intraocular pressure (IOP) measurements, and dilated funduscopic examinations. The axial length (AL) measurements of the eyes were taken using an IOLMaster (Carl Zeiss Meditec, Dublin, CA). The choroidal images of the eyes were obtained using the EDI mode of Cirrus HD-OCT 4000 (Carl Zeiss Meditec, Dublin, CA). The scan pattern was a 6-mm line consisting of 4096 A-scans. All measurements were performed between 8:00 AM and 2:00 PM. After 12 months of CPAP therapy, second EDI-OCT examinations in the OSAS group were performed.
Polysomnography (PSG)
All patients in the OSAS group underwent full-night PSG (Comet-PLUS, Grass Technologies, West Warwick, RI). The patients were advised not to consume caffeine-containing beverages, not to take medications, and not to sleep during the day, which might affect the results. The PSG recordings included the following parameters: central, temporal, and occipital electroencephalogram; bilateral electrooculogram; submental and anterior tibialis electromyogram; electrocardiogram; nasal airflow (using a nasal cannula and pressure transducer); naso-oral airflow (using a thermistor); and respiratory effort (using chest and abdominal piezoelectric belts). Oxygen saturation was monitored using an oximeter probe. Respiratory parameters were evaluated according to the standard criteria of the American Academy of Sleep Medicine (AASM) [16]. Apnoea was defined as a complete cessation of airflow for ≥10 s. Hypopnea was defined as a reduction in airflow of ≥50% accompanied by ≥3% desaturation. OSAS was categorized as mild, moderate, and severe according to the apnoea-hypopnea index (AHI). The AHI values of five to 14 events per hour were evaluated as mild OSAS, 15–29 events per hour as moderate OSAS, and 30 events or more per hour as severe OSAS.
Healthy controls were questioned in terms of OSAS symptoms using the Berlin Questionnaire (BQ), and the individuals with scores equal to zero were included.
CPAP therapy
Patients in the OSAS group were offered CPAP treatment. Patients were considered to be CPAP compliant if they used the device at least 4 hours per night and 5 days per week [17].
Image Binarization method
Image binarizations were performed using public domain software, ImageJ ((version 1.53e); http://imagej.nih.gov/ij/). Two ophthalmologists (MGA and HU) who were masked to the study groups, analysed all choroidal images independently and the mean values of the analyses were taken. Choroidal EDI-OCT images that passed through the fovea were selected. First, the line tool of ImageJ was used to determine the scale set by calibrating the pixel length of the image. Then, the image was converted to the 8-bit format, and the auto-local threshold and Niblack threshold settings were selected. The region of interest (ROI) was selected as total subfoveal choroidal area of the image using the polygon tool and added to the ROI manager. The upper edge of the selected area was along the choroid–RPE intersection and the lower edge was along the choroid–sclera intersection. Then, the colour threshold setting was applied after the image was converted to red, green, blue (RGB). In the brightness section, the first bar was adjusted to zero, and the second bar was adjusted to 250. After this process, the second measurement was also added to the ROI manager. The first and second measurements that were previously added to the ROI manager were selected and merged to measure the composite area with the AND tool of the ROI manager. Finally, LA and TCAs were measured using the ROI manager. By dividing the LA into the TCA, the CVI could be measured (Supplementary Fig. 1).
Statistical analysis
Data were analysed using the IBM SPSS Statistics 26 software package (IBM SPSS, Turkey). The Shapiro-Wilk test was used to examine the normality of data distribution. The data were obtained in the forms of mean, standard deviation (SD), minimum, maximum, median, frequency, and percentage. The significance of the differences between two independent groups was determined using the independent sample t test. The significance of the difference between two dependent groups was evaluated using the paired sample t test. The Chi-square test was used to determine whether there was a relationship between two independent categorical variables. Spearman’s rho correlation coefficient was used for the determination of the degree of non-causal relationships between two numerical variables. A p value of <0.05 was used to declare a statistically significant difference between the groups.
Results
A total of 48 eyes of 48 patients were included. The mean age of the patients was 47.21 ± 8.82 (range, 30–63) years. Twenty-two eyes of 22 patients with OSAS and 26 eyes of 26 healthy controls were evaluated in the study. The demographic characteristics of the patients and controls were similar; however, the mean body mass index (BMI) was significantly higher in the OSAS group than in the control group (p < 0.05). The comparison of the demographic characteristics between the groups is shown in Table 1.
