Abstract
Background
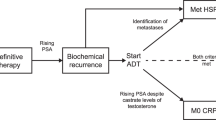
With the availability of second-generation androgen receptor inhibitors (SGARIs), the treatment landscape has changed dramatically for patients with nonmetastatic castration-resistant prostate cancer (nmCRPC). In clinical trials, the SGARIs (apalutamide, enzalutamide, darolutamide) increased metastasis-free survival (MFS), overall survival (OS), and patient quality of life compared to placebo. These drugs were subsequently integrated into nmCRPC clinical practice guidelines. With advances in radiographic imaging, disease assessment, and patient monitoring, nmCRPC strategies are evolving to address limitations related to tracking disease progression using prostate-specific antigen (PSA) kinetics.
Methods
A panel of 10 multidisciplinary experts in prostate cancer conducted reviews and discussions of unmet needs in the management and monitoring of patients with nmCRPC in order to develop consensus recommendations.
Results
Across the SGARI literature, patient MFS and OS are generally comparable for all treatments, but important distinctions exist regarding short- and long-term drug safety profiles and drug-drug interactions. With respect to disease monitoring, a substantial proportion of patients using SGARIs may experience disease progression without rising PSA levels, suggesting a need for enhanced radiographic imaging in addition to PSA monitoring. Recent data also indicate that novel prostate-specific membrane antigen positron emission tomography radiotracers provide enhanced accuracy for disease detection, as compared to conventional imaging.
Conclusions
Clinical decision-making in nmCRPC has become more complex, with new opportunities to apply precision medicine to patient care. Multidisciplinary teams can ensure that patients with nmCRPC receive optimal and individualized disease management.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Change history
20 April 2024
A Correction to this paper has been published: https://doi.org/10.1038/s41391-024-00835-x
References
National Cancer Institute (NCI). Cancer stat facts: prostate cancer. Surveillance, Epidemiology, and End Results Program (SEER). https://seer.cancer.gov/statfacts/html/prost.html.
Berruti A, Bracarda S, Caffo O, Cortesi E, D’Angelillo R, Del Re M, et al. nmCRPC, a look in the continuous care of prostate cancer patients: state of art and future perspectives. Cancer Treat Rev. 2023;115:102525.
NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®). Prostate Cancer. Version 1.2023. September, 2022. https://www.nccn.org/professionals/physician_gls/pdf/prostate.pdf.
Rice MA, Malhotra SV, Stoyanova T. Second-generation antiandrogens: from discovery to standard of care in castration resistant prostate cancer. Front Oncol. 2019;9:801.
Smith MR, Saad F, Oudard S, Shore N, Fizazi K, Sieber P, et al. Denosumab and bone metastasis-free survival in men with nonmetastatic castration-resistant prostate cancer: exploratory analyses by baseline prostate-specific antigen doubling time. J Clin Oncol. 2013;31:3800–6.
Scher HI, Morris MJ, Stadler WM, Higano C, Basch E, Fizazi K, et al. Trial design and objectives for castration-resistant prostate cancer: updated recommendations from the prostate cancer clinical trials working group 3. J Clin Oncol. 2016;34:1402–18.
Saad F, Bogemann M, Suzuki K, Shore N. Treatment of nonmetastatic castration-resistant prostate cancer: focus on second-generation androgen receptor inhibitors. Prostate Cancer Prostatic Dis. 2021;24:323–34.
ERLEADA®. Prescribing information. Janssen Products, LP. 2023. https://www.janssenlabels.com/package-insert/product-monograph/prescribing-information/ERLEADA-pi.pdf.
NUBEQA®. Prescribing information. Bayer HealthCare Pharmaceuticals Inc. 2022. https://labeling.bayerhealthcare.com/html/products/pi/Nubeqa_PI.pdf?inline=.
XTANDI®. Prescribing information. Astellas Pharma Inc. 2022. https://www.astellas.us/docs/us/12A005-ENZ-WPI.pdf.
Lowrance W, Dreicer R, Jarrard DF, Scarpato KR, Kim SK, Kirkby E, et al. Updates to advanced prostate cancer: AUA/SUO guideline (2023). J Urol. (2023). https://doi.org/10.1097/JU.0000000000003452.
Parker C, Castro E, Fizazi K, Heidenreich A, Ost P, Procopio G, et al. Prostate cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2020;31:1119–34.
