Abstract
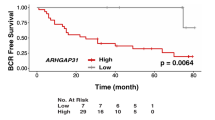
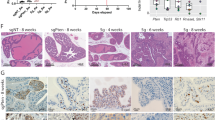
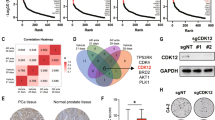
Metastatic castration-resistant prostate cancer (mCRPC) is associated with a poor prognosis and remains an incurable fatal disease. Therefore, the identification of molecular markers involved in cancer progression is urgently needed to develop more-effective therapies. The present study investigated the role of the Wnt signaling modulator Dickkopf-1 (DKK1) in the growth and metastatic progression of mCRPC. DKK1 silencing through siRNA and deletion via CRISPR/Cas9 editing were performed in two different metastatic castration-resistant prostate cancer cell lines (PC3 and DU145). A xenograft tumor model was used to assess tumor growth and metastases. In in vitro experiments, both DKK1 silencing and deletion reduced cell growth and migration of both cell lines. DKK1 knockout clones (DKK1-KO) exhibited cell cycle arrest, tubulin reorganization, and modulation of tumor metastasis-associated genes. Furthermore, in DKK1-KO cells, E-cadherin re-expression and its membrane co-localization with β-catenin were observed, contributing to reduced migration; Cadherin-11, known to increase during epithelial-mesenchymal transition, was down-regulated in DKK1-KO cells. In the xenograft mouse model, DKK1 deletion not only reduced tumor growth but also inhibited the formation of lung metastases. In conclusion, our findings support the key role of DKK1 in the growth and metastatic dissemination of mCRPC, both in vitro and in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The data generated and analyzed during this study can be found within the published article and its supplementary files. Additional data are available from the corresponding author upon reasonable request.
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71:209–49.
Bekelman JE, Rumble RB, Chen RC, Pisansky TM, Finelli A, Feifer A, et al. Clinically Localized Prostate Cancer: ASCO Clinical Practice Guideline Endorsement of an American Urological Association/American Society for Radiation Oncology/Society of Urologic Oncology Guideline. J Clin Oncol. 2018;36:3251–8.
Sweeney CJ, Chen YH, Carducci M, Liu G, Jarrard DF, Eisenberger M, et al. Chemohormonal therapy in metastatic hormone-sensitive prostate cancer. N Engl J Med. 2015;373:737–46.
Hahn AW, Siddiqui BA, Leo J, Dondossola E, Basham KJ, Miranti CK, et al. Cancer cell-extrinsic roles for the androgen receptor in prostate cancer. Endocrinology. 2023;164:bqad078.
Gillette CM, Yette GA, Cramer SD, Graham LS. Management of advanced prostate cancer in the precision oncology era. Cancers (Basel). 2023;15:2552.
Watson PA, Arora VK, Sawyers CL. Emerging mechanisms of resistance to androgen receptor inhibitors in prostate cancer. Nat Rev Cancer. 2015;15:701–11.
Karantanos T, Corn PG, Thompson TC. Prostate cancer progression after androgen deprivation therapy: mechanisms of castrate resistance and novel therapeutic approaches. Oncogene. 2013;32:5501–11.
Zhan T, Rindtorff N, Boutros M. Wnt signaling in cancer. Oncogene. 2017;36:1461–73.
Wang L, Dehm SM, Hillman DW, Sicotte H, Tan W, Gormley M, et al. A prospective genome-wide study of prostate cancer metastases reveals association of wnt pathway activation and increased cell cycle proliferation with primary resistance to abiraterone acetate-prednisone. Ann Oncol. 2018;29:352–60.
Isaacsson Velho P, Fu W, Wang H, Mirkheshti N, Qazi F, Lima FAS, et al. Wnt-pathway activating mutations are associated with resistance to first-line abiraterone and enzalutamide in castration-resistant prostate cancer. European Urology. 2020;77:14–21.
Patel R, Brzezinska EA, Repiscak P, Ahmad I, Mui E, Gao M, et al. Activation of β-catenin cooperates with loss of pten to drive AR-independent castration-resistant prostate cancer. Cancer Res. 2020;80:576–90.
Hayat R, Manzoor M, Hussain A. Wnt signaling pathway: a comprehensive review. Cell Biol Int. 2022;46:863–77.
