Abstract
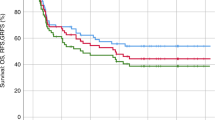
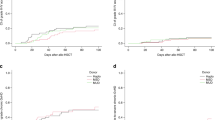
While post-transplant cyclophosphamide (PTCy) is commonly used as graft-versus-host disease (GvHD) prophylaxis in haploidentical stem cell transplantation (haplo-HSCT), its dose remains a matter of debate due to side effect concerns. Standard dose of 100 mg/kg associated with tacrolimus and post-engraftment anti-thymocyte globulin (ATG) was used as the reference GvHD prophylaxis in our center and had demonstrated encouraging results. Though PTCy 80 mg/kg was shown to be feasible in patients in reduced-intensity conditioning, whether it exerts equivalent GvHD prophylactic efficacy in myeloablative conditioning (MAC) setting has not been confirmed. Here, we retrospectively analyzed the efficacy and safety of PTCy 80 mg/kg combined with tacrolimus and post-engraftment ATG as GvHD prophylaxis in patients aged more than 55 years or with cardiac antecedents or HCT-CI score >2 undergoing haplo-HSCT with MAC. The cumulative incidence of grade III-IV aGvHD at day 100 and moderate-to-severe cGvHD at 1 year was 4.8% ± 3.4% and 19.9% ± 7.0%, respectively. When compared with patients receiving the reference regimen, patients from the PTCy 80 mg/kg group had similar incidence of GvHDs and survival as their younger counterparts. Thus, PTCy 80 mg/kg seems to be feasible for patients treated with MAC conditioning regimens in haplo-HSCT, inviting further investigation notably in frail patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Ruggeri A, Sun Y, Labopin M, Bacigalupo A, Lorentino F, Arcese W, et al. Post-transplant cyclophosphamide versus anti-thymocyte globulin as graft- versus-host disease prophylaxis in haploidentical transplant. Haematologica. 2017;102:401–10. https://doi.org/10.3324/haematol.2016.151779.
Shaw BE, Jimenez-Jimenez AM, Burns LJ, Logan BR, Khimani F, Shaffer BC, et al. National Marrow Donor Program-sponsored multicenter, phase II trial of HLA-mismatched unrelated donor bone marrow transplantation using post-transplant cyclophosphamide. J Clin Oncol. 2021;39:1971–82. https://doi.org/10.1200/JCO.20.03502.
Sanz J, Galimard JE, Labopin M, Afanasyev B, Angelucci E, Ciceri F, et al. Post-transplant cyclophosphamide after matched sibling, unrelated and haploidentical donor transplants in patients with acute myeloid leukemia: a comparative study of the ALWP EBMT. J Hematol Oncol. 2020;13:46. https://doi.org/10.1186/s13045-020-00882-6.
Perez-Valencia AI, Cascos E, Carbonell-Ordeig S, Charry P, Gomez-Hernando M, Rodriguez-Lobato LG, et al. Incidence, risk factors, and impact of early cardiac toxicity after allogeneic hematopoietic cell transplant. Blood Adv. 2023;7:2018–31. https://doi.org/10.1182/bloodadvances.2022008792.
Dulery R, Mohty R, Labopin M, Sestili S, Malard F, Brissot E, et al. Early cardiac toxicity associated with post-transplant cyclophosphamide in allogeneic stem cell transplantation. JACC CardioOncol. 2021;3:250–9. https://doi.org/10.1016/j.jaccao.2021.02.011.
Yeh J, Whited L, Saliba RM, Rondon G, Banchs J, Shpall E, et al. Cardiac toxicity after matched allogeneic hematopoietic cell transplant in the posttransplant cyclophosphamide era. Blood Adv. 2021;5:5599–607. https://doi.org/10.1182/bloodadvances.2021004846.
Ngo D, Samuels D, Chen J, Koller PB, Al Malki MM. A clinical review of the different strategies to minimize hemorrhagic cystitis associated with the use of post-transplantation cyclophosphamide in an allogeneic transplant. Transpl Cell Ther. 2022;28:349–54. https://doi.org/10.1016/j.jtct.2022.05.012.
Dulery R, Goudet C, Mannina D, Bianchessi A, Granata A, Harbi S, et al. Reduced post-transplant cyclophosphamide doses in haploidentical hematopoietic cell transplantation for elderly patients with hematological malignancies. Bone Marrow Transpl. 2023;58:386–92. https://doi.org/10.1038/s41409-022-01908-y.
Dulery R, Malard F, Brissot E, Banet A, Sestili S, Belhocine R et al. Reduced post-transplant cyclophosphamide dose with antithymocyte globulin in peripheral blood stem cell haploidentical transplantation. Bone Marrow Transpl. 2023. https://doi.org/10.1038/s41409-023-02085-2.
