Abstract
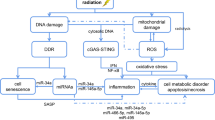
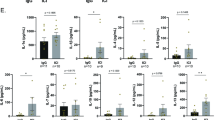
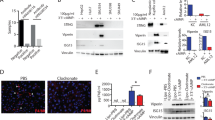
Liver damage upon exposure to ionizing radiation (IR), whether accidental or therapeutic, can contribute to liver dysfunction. Currently, radiotherapy (RT) is used for various cancers including hepatocellular carcinoma (HCC); however, the treatment dose is limited by radiation-induced liver disease (RILD) with a high mortality rate. Furthermore, the precise molecular mechanisms of RILD remain poorly understood. Here, we investigated RILD pathogenesis using various knockout mouse strains subjected to whole-liver irradiation. We found that hepatocytes released a large quantity of double-stranded DNA (dsDNA) after irradiation. The cGAS-STING pathway in non-parenchymal cells (NPCs) was promptly activated by this dsDNA, causing interferon (IFN)-I production and release and concomitant hepatocyte damage. Genetic and pharmacological ablation of the IFN-I signaling pathway protected against RILD. Moreover, clinically irradiated human peri-HCC liver tissues exhibited substantially higher STING and IFNβ expression than non-irradiated tissues. Increased serum IFNβ concentrations post-radiation were associated with RILD development in patients. These results delineate cGAS-STING induced type 1 interferon release in NPCs as a key mediator of IR-induced liver damage and described a mechanism of innate-immunity-driven pathology, linking cGAS-STING activation with amplification of initial radiation-induced liver injury.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Jemal, A. et al. Global cancer statistics. CA Cancer J. Clin. 61, 69–90. (2011).
Karaman, B., Battal, B., Sari, S. & Verim, S. Hepatocellular carcinoma review: current treatment, and evidence-based medicine. World J. Gastroenterol. 20, 18059–18060 (2014).
Gerum, S., Jensen, A. D. & Roeder, F. Stereotactic body radiation therapy in patients with hepatocellular carcinoma: a mini-review. World J. Gastrointest. Oncol. 11, 367–376 (2019).
Koay, E. J., Owen, D. & Das, P. Radiation-induced liver disease and modern radiotherapy. Semin. Radiat. Oncol. 28, 321–331 (2018).
Dawson, L. A. et al. Analysis of radiation-induced liver disease using the Lyman NTCP model. Int. J. Radiat. Oncol. Biol. Phys. 53, 810–821 (2002).
Wang, S. et al. Liver toxicity induced by combined external-beam irradiation and radioimmunoglobulin therapy. Radiat. Res. 141, 294–302 (1995).
Du, S. S. et al. Inactivation of kupffer cells by gadolinium chloride protects murine liver from radiation-induced apoptosis. Int. J. Radiat. Oncol. Biol. Phys. 76, 1225–1234 (2010).
Christiansen, H. et al. Irradiation leads to susceptibility of hepatocytes to TNF-alpha mediated apoptosis. Radiother. Oncol. 72, 291–296 (2004).
Dong, Y. et al. Activation of the JNK-c-Jun pathway in response to irradiation facilitates Fas ligand secretion in hepatoma cells and increases hepatocyte injury. J. Exp. Clin. Cancer Res. 35, 114 (2016).
Chen, Q., Sun, L. & Chen, Z. J. Regulation and function of the cGAS-STING pathway of cytosolic DNA sensing. Nat. Immunol. 17, 1142–1149 (2016).
Sun, L., Wu, J., Du, F., Chen, X. & Chen, Z. J. Cyclic GMP-AMP synthase is a cytosolic DNA sensor that activates the type I interferon pathway. Science 339, 786–791 (2013).
Wu, J. et al. Cyclic GMP-AMP is an endogenous second messenger in innate immune signaling by cytosolic DNA. Science 339, 826–830 (2013).
Ishikawa, H. & Barber, G. N. STING is an endoplasmic reticulum adaptor that facilitates innate immune signalling. Nature 455, 674–678 (2008).
Gao, D. et al. Activation of cyclic GMP-AMP synthase by self-DNA causes autoimmune diseases. Proc. Natl. Acad. Sci. USA 112, E5699–E5705 (2015).
