Abstract
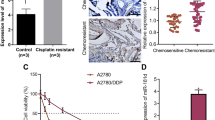
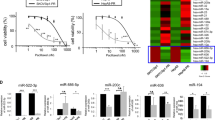
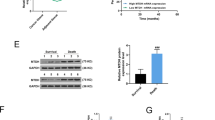
Our previous study confirmed that miR-219-5p inhibits the progression of ovarian cancer (OC) by targeting high mobility group AT-hook 2 (HMGA2), while the role of miR-219-5p on the chemoresistance of OC is unclear. HMGA2 and miR-219-5p expression in OC tumors and various types of OC cells were determined by reverse transcription-quantitative PCR (RT-qPCR) and western blotting. The miRNA profiles in A2780 and cisplatin-resistant A2780 cells were investigated via bulk miRNA sequencing, and the interactions of miR-219-5p and HMGA2 were determined by luciferase reporter activity assay. Cell function was verified through Cell Counting Kit-8, invasion assay, wound-healing, and TUNEL assays. HMGA2 level is highly expressed in cisplatin-resistant OC cell lines compared to normal OC cells, while the expression trend of miR-219-5p is the opposite. In addition, we found that miR-219-5p is one of the miRNAs that have the most significant reduction in levels in the cisplatin-resistant A2780/DDP cell line compared to A2780 cells. Then, we reveal that miR-219-5p directly targets HMGA2 in cisplatin-resistant OC cells, and upregulation of miR-219-5p significantly reduces the resistance of OC cells to cisplatin both in vitro and in vivo. Finally, our results suggest that Wnt/β-catenin signaling and autophagy pathway is involved in the role of miR-219-5p/HMGA2 on resistance of OC cells to cisplatin via gain-of-function experiments. Collectively, the present study shows that miR-219-5p decreases the resistance of OC cells to cisplatin via Wnt/β-catenin signaling and autophagy by regulating HMGA2, which provides a feasible solution for the resistance of OC to chemotherapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The original contributions presented in the study are included in the article. Further inquiries can be directed to the corresponding authors.
References
Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2021. CA Cancer J Clin. 2021;71:7–33.
Lheureux S, Gourley C, Vergote I, Oza AM. Epithelial ovarian cancer. Lancet. 2019;393:1240–53.
Jelovac D, Armstrong DK. Recent progress in the diagnosis and treatment of ovarian cancer. CA Cancer J Clin. 2011;61:183–203.
Ozols RF, Bundy BN, Greer BE, Fowler JM, Clarke-Pearson D, Burger RA, et al. Phase III trial of carboplatin and paclitaxel compared with cisplatin and paclitaxel in patients with optimally resected stage III ovarian cancer: a Gynecologic Oncology Group study. J Clin Oncol. 2003;21:3194–200.
Zhou X, Chada K. HMGI family proteins: architectural transcription factors in mammalian development and cancer. Keio J Med. 1998;47:73–7.
Fusco A, Fedele M. Roles of HMGA proteins in cancer. Nat Rev Cancer. 2007;7:899–910.
Hammond SM, Sharpless NE. HMGA2, microRNAs, and stem cell aging. Cell. 2008;135:1013–6.
Wu J, Zhang S, Shan J, Hu Z, Liu X, Chen L, et al. Elevated HMGA2 expression is associated with cancer aggressiveness and predicts poor outcome in breast cancer. Cancer Lett. 2016;376:284–92.
Palumbo A Jr, Da Costa NM, Esposito F, De Martino M, D’Angelo D, de Sousa VP, et al. HMGA2 overexpression plays a critical role in the progression of esophageal squamous carcinoma. Oncotarget. 2016;7:25872–84.
Wu J, Wang Y, Xu X, Cao H, Sahengbieke S, Sheng H, et al. Transcriptional activation of FN1 and IL11 by HMGA2 promotes the malignant behavior of colorectal cancer. Carcinogenesis. 2016;37:511–21.
Agostini A, Brunetti M, Davidson B, Trope CG, Heim S, Panagopoulos I, et al. Genomic imbalances are involved in miR-30c and let-7a deregulation in ovarian tumors: implications for HMGA2 expression. Oncotarget. 2017;8:21554–60.