Spyridonidis A, Labopin M, Savani BN, Niittyvuopio R, Blaise D, Craddock C, et al. Redefining and measuring transplant conditioning intensity in current era: a study in acute myeloid leukemia patients. Bone Marrow Transpl. 2020;55:1114–25. https://doi.org/10.1038/s41409-020-0803-y.
Armand P, Kim HT, Logan BR, Wang Z, Alyea EP, Kalaycio ME, et al. Validation and refinement of the Disease Risk Index for allogeneic stem cell transplantation. Blood. 2014;123:3664–71. https://doi.org/10.1182/blood-2014-01-552984.
Wang L, Dai B, Gao W, Wang J, Wan M, Wang R, et al. Clinical significance of haplo-fever and cytokine profiling after graft infusion in allogeneic stem cell transplantation from haplo-identical donors. Front Med. 2022;9:820591. https://doi.org/10.3389/fmed.2022.820591.
Law AD, Salas MQ, Lam W, Michelis FV, Thyagu S, Kim DDH, et al. Reduced-intensity conditioning and dual T lymphocyte suppression with antithymocyte globulin and post-transplant cyclophosphamide as graft-versus-host disease prophylaxis in haploidentical hematopoietic stem cell transplants for hematological malignancies. Biol Blood Marrow Transpl. 2018;24:2259–64. https://doi.org/10.1016/j.bbmt.2018.07.008.
Alanazi W, Chen S, Lipton JH, Kim DD, Viswabandya A, Kumar R, et al. Post-transplant cyclophosphamide combined with anti-thymocyte globulin as graft-versus-host disease prophylaxis for allogeneic hematopoietic cell transplantation in high-risk acute myeloid leukemia and myelodysplastic syndrome. Acta Haematol. 2021;144:66–73. https://doi.org/10.1159/000507536.
Dulery R, Brissot E, Mohty M. Combining post-transplant cyclophosphamide with antithymocyte globulin for graft-versus-host disease prophylaxis in hematological malignancies. Blood Rev. 2023: 101080. https://doi.org/10.1016/j.blre.2023.101080.
Zu Y, Li Z, Gui R, Liu Y, Zhang Y, Yu F, et al. Low-dose post-transplant cyclophosphamide with low-dose antithymocyte globulin for prevention of graft-versus-host disease in first complete remission undergoing 10/10 HLA-matched unrelated donor peripheral blood stem cell transplants: a multicentre, randomized controlled trial. Bone Marrow Transpl. 2022;57:1573–80. https://doi.org/10.1038/s41409-022-01754-y.
Spyridonidis A, Labopin M, Brissot E, Moiseev I, Cornelissen J, Choi G, et al. Should anti-thymocyte globulin be added in post-transplant cyclophosphamide based matched unrelated donor peripheral blood stem cell transplantation for acute myeloid leukemia? A study on behalf of the Acute Leukemia Working Party of the EBMT. Bone Marrow Transpl. 2022;57:1774–80. https://doi.org/10.1038/s41409-022-01816-1.
Zhang W, Gui R, Zu Y, Zhang B, Li Z, Zhang Y, et al. Reduced-dose post-transplant cyclophosphamide plus low-dose post-transplant anti-thymocyte globulin as graft-versus-host disease prophylaxis with fludarabine-busulfan-cytarabine conditioning in haploidentical peripheral blood stem cell transplantation: a multicentre, randomized controlled clinical trial. Br J Haematol. 2023;200:210–21. https://doi.org/10.1111/bjh.18483.
Xue E, Lorentino F, Lupo Stanghellini MT, Giglio F, Piemontese S, Clerici DT, et al. Addition of a single low dose of anti T-lymphocyte globulin to post-transplant cyclophosphamide after allogeneic hematopoietic stem cell transplant: a pilot study. J Clin Med. 2022;11. https://doi.org/10.3390/jcm11041106.
Gao WH, Zhu JY, Wang LN, Wan M, Wang L, Devillier R, et al. Post-transplantation cyclophosphamide combined with tacrolimus and low-dose post-engraftment anti-thymoglobulin as GVHD prophylaxis for patients undergoing peripheral blood stem cell transplantation from haploidentical family donor: A single center analysis. Front Med. 2023;10:1140217. https://doi.org/10.3389/fmed.2023.1140217.
Author information
Authors and Affiliations
Contributions
Wang Lining and Xu Guilin performed the transplants in the patients, collected patient data, and wrote the manuscript. Wang Lining, Gao Wenhui and Wan Ming performed statistical analysis. Wang Ling and Jiang Jieling supervised the transplants and followed up the patients. Blaise Didier and Hu Jiong designed and supervised the study and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, L., Xu, G., Wang, L. et al. Post-transplant cyclophosphamide at 80 mg/kg with low dose post-engraftment anti-thymocyte globulin in haploidentical transplantation with myeloablative conditioning. Bone Marrow Transplant (2024). https://doi.org/10.1038/s41409-024-02277-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41409-024-02277-4