Kagey MH, He X. Rationale for targeting the Wnt signalling modulator Dickkopf-1 for oncology. Br J Pharmacol. 2017;174:4637–50.
Jiang H, Zhang Z, Yu Y, Chu HY, Yu S, Yao S, et al. Drug discovery of DKK1 inhibitors. Front Pharmacol. 2022;13:847387.
Tian E, Zhan F, Walker R, Rasmussen E, Ma Y, Barlogie B, et al. The role of the Wnt-signaling antagonist DKK1 in the development of osteolytic lesions in multiple myeloma. N Engl J Med. 2003;349:2483–94.
Liu Y, Tang W, Xie L, Wang J, Deng Y, Peng Q, et al. Prognostic significance of dickkopf-1 overexpression in solid tumors: a meta-analysis. Tumour Biol. 2014;35:3145–54.
Sheng SL, Huang G, Yu B, Qin WX. Clinical significance and prognostic value of serum Dickkopf-1 concentrations in patients with lung cancer. Clin Chem. 2009;55:1656–64.
Kim SU, Park JH, Kim HS, Lee JM, Lee HG, Kim H, et al. Serum Dickkopf-1 as a biomarker for the diagnosis of hepatocellular carcinoma. Yonsei Med J. 2015;56:1296–306.
Jiang T, Huang L, Zhang S. DKK-1 in serum as a clinical and prognostic factor in patients with cervical cancer. Int J Biol Markers. 2013;28:221–5.
Aguilera O, Fraga MF, Ballestar E, Paz MF, Herranz M, Espada J, et al. Epigenetic inactivation of the Wnt antagonist DICKKOPF-1 (DKK-1) gene in human colorectal cancer. Oncogene. 2006;25:4116–21.
Mikheev AM, Mikheeva SA, Maxwell JP, Rivo JV, Rostomily R, Swisshelm K, et al. Dickkopf-1 mediated tumor suppression in human breast carcinoma cells. Breast Cancer Res Treat. 2008;112:263–73.
Cho SW, Lee EJ, Kim H, Kim SH, Ahn HY, Kim YA, et al. Dickkopf-1 inhibits thyroid cancer cell survival and migration through regulation of β-catenin/E-cadherin signaling. Mol Cell Endocrinol. 2013;366:90–8.
Thudi NK, Martin CK, Murahari S, Shu ST, Lanigan LG, Werbeck JL, et al. Dickkopf-1 (DKK-1) stimulated prostate cancer growth and metastasis and inhibited bone formation in osteoblastic bone metastases. Prostate. 2011;71:615–25.
Rachner TD, Thiele S, Göbel A, Browne A, Fuessel S, Erdmann K, et al. High serum levels of Dickkopf-1 are associated with a poor prognosis in prostate cancer patients. BMC Cancer. 2014;14:649.
D’Amelio P, Roato I, Oderda M, Soria F, Zitella A, Ferracini R, et al. DKK-1 in prostate cancer diagnosis and follow up. BMC Clin Pathol. 2014;14:11.
Hall CL, Daignault SD, Shah RB, Pienta KJ, Keller ET. Dickkopf-1 expression increases early in prostate cancer development and decreases during progression from primary tumor to metastasis. Prostate. 2008;68:1396–404.
Rinella L, Marano F, Paletto L, Fraccalvieri M, Annaratone L, Castellano I, et al. Extracorporeal shock waves trigger tenogenic differentiation of human adipose-derived stem cells. Connect Tissue Res. 2018;59:561–73.
Roato I, D’Amelio P, Gorassini E, Grimaldi A, Bonello L, Fiori C, et al. Osteoclasts are active in bone forming metastases of prostate cancer patients. PLoS ONE. 2008;3:e3627.
Giralt I, Gallo-Oller G, Navarro N, Zarzosa P, Pons G, Magdaleno A, et al. Dickkopf-1 inhibition reactivates Wnt/β-catenin signaling in rhabdomyosarcoma, induces myogenic markers in vitro and impairs tumor cell survival in vivo. IJMS. 2021;22:12921.
Hall CL, Zhang H, Baile S, Ljungman M, Kuhstoss S, Keller ET. p21CIP-1/WAF-1 induction is required to inhibit prostate cancer growth elicited by deficient expression of the Wnt inhibitor Dickkopf-1. Cancer Res. 2010;70:9916–26.