Hussain M, Fizazi K, Saad F, Rathenborg P, Shore N, Ferreira U, et al. Enzalutamide in men with nonmetastatic, castration-resistant prostate cancer. N Engl J Med. 2018;378:2465–74.
Smith MR, Saad F, Chowdhury S, Oudard S, Hadaschik BA, Graff JN, et al. Apalutamide treatment and metastasis-free survival in prostate cancer. N Engl J Med. 2018;378:1408–18.
Fizazi K, Shore N, Tammela TL, Ulys A, Vjaters E, Polyakov S, et al. Darolutamide in nonmetastatic, castration-resistant prostate cancer. N Engl J Med. 2019;380:1235–46.
Fizazi K, Shore N, Tammela TL, Ulys A, Vjaters E, Polyakov S, et al. Nonmetastatic, castration-resistant prostate cancer and survival with darolutamide. N Engl J Med. 2020;383:1040.
Smith MR, Saad F, Chowdhury S, Oudard S, Hadaschik BA, Graff JN, et al. Apalutamide and overall survival in prostate cancer. Eur Urol. 2021;79:150–8.
Sternberg CN, Fizazi K, Saad F, Shore ND, De Giorgi U, Penson DF, et al. Enzalutamide and survival in nonmetastatic, castration-resistant prostate cancer. N Engl J Med. 2020;382:2197–206.
Oudard S, Hadaschik B, Saad F, Cella D, Basch E, Graff JN, et al. Health-related quality of life at the SPARTAN final analysis of apalutamide for nonmetastatic castration-resistant prostate cancer patients receiving androgen deprivation therapy. Eur Urol Focus. 2022;8:958–67.
Saad F, Cella D, Basch E, Hadaschik BA, Mainwaring PN, Oudard S, et al. Effect of apalutamide on health-related quality of life in patients with non-metastatic castration-resistant prostate cancer: an analysis of the SPARTAN randomised, placebo-controlled, phase 3 trial. Lancet Oncol. 2018;19:1404–16.
Smith MR, Shore N, Tammela TL, Ulys A, Vjaters E, Polyakov S, et al. Darolutamide and health-related quality of life in patients with non-metastatic castration-resistant prostate cancer: an analysis of the phase III ARAMIS trial. Eur J Cancer. 2021;154:138–46.
Tombal B, Saad F, Penson D, Hussain M, Sternberg CN, Morlock R, et al. Patient-reported outcomes following enzalutamide or placebo in men with non-metastatic, castration-resistant prostate cancer (PROSPER): a multicentre, randomised, double-blind, phase 3 trial. Lancet Oncol. 2019;20:556–69.
Xie W, Regan MM, Buyse M, Halabi S, Kantoff PW, Sartor O, et al. Metastasis-free survival is a strong surrogate of overall survival in localized prostate cancer. J Clin Oncol. 2017;35:3097–104.
U.S. Department of Health and Human Services, Food and Drug Administration (FDA), Center for Drug Evaluation and Research (CDER), Center for Biologics Evaluation and Research (CBER). Nonmetastatic castration-resistant prostate cancer: considerations for metastasis-free survival endpoint in clinical trials — Guidance for Industry. August 2021. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/nonmetastatic-castration-resistant-prostate-cancer-considerations-metastasis-free-survival-endpoint.
Brave M, Weinstock C, Brewer JR, Chi DC, Suzman DL, Cheng J, et al. An FDA review of drug development in nonmetastatic castration-resistant prostate cancer. Clin Cancer Res. 2020;26:4717–22.
Beaver JA, Kluetz PG, Pazdur R. Metastasis-free survival - a new end point in prostate cancer trials. N. Engl J Med. 2018;378:2458–60.
Cattrini C, Caffo O, De Giorgi U, Mennitto A, Gennari A, Olmos D, et al. Apalutamide, darolutamide and enzalutamide for nonmetastatic castration-resistant prostate cancer (nmCRPC): a critical review. Cancers (Basel). 2022;14.
Shore ND, Luz MdA, Ulys A, Ortiz JA, Srinivasan S, Kurland E, et al. Long-term safety and tolerability of darolutamide and duration of treatment in patients with nonmetastatic castration-resistant prostate cancer (nmCRPC) from the ARAMIS rollover study. J Clin Oncol. 2023;41:147.