All patients in the OSAS group underwent PSG before the study. According to the AHI scores of the PSG results, there were two patients with mild OSAS, 11 patients with moderate OSAS, and nine patients with severe OSAS. The mean values of the parameters of PSG tests are given in Table 2.
The mean CVI value was 68.10 ± 1.80% in the OSAS group before the CPAP therapy, and 69.22 ± 1.40% in the control group (p < 0.05). There was no statistically significant difference for the mean values of LA, SA, and TCA between the OSAS group before CPAP therapy and the control group (p > 0.05). None of the choroidal parameters was significantly different between the OSAS group after the CPAP therapy and the control group (p > 0.05). The comparison of the different choroidal parameters between the OSAS subgroups and the control group is shown in Table 3.
The mean CVI value increased from 68.10 ± 1.80% to 69.15 ± 1.77% after the CPAP therapy in the OSAS group (p < 0.01). The changes of the mean LA and TCA values were not statistically significant; however, the mean SA value significantly decreased from 0.51 ± 0.07 mm2 to 0.48 ± 0.07 mm2 (p < 0.05) (Table 4).
There was no statistically significant correlation between the choroidal structural parameters and the PSG parameters (p > 0.05). The correlation between the parameters is shown in Table 5.
Discussion
OSAS is primarily a disorder related to the collapse of the upper airway during sleep; however, its pathophysiologic impact on other body systems is being increasingly recognized. Cessation of airflow during sleep leads to intermittent hypoxia, increased respiratory effort, sleep fragmentation, and sympathetic activation, which leads to elevated systemic blood pressure, endothelial dysfunction, and potentially oxidative stress [18]. OSAS may also affect choroidal blood flow by these associated mechanisms.
There are several reports in the literature evaluating the effect of OSAS on CT; however, the results are contradictory [13]. In some reports, it was determined that patients with OSAS had abnormal choroidal structural alterations [19,20,21,22], whereas in others, no effect of OSAS on the choroid was reported [23, 24]. In some studies, it was shown that the choroid had an autoregulation capability [25]. Tonini et al. [26] reported that choroidal vascular reactivity to hyperoxia and hypercapnia in OSAS was not significantly different from that in healthy individuals. This result can be explained by the autoregulation mechanism of choroidal blood flow, and it can be thought that that the choroidal blood flow may be preserved in patients with mild OSAS. However, Geisser et al. [27] reported that an increase in arterial carbon dioxide (CO2) pressure per 1 mm Hg PaCO2 increases choroidal blood flow by ~1.5%.
Unlike other studies in the literature, we calculated choroidal structural areas using the ImageJ software instead of using CT. CT gives information about a localized choroidal area, whereas TCA gives information about the entire choroidal area in EDI-OCT images. In addition, in our study, SA and LA were calculated using image binarization. We found that the mean LA, SA, and TCA values were higher in the OSAS group before CPAP therapy than in the control group, although the differences were not statistically significant (p > 0.05). These results can be explained by the inflammation due to oxidative stress and endothelial dysfunction in OSAS. Similar to our results, Durusoy et al. [28] reported that hypercapnia due to prolonged use of N95 masks might cause choroidal hemodynamic changes and transient increased CT in healthy individuals. CVI was found significantly lower in the OSAS group before CPAP therapy because the percentage of involvement in SA was higher.
CPAP is the first-line therapy for OSAS. CPAP suppresses abnormal respiratory events during sleep, reverses partly or completely acute and chronic cardiovascular changes due to OSAS, and may improve microvascular endothelial function [29, 30]. In our study, consistent with these results, SA decreased significantly after 12 months of regular CPAP therapy (p < 0.05), which can be explained by a decrease in stromal oedema as a result of CPAP therapy. Consequently, CVI increased after CPAP therapy.
He et al. [13] reported that the CT tended to decrease with increasing severity of OSAS, and suggested that the choroidal blood flow might be preserved in the mild stages of OSAS; however, patients with moderate-to-severe OSAS had decreased choroidal blood flow. As OSAS becomes chronic, atrophy may develop in the choroidal tissue, which may cause a decrease in CT. However, in our study, all patients had newly diagnosed OSAS, they were relatively young and never-treated patients, so this may have affected the results.