Xing F, Song Z, He Y. MiR-219-5p inhibits growth and metastasis of ovarian cancer cells by targeting HMGA2. Biol Res. 2018;51:50.
Ferrarelli LK. Treating WNT-driven colorectal cancer. Science. 2017;356:1346–8.
Matsuzaki S, Botchorishvili R, Pouly JL, Canis M. Targeting the Wnt/beta-catenin pathway in endometriosis: a potentially effective approach for treatment and prevention. Mol Cell Ther. 2014;2:36.
Coopes A, Henry CE, Llamosas E, Ford CE. An update of Wnt signalling in endometrial cancer and its potential as a therapeutic target. Endocr Relat Cancer. 2018;ERC-18-0112.
Huang M, Zhang D, Wu JY, Xing K, Yeo E, Li C et al. Wnt-mediated endothelial transformation into mesenchymal stem cell-like cells induces chemoresistance in glioblastoma. Sci Transl Med. 2020;12:eaay7522.
Wickstrom M, Dyberg C, Milosevic J, Einvik C, Calero R, Sveinbjornsson B, et al. Wnt/beta-catenin pathway regulates MGMT gene expression in cancer and inhibition of Wnt signalling prevents chemoresistance. Nat Commun. 2015;6:8904.
Zhang SF, Wang XY, Fu ZQ, Peng QH, Zhang JY, Ye F, et al. TXNDC17 promotes paclitaxel resistance via inducing autophagy in ovarian cancer. Autophagy. 2015;11:225–38.
Zhou C, Yi C, Yi Y, Qin W, Yan Y, Dong X, et al. LncRNA PVT1 promotes gemcitabine resistance of pancreatic cancer via activating Wnt/beta-catenin and autophagy pathway through modulating the miR-619-5p/Pygo2 and miR-619-5p/ATG14 axes. Mol Cancer. 2020;19:118.
Karasic TB, O’Hara MH, Loaiza-Bonilla A, Reiss KA, Teitelbaum UR, Borazanci E, et al. Effect of gemcitabine and nab-paclitaxel with or without hydroxychloroquine on patients with advanced pancreatic cancer: a phase 2 randomized clinical trial. JAMA Oncol. 2019;5:993–8.
Oronsky B, Ray CM, Spira AI, Trepel JB, Carter CA, Cottrill HM. A brief review of the management of platinum-resistant-platinum-refractory ovarian cancer. Med Oncol. 2017;34:103.
Yu T, Guo F, Yu Y, Sun T, Ma D, Han J, et al. Fusobacterium nucleatum promotes chemoresistance to colorectal cancer by modulating autophagy. Cell. 2017;170:548–63. e16
Menon U, Gentry-Maharaj A, Burnell M, Singh N, Ryan A, Karpinskyj C, et al. Ovarian cancer population screening and mortality after long-term follow-up in the UK Collaborative Trial of Ovarian Cancer Screening (UKCTOCS): a randomised controlled trial. Lancet. 2021;397:2182–93.
Kleppe M, van der Aa MA, Van Gorp T, Slangen BF, Kruitwagen RF. The impact of lymph node dissection and adjuvant chemotherapy on survival: a nationwide cohort study of patients with clinical early-stage ovarian cancer. Eur J Cancer. 2016;66:83–90.
Cocco E, Deng Y, Shapiro EM, Bortolomai I, Lopez S, Lin K, et al. Dual-targeting nanoparticles for in vivo delivery of suicide genes to chemotherapy-resistant ovarian cancer cells. Mol Cancer Ther. 2017;16:323–33.
Cheng J, Deng R, Zhang P, Wu C, Wu K, Shi L, et al. miR-219-5p plays a tumor suppressive role in colon cancer by targeting oncogene Sall4. Oncol Rep. 2015;34:1923–32.
Wang Q, Zhu L, Jiang Y, Xu J, Wang F, He Z. miR-219-5p suppresses the proliferation and invasion of colorectal cancer cells by targeting calcyphosin. Oncol Lett. 2017;13:1319–24.
Ting G, Li X, Kwon HY, Ding T, Zhang Z, Chen Z, et al. microRNA-219-5p targets NEK6 to inhibit hepatocellular carcinoma progression. Am J Transl Res. 2020;12:7528–41.