Wang Z, Wang J, Chen Z, Wang K, Shi L. MicroRNA-1-3p inhibits the proliferation and migration of oral squamous cell carcinoma cells by targeting DKK1. Biochem Cell Biol. 2018;96:355–64.
Takahashi N, Fukushima T, Yorita K, Tanaka H, Chijiiwa K, Kataoka H. Dickkopf-1 is overexpressed in human pancreatic ductal adenocarcinoma cells and is involved in invasive growth. Int J Cancer. 2010;126:1611–20.
Qi L, Sun B, Liu Z, Li H, Gao J, Leng X. Dickkopf-1 inhibits epithelial-mesenchymal transition of colon cancer cells and contributes to colon cancer suppression. Cancer Sci. 2012;103:828–35.
Vitale I, Galluzzi L, Castedo M, Kroemer G. Mitotic catastrophe: a mechanism for avoiding genomic instability. Nat Rev Mol Cell Biol. 2011;12:385–92.
Vakifahmetoglu H, Olsson M, Zhivotovsky B. Death through a tragedy: mitotic catastrophe. Cell Death Differ. 2008;15:1153–62.
Kim S, Min A, Lee KH, Yang Y, Kim TY, Lim JM, et al. Antitumor effect of KX-01 through inhibiting Src family kinases and mitosis. Cancer Res Treat. 2017;49:643–55.
Wong SHM, Fang CM, Chuah LH, Leong CO, Ngai SC. E-cadherin: Its dysregulation in carcinogenesis and clinical implications. Crit Rev Oncol Hematol. 2018;121:11–22.
Loh CY, Chai JY, Tang TF, Wong WF, Sethi G, Shanmugam MK, et al. The E-Cadherin and N-Cadherin switch in epithelial-to-mesenchymal transition: signaling, therapeutic implications, and challenges. Cells. 2019;8:1118.
Yao L, Zhang D, Zhao X, Sun B, Liu Y, Gu Q, et al. Dickkopf-1-promoted vasculogenic mimicry in non-small cell lung cancer is associated with EMT and development of a cancer stem-like cell phenotype. J Cell Mol Med. 2016;20:1673–85.
Zhang P, Li S, Lv C, Si J, Xiong Y, Ding L, et al. BPI-9016M, a c-Met inhibitor, suppresses tumor cell growth, migration and invasion of lung adenocarcinoma via miR203-DKK1. Theranostics. 2018;8:5890–902.
Pang H, Ma N, Shen W, Zhao Q, Wang J, Duan L, et al. Effects of DKK1 overexpression on bone metastasis of SBC-3 cells. Oncol Lett. 2018;15:6739–44.
Song Q, Liu H, Li C, Liang H. miR-33a-5p inhibits the progression of esophageal cancer through the DKK1-mediated Wnt/β-catenin pathway. Aging (Albany NY). 2021;13:20481–94.
Huang CF, Lira C, Chu K, Bilen MA, Lee YC, Ye X, et al. Cadherin-11 increases migration and invasion of prostate cancer cells and enhances their interaction with osteoblasts. Cancer Res. 2010;70:4580–9.
Chu K, Cheng CJ, Ye X, Lee YC, Zurita AJ, Chen DT, et al. Cadherin-11 promotes the metastasis of prostate cancer cells to bone. Mol Cancer Res. 2008;6:1259–67.
Lee YC, Cheng CJ, Huang M, Bilen MA, Ye X, Navone NM, et al. Androgen depletion up-regulates cadherin-11 expression in prostate cancer. J Pathol. 2010;221:68–76.
Seo SH, Cho KJ, Park HJ, Kim H, Lee HW, Kim BK, et al. Dickkopf-1 promotes angiogenesis by upregulating VEGF receptor 2-mediated mTOR/p70S6K signaling in hepatocellular carcinoma. Am J Cancer Res. 2021;11:4788–806.
Yang RH, Qin J, Cao JL, Zhang MZ, Li YY, Wang MQ, et al. Dickkopf-1 drives tumor immune evasion by inducing PD-L1 expression in hepatocellular carcinoma. Biochem Pharmacol. 2023;208:115378.
Fradet A, Sorel H, Depalle B, Serre CM, Farlay D, Turtoi A, et al. A new murine model of osteoblastic/osteolytic lesions from human androgen-resistant prostate cancer. PLoS ONE. 2013;8:e75092.