Our study had some limitations. The exact OSAS duration was not known, the sample was relatively small, the participants in the control group were evaluated using only the BQ, and the mean BMI in the OSAS group was significantly higher than in the control group. In our study, all measurements were taken between 8:00 AM and 2:00 PM. It would have been ideal to take all the measurements in a shorter time frame as it could be important that some diurnal variation could affect choroidal measurements.
Conclusion
In our study, OSAS was found to be associated with increased stromal oedema in the choroid, which improved after 12 months of regular CPAP therapy. CVI can be an important parameter for the follow-up of patients with OSAS. Further prospective studies with larger sample sizes are needed on this issue.
Summary
What was known before
-
There are several reports in the literature evaluating the effect of OSAS on CT; however, the results are contradictory. In some reports, it was determined that patients with OSAS had abnormal choroidal structural alterations, whereas in others, no effect of OSAS on the choroid was reported.
What this study adds
-
Unlike other studies in the literature, we calculated choroidal structural areas using the ImageJ software instead of using CT. CT gives information about a localized choroidal area, whereas TCA gives information about the entire choroidal area in EDI-OCT images. In addition, in our study, SA and LA were calculated using image binarization.
References
White DP. Sleep-related breathing disorder: 2—pathophysiology of obstructive sleep apnoea. Thorax. 1995;50:797–804.
Peppard PE, Young T, Barnet JH, Palta M, Hagen EW, Hla KM. Increased prevalence of sleep-disordered breathing in adults. Am J Epidemiol. 2013;177:1006–14.
Basner RC. Continuous positive airway pressure for obstructive sleep apnea. N Engl J Med. 2007;356:1751–8.
Pepperell JC, Ramdassingh-Dow S, Crosthwaite N, Mullins R, Jenkinson C, Stradling JR, et al. Ambulatory blood pressure after therapeutic and subtherapeutic nasal continuous positive airway pressure for obstructive sleep apnea: a randomized parallel trial. Lancet. 2002;359:204–10.
Bayram NA, Ciftci B, Keles T, Durmaz T, Turhan S, Bozkurt E, et al. Endothelial function in normotensive men with obstructive sleep apnea before and 6 months after CPAP treatment. Sleep. 2009;32:1257–63.
Heinzer R, Vat S, Marques-Vidal P, Marti-Soler H, Andries D, Tobback N, et al. Prevalence of sleep-disordered breathing in the general population: the HypnoLaus study. Lancet Respir Med. 2015;3:310–8.
Santos M, Hofmann RJ. Ocular manifestations of obstructive sleep apnea. J Clin Sleep Med. 2017;13:1345–8.
Alm A, Bill A. Ocular and optic nerve blood flow at normal and increased intraocular pressures in monkeys (Macacairus): a study with radioactively labelled microspheres including flow determinations in brain and some other tissues. Exp Eye Res. 1973;15:15–29.
Tan KA, Gupta P, Agarwal A, Chhablani J, Cheng CY, Keane PA, et al. State of science: choroidal thickness and systemic health. Surv Ophthalmol. 2016;61:566–81.
Invernizzi A, Benatti E, Cozzi M, Erba S, Vaishnavi S, Vupparaboina KK, et al. Choroidal structural changes correlate with neovascular activity in neovascular age related macular degeneration. Invest Ophthalmol Vis Sci. 2018;59:3836–41.
Wang H, Tao Y. Choroidal structural changes correlate with severity of diabetic retinopathy in diabetes mellitus. BMC Ophthalmol. 2019;19:186.
Spaide RF, Koizumi H, Pozzoni MC. Enhanced depth imaging spectral-domain optical coherence tomography. Am J Ophthalmol. 2008;146:496–500.
He M, Han X, Wu H, Huang W. Choroidal thickness changes in obstructive sleep apnea syndrome: a systematic review and meta-analysis. Sleep Breath. 2016;20:369–78.
Agrawal R, Gupta P, Tan KA, Cheung CMG, Wong TY, Cheng CY. Choroidal vascularity index as a measure of vascular status of the choroid: measurements in healthy eyes from a population-based study. Sci Rep. 2016;6:1–9.
Agrawal R, Salman M, Tan K, Karampelas M. Choroidal vascularity index (CVI)—A novel optical coherence tomography parameter for monitoring patients with panuveitis? Plos One. 2016;11:e0146344.