Huang WT, Zhang H, Jin Z, Li K, Hu C, Li ML, et al. MiR-219-5p inhibits prostate cancer cell growth and metastasis by targeting HMGA2. Eur Rev Med Pharm Sci. 2020;24:4710–8.
Ye Q, Wang X, Yuan M, Cui S, Chen Y, Hu Z et al. miR-219-5p targets TBXT and inhibits breast cancer cell EMT and cell migration and invasion. Biosci Rep 2021; 41:BSR20210318.
Li X, Sun L, Chen L, Xu Y, Kong X. Upregulation of microRNA-219-5p relieves ulcerative colitis through balancing the differentiation of Treg/Th17 cells. Eur J Gastroenterol Hepatol. 2020;32:813–20.
Lou W, Zhang X, Hu XY, Hu AR. MicroRNA-219-5p inhibits morphine-induced apoptosis by targeting key cell cycle regulator WEE1. Med Sci Monit. 2016;22:1872–9.
Wei JJ, Wu J, Luan C, Yeldandi A, Lee P, Keh P, et al. HMGA2: a potential biomarker complement to P53 for detection of early-stage high-grade papillary serous carcinoma in fallopian tubes. Am J Surg Pathol. 2010;34:18–26.
Mahajan A, Liu Z, Gellert L, Zou X, Yang G, Lee P, et al. HMGA2: a biomarker significantly overexpressed in high-grade ovarian serous carcinoma. Mod Pathol. 2010;23:673–81.
Hristov AC, Cope L, Reyes MD, Singh M, Iacobuzio-Donahue C, Maitra A, et al. HMGA2 protein expression correlates with lymph node metastasis and increased tumor grade in pancreatic ductal adenocarcinoma. Mod Pathol. 2009;22:43–9.
Shell S, Park SM, Radjabi AR, Schickel R, Kistner EO, Jewell DA, et al. Let-7 expression defines two differentiation stages of cancer. Proc Natl Acad Sci USA. 2007;104:11400–5.
Nishino J, Kim I, Chada K, Morrison SJ. Hmga2 promotes neural stem cell self-renewal in young but not old mice by reducing p16Ink4a and p19Arf Expression. Cell. 2008;135:227–39.
Watanabe S, Ueda Y, Akaboshi S, Hino Y, Sekita Y, Nakao M. HMGA2 maintains oncogenic RAS-induced epithelial-mesenchymal transition in human pancreatic cancer cells. Am J Pathol. 2009;174:854–68.
Malek A, Bakhidze E, Noske A, Sers C, Aigner A, Schafer R, et al. HMGA2 gene is a promising target for ovarian cancer silencing therapy. Int J Cancer. 2008;123:348–56.
Li VS, Ng SS, Boersema PJ, Low TY, Karthaus WR, Gerlach JP, et al. Wnt signaling through inhibition of beta-catenin degradation in an intact Axin1 complex. Cell. 2012;149:1245–56.
Clevers H, Nusse R. Wnt/beta-catenin signaling and disease. Cell. 2012;149:1192–205.
Petherick KJ, Williams AC, Lane JD, Ordonez-Moran P, Huelsken J, Collard TJ, et al. Autolysosomal beta-catenin degradation regulates Wnt-autophagy-p62 crosstalk. EMBO J. 2013;32:1903–16.
Funding
This study was supported by grants from the project of Shanghai Zhabei Central Hospital construction of important and weak medical disciplines (2021BR03).
Author information
Authors and Affiliations
Contributions
Concepts: F.X., Z.S., and S.W.; resources: F.X. and S.W.; experiments and data analysis: Z.S., L.Y., X.X., X.S., and S.T.; bioinformatics and manuscript preparation: C.L. and F.X.; revision and editing: Z.S., F.X., and S.W. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethics statement
This study received approval from the Ethics Committee of Shanghai Zhabei Central Hospital, and all patients and participants signed informed consent. The procedures involving animals were approved by the Institutional Animal Care and Use Committee (IACUC) of Tongji University.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Song, Z., Liao, C., Yao, L. et al. miR-219-5p attenuates cisplatin resistance of ovarian cancer by inactivating Wnt/β-catenin signaling and autophagy via targeting HMGA2. Cancer Gene Ther 30, 596–607 (2023). https://doi.org/10.1038/s41417-022-00574-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41417-022-00574-y