Cui N, Hu M, Khalil RA. Biochemical and biological attributes of matrix metalloproteinases. Prog Mol Biol Transl Sci. 2017;147:1–73.
Mahmood N, Rabbani SA. Fibrinolytic system and cancer: diagnostic and therapeutic applications. Int J Mol Sci. 2021;22:4358.
Longstreth JH, Wang K. The role of fibronectin in mediating cell migration. Am J Physiol Cell Physiol. 2024. https://doi.org/10.1152/ajpcell.00633.2023
Jiang Y, Wang M, Celiker MY, Liu YE, Sang QX, Goldberg ID, et al. Stimulation of mammary tumorigenesis by systemic tissue inhibitor of matrix metalloproteinase 4 gene delivery. Cancer Res. 2001;61:2365–70.
Han J, Jing Y, Han F, Sun P. Comprehensive analysis of expression, prognosis and immune infiltration for TIMPs in glioblastoma. BMC Neurol. 2021;21:447.
Janiak M, Paskal W, Rak B, Garbicz F, Jarema R, Sikora K, et al. TIMP4 expression is regulated by miR-200b-3p in prostate cancer cells. APMIS. 2017;125:101–5.
Rorive S, Lopez XM, Maris C, Trepant AL, Sauvage S, Sadeghi N, et al. TIMP-4 and CD63: new prognostic biomarkers in human astrocytomas. Mod Pathol. 2010;23:1418–28.
Chu T, Teng J, Jiang L, Zhong H, Han B. Lung cancer-derived Dickkopf1 is associated with bone metastasis and the mechanism involves the inhibition of osteoblast differentiation. Biochem Biophys Res Commun. 2014;443:962–8.
Zhuang X, Zhang H, Li X, Li X, Cong M, Peng F, et al. Differential effects on lung and bone metastasis of breast cancer by Wnt signalling inhibitor DKK1. Nat Cell Biol. 2017;19:1274–85.
Doucet D, Brubaker C, Turner D, Gregory CA. Factors affecting the role of canonical Wnt inhibitor Dickkopf-1 in cancer progression. Front Oncol. 2023;13:1114822.
Goldstein SD, Trucco M, Guzman WB, Hayashi M, Loeb DM. A monoclonal antibody against the Wnt signaling inhibitor dickkopf-1 inhibits osteosarcoma metastasis in a preclinical model. Oncotarget. 2016;7:21114–23.
Betella I, Turbitt WJ, Szul T, Wu B, Martinez AA, Arend RC, et al. Dkn-01: a promising strategy for targeting the Wnt pathway in ovarian cancer. Gynecol Oncol. 2019;154:55.
Betella I, Turbitt WJ, Szul T, Wu B, Martinez A, Katre A, et al. Wnt signaling modulator DKK1 as an immunotherapeutic target in ovarian cancer. Gynecol Oncol. 2020;157:765–74.
Acknowledgements
The authors thank Chiara Pighi for production of Luc/GFP PC3 expressing cells, and Federica Antico for technical support in histology. Funding: This work was supported by Fondazione CRT (grant number = RF 2020.1694), Turin, Italy to EA; and by “Research Fund ex-60%”, University of Turin, Turin, Italy to MGC.
Funding
This work was supported by Fondazione CRT (grant Number= RF 2020.1694), Turin, Italy to EA; and by “Research Fund ex-60%”, University of Turin, Turin, Italy to MGC.
Author information
Authors and Affiliations
Contributions
Conceptualization: LR, MC, CG, PD, BB, MGC. Investigation: LR, GF, MC, CG, MC, FM, OB, EV, LD. Analysis of the data: LR, GF, CG, MGC. Drafting of the manuscript: LR, GF, MC, CG, MGC. Critically revising of the manuscript: PD, BB, EA, MCG. Supervision: EA, MGC. Funding: EA, MGC.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
Animal studies were conducted in accordance with the Italian National Institute of Health Guide for the Care and Use of Laboratory Animals (authorization n° 521/2021-PR, July 19, 2021). Mice were housed according to the guidelines of the Federation of European Laboratory Animal Science Association and the Ethical Committee of the University of Turin.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rinella, L., Fiorentino, G., Compagno, M. et al. Dickkopf-1 (DKK1) drives growth and metastases in castration-resistant prostate cancer. Cancer Gene Ther (2024). https://doi.org/10.1038/s41417-024-00783-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41417-024-00783-7