American Academy of Sleep Medicine. Task Force Report. Sleep-related breathing disorders in adults: recommendations for syndrome definition and measurement techniques in clinical research. Sleep. 1999;22:667–89.
Richard W, Venker J, den Herder C, Kox D, van den Berg B, Laman M, et al. Acceptance and longterm compliance of nCPAP in obstructive sleep apnea. Eur Arch Otorhinolaryngol. 2007;264:1081–6.
Kohler M, Stradling JR. Mechanisms of vascular damage in obstructive sleep apnea. Nat Rev Cardiol. 2010;7:677–85.
Xin C, Wang J, Zhang W, Wang L, Peng X. Retinal and choroidal thickness evaluation by SD-OCT in adults with obstructive sleep apnea-hypopnea syndrome (OSAS). Eye. 2014;28:415–21.
Ekinci M, Huseyinoglu N, Cagatay HH, Keles S, Ceylan E, Gokce G. Choroidal thickening in patients with sleep apnea syndrome. Neuro-Ophthalmol. 2014;38:8–13.
Kara S, Ozcimen M, Bekci TT, Sakarya Y, Gencer B, Tufan HA, et al. Evaluation of choroidal thickness in patients with obstructive sleep apnea/hypopnea syndrome. Arq Bras Oftalmol. 2014;77:280–4.
Karalezli A, Eroglu FC, Kivanc T, Dogan R. Evaluation of choroidal thickness using spectral-domain optical coherence tomography in patients with severe obstructive sleep apnea syndrome: a comparative study. Int J Ophthalmol. 2014;7:1030–4.
Karaca EE, Ekici F, Yalcin NG, Ciftci TU, Ozdek S. Macular choroidal thickness measurements in patients with obstructive sleep apnea syndrome. Sleep Breath. 2014;19:335–41.
Zengin MO, Oz T, Baysak A, Cinar E, Kucukerdonmez C. Changes in choroidal thickness in patients with obstructive sleep apnea syndrome. Ophthalmic Surg Lasers Imaging Retin. 2014;45:298–304.
Kiel JW, van Heuven WA. Ocular perfusion pressure and choroidal blood flow in the rabbit. Invest Ophthalmol Vis Sci. 1995;36:579–85.
Tonini M, Khayi H, Pepin JL, Renard E, Baguet JP, Levy P, et al. Choroidal blood-flow responses to hyperoxia and hypercapnia in men with obstructive sleep apnea. Sleep 2010;33:811–8.
Geiser MH, Riva CE, Dorner GT, Diermann U, Luksch A, Schmetterer L. Response of choroidal blood flow in the foveal region to hyperoxia and hyperoxia-hypercapnia. Curr Eye Res. 2000;21:669–76.
Karatas Durusoy G, Gumus G, Demir G Evaluati̇on of the effecT OF N95 face mask used by healthcare professi̇onals on choroi̇dal thi̇ckness. Photodiagnosis Photodyn Ther. (2021) https://doi.org/10.1016/j.pdpdt.2021.102279.
Jelic S, Padeletti M, Kawut SM, Higgins C, Canfield SM, Onat D, et al. Inflammation, oxidative stress, and repair capacity of the vascular endothelium in obstructive sleep apnea. Circulation. 2008;117:2270–8.
Lattimore JL, Wilcox I, Skilton M, Langenfeld M, Celermajer DS. Treatment of obstructive sleep apnoea leads to improved microvascular endothelial function in the systemic circulation. Thorax. 2006;61:491–5.
Author information
Authors and Affiliations
Contributions
MGA conceived of the presented idea. MGA and HU developed the theory and performed the computations. HU and OD were responsible for collecting the data. AYK and OD verified the analytical methods. MGA was responsible for writing the report, arbitrating potentially eligible studies, extracting and analysing data and interpreting results. All authors discussed the results and contributed to the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
All procedures performed in experiments involving human participants were in accordance with the ethical standards of the institutional and national research committee and with the 1964 Declaration of Helsinki. The study was approved by the Scientific Research Commission of Hisar Hospital.
Informed consent
Written informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Altinel, M.G., Uslu, H., Kanra, A.Y. et al. Effect of obstructive sleep apnoea syndrome and continuous positive airway pressure treatment on choroidal structure. Eye 36, 1977–1981 (2022). https://doi.org/10.1038/s41433-021-01790-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-021-01790-w