Gray, E. E., Treuting, P. M., Woodward, J. J. & Stetson, D. B. Cutting edge: cGAS is required for lethal autoimmune disease in the Trex1-deficient mouse model of aicardi-goutieres syndrome. J. Immunol. 195, 1939–1943 (2015).
Deng, L. et al. STING-dependent cytosolic DNA sensing promotes radiation-induced type I interferon-dependent antitumor immunity in immunogenic tumors. Immunity 41, 843–852 (2014).
Demaria, O. et al. STING activation of tumor endothelial cells initiates spontaneous and therapeutic antitumor immunity. Proc. Natl. Acad. Sci. USA 112, 15408–15413 (2015).
Wang, M. J., Chen, F., Lau, J. T. Y. & Hu, Y. P. Hepatocyte polyploidization and its association with pathophysiological processes. Cell Death Dis. 8, e2805 (2017).
Jackson, S. P. & Bartek, J. The DNA-damage response in human biology and disease. Nature 461, 1071–1078 (2009).
Petrasek, J. et al. STING-IRF3 pathway links endoplasmic reticulum stress with hepatocyte apoptosis in early alcoholic liver disease. Proc. Natl. Acad. Sci. USA 110, 16544–16549 (2013).
Ahn, J., Gutman, D., Saijo, S. & Barber, G. N. STING manifests self DNA-dependent inflammatory disease. Proc. Natl. Acad. Sci. USA 109, 19386–19391 (2012).
Lawrence, T. S. et al. Hepatic toxicity resulting from cancer treatment. Int. J. Radiat. Oncol. Biol. Phys. 31, 1237–1248 (1995).
Araujo A. M., et al. Liver immune cells release type 1 interferon due to DNA sensing and amplify liver injury from acetaminophen overdose. Cells 7, 1–16. https://doi.org/10.3390/cells7080088 (2018).
Thomsen, M. K. et al. Lack of immunological DNA sensing in hepatocytes facilitates hepatitis B virus infection. Hepatology 64, 746–759 (2016).
Barber, G. N. STING: infection, inflammation and cancer. Nat. Rev. Immunol. 15, 760–770 (2015).
Bhattacharya, A. et al. Superoxide dismutase 1 protects hepatocytes from type I interferon-driven oxidative damage. Immunity 43, 974–986 (2015).
Mapuskar, K. A., Anderson, C. M., Spitz, D. R., Batinic-Haberle, I. & Allen, B. G. Utilizing superoxide dismutase mimetics to enhance radiation therapy response while protecting normal tissues. Semin. Radiat. Oncol. 29, 72–80 (2019).
Nagata, S. Apoptosis and autoimmune diseases. Ann. N. Y. Acad. Sci. 1209, 10–16 (2010).
Munoz-Schuffenegger, P., Ng, S. & Dawson, L. A. Radiation-induced liver toxicity. Semin. Radiat. Oncol. 27, 350–357 (2017).
Deng, Z. B. et al. Immature myeloid cells induced by a high-fat diet contribute to liver inflammation. Hepatology 50, 1412–1420 (2009).
Xu, H. et al. Myeloid cell-specific disruption of Period1 and Period2 exacerbates diet-induced inflammation and insulin resistance. J. Biol. Chem. 289, 16374–16388 (2014).
Iracheta-Vellve, A. et al. Endoplasmic reticulum stress-induced hepatocellular death pathways mediate liver injury and fibrosis via stimulator of interferon genes. J. Biol. Chem. 291, 26794–26805 (2016).
Qiao, J. T. et al. Activation of the STING-IRF3 pathway promotes hepatocyte inflammation, apoptosis and induces metabolic disorders in nonalcoholic fatty liver disease. Metabolism 81, 13–24 (2018).
Cho, C. S. et al. Lipotoxicity induces hepatic protein inclusions through TANK binding kinase 1-mediated p62/sequestosome 1 phosphorylation. Hepatology 68, 1331–1346 (2018).
Malhi, H. & Gores, G. J. Molecular mechanisms of lipotoxicity in nonalcoholic fatty liver disease. Semin. Liver Dis. 28, 360–369 (2008).
Tosello-Trampont, A. C., Landes, S. G., Nguyen, V., Novobrantseva, T. I. & Hahn, Y. S. Kuppfer cells trigger nonalcoholic steatohepatitis development in diet-induced mouse model through tumor necrosis factor-alpha production. J. Biol. Chem. 287, 40161–40172 (2012).
Alati, T., Van Cleeff, M., Strom, S. C. & Jirtle, R. L. Radiation sensitivity of adult human parenchymal hepatocytes. Radiat. Res. 115, 152–160 (1988).
Radoshevich, L. & Dussurget, O. Cytosolic innate immune sensing and signaling upon infection. Front. Microbiol. 7, 313 (2016).
Benci, J. L. et al. Tumor interferon signaling regulates a multigenic resistance program to immune checkpoint blockade. Cell 167, 1540–1554.e12 (2016).
Katlinski, K. V. et al. Inactivation of interferon receptor promotes the establishment of immune privileged tumor microenvironment. Cancer Cell 31, 194–207 (2017).
Woo, S.-R. et al. STING-dependent cytosolic DNA sensing mediates innate immune recognition of immunogenic tumors. Immunity 41, 830–842 (2014).
Spranger, S. et al. Up-regulation of PD-L1, IDO, and T(regs) in the melanoma tumor microenvironment is driven by CD8(+) T cells. Sci. Transl. Med. 5, 200ra116 (2013).
Taube, J. M. et al. Colocalization of inflammatory response with B7-h1 expression in human melanocytic lesions supports an adaptive resistance mechanism of immune escape. Sci. Transl. Med. 4, 127ra37 (2012).
Weichselbaum, R. R. et al. An interferon-related gene signature for DNA damage resistance is a predictive marker for chemotherapy and radiation for breast cancer. Proc. Natl. Acad. Sci. USA 105, 18490–18495 (2008).
Boelens, M. C. et al. Exosome transfer from stromal to breast cancer cells regulates therapy resistance pathways. Cell 159, 499–513 (2014).
Erdal, E., Haider, S., Rehwinkel, J., Harris, A. L. & McHugh, P. J. A prosurvival DNA damage-induced cytoplasmic interferon response is mediated by end resection factors and is limited by Trex1. Genes Dev. 31, 353–369 (2017).
Post, A. E. M. et al. Interferon-stimulated genes are involved in cross-resistance to radiotherapy in tamoxifen-resistant breast cancer. Clin. Cancer Res. 24, 3397–3408 (2018).
Chen, J. et al. IFN protects cancer cells from CD8+ T cell-mediated cytotoxicity after radiation. J. Clin. Invest. 129, 4224–4238 (2019).
Xu, H. et al. ADAMTS13 controls vascular remodeling by modifying VWF reactivity during stroke recovery. Blood 130, 11–22 (2017).
Wu, Z. F. et al. Toll-like receptor 4 and its associated proteins as prognostic factors for HCC treated by post-radiotherapy surgery. Oncol. Lett. 15, 9599–9608 (2018).
Acknowledgements
This work was supported by the National Nature Science Foundation of China (No. 81773220 and U1505229).
Author information
Authors and Affiliations
Contributions
Study concept and design: S.D. and Z.Z.; acquisition of data: G.C. and S.D.; analysis and interpretation of data: S.D. and G.C.; acquisition of patient specimens: P.Y., Y.C., Y.H. and S.D.; drafting of the manuscript: S.D. and Z.Z.; critical revision of the manuscript: J.Z., J.F. and Z.Z.; obtained funding: administrative, Z.Z.; technical or other material support: S.D. and G.C.; study supervision: Z.Z.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Du, S., Chen, G., Yuan, B. et al. DNA sensing and associated type 1 interferon signaling contributes to progression of radiation-induced liver injury. Cell Mol Immunol 18, 1718–1728 (2021). https://doi.org/10.1038/s41423-020-0395-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41423-020-0395-x
Keywords
This article is cited by
-
The dual function of cGAS-STING signaling axis in liver diseases
Acta Pharmacologica Sinica (2024)
-
Deletion of Cyclic GMP-AMP Synthase Aggravates Concanavalin A-Induced Acute Hepatic Injury by Facilitating Leukocyte Chemotaxis
Inflammation (2023)
-
Activation of cDCs and iNKT cells contributes to triptolide-induced hepatotoxicity via STING signaling pathway and endoplasmic reticulum stress
Cell Biology and Toxicology (2023)
-
TLD calibration and absorbed dose measurement in a radiation-induced liver injury model under a linear accelerator
Nuclear Science and Techniques (2023)
-
Emodin Attenuates Acetaminophen-Induced Hepatotoxicity via the cGAS-STING Pathway
Inflammation (2022)