Halabi S, Jiang S, Terasawa E, Garcia-Horton V, Ayyagari R, Waldeck AR, et al. Indirect comparison of darolutamide versus apalutamide and enzalutamide for nonmetastatic castration-resistant prostate cancer. J Urol. 2021;206:298–307.
Maggi M, Salciccia S, Del Giudice F, Busetto GM, Falagario UG, Carrieri G, et al. A systematic review and meta-analysis of randomized controlled trials with novel hormonal therapies for non-metastatic castration-resistant prostate cancer: an update from mature overall survival data. Front Oncol. 2021;11:700258 https://doi.org/10.3389/fonc.2021.700258
Mulati Y, Fan Y, Yu W, Zhang Q, He Z. Novel androgen receptor inhibitors in non-metastatic, castration-resistant prostate cancer: a systematic review and network meta-analysis. Front Oncol. 2021;11:733202 https://doi.org/10.3389/fonc.2021.733202
Benoist GE, van Oort IM, Smeenk S, Javad A, Somford DM, Burger DM, et al. Drug-drug interaction potential in men treated with enzalutamide: mind the gap. Br J Clin Pharm. 2018;84:122–9.
Conde-Estevez D, Henriquez I, Munoz-Rodriguez J, Rodriguez-Vida A. Treatment of non-metastatic castration-resistant prostate cancer: facing age-related comorbidities and drug-drug interactions. Expert Opin Drug Metab Toxicol. 2022;18:601–13.
Maher RL, Hanlon J, Hajjar ER. Clinical consequences of polypharmacy in elderly. Expert Opin Drug Saf. 2014;13:57–65.
Nightingale G, Skonecki E, Boparai MK. The impact of polypharmacy on patient outcomes in older adults with cancer. Cancer J. 2017;23:211–8.
Shi S, Klotz U. Age-related changes in pharmacokinetics. Curr Drug Metab. 2011;12:601–10.
Derendorf H, Schmidt S. Rowland and Tozer’s Clinical Pharmacokinetics and Pharmacodynamics: Concepts and Applications. Fifth edn. (Wolters Kluwer, 2020).
Mohamed MR, Mohile SG, Juba KM, Awad H, Wells M, Loh KP, et al. Association of polypharmacy and potential drug-drug interactions with adverse treatment outcomes in older adults with advanced cancer. Cancer. 2023;129:1096–104.
Ramsdale E, Mohamed M, Yu V, Otto E, Juba K, Awad H, et al. Polypharmacy, potentially inappropriate medications, and drug-drug interactions in vulnerable older adults with advanced cancer initiating cancer treatment. Oncologist. 2022;27:e580–e8.
Gillessen S, Bossi A, Davis ID, de Bono J, Fizazi K, James ND, et al. Management of patients with advanced prostate cancer. part I: intermediate-/high-risk and locally advanced disease, biochemical relapse, and side effects of hormonal treatment: report of the Advanced Prostate Cancer Consensus Conference 2022. Eur Urol. 2023;83:267–93.
Figueiredo A, Costa L, Mauricio MJ, Figueira L, Ramos R, Martins-da-Silva C. Nonmetastatic castration-resistant prostate cancer: current challenges and trends. Clin Drug Investig. 2022;42:631–42.
Gibbons JA, de Vries M, Krauwinkel W, Ohtsu Y, Noukens J, van der Walt JS, et al. Pharmacokinetic drug interaction studies with enzalutamide. Clin Pharmacokinet. 2015;54:1057–69.
Del Re M, Fogli S, Derosa L, Massari F, De Souza P, Crucitta S, et al. The role of drug-drug interactions in prostate cancer treatment: Focus on abiraterone acetate/prednisone and enzalutamide. Cancer Treat Rev. 2017;55:71–82. 20170309
Lopez-Campos F, Conde-Moreno A, Barrado Los Arcos M, Gomez-Caamano A, Garcia-Gomez R, Hervas Moron A. Treatment landscape of nonmetastatic castration-resistant prostate cancer: a window of opportunity. J Pers Med. 11, (2021).
CRESTOR. Prescribing information. AstraZeneca. 2023. https://den8dhaj6zs0e.cloudfront.net/50fd68b9-106b-4550-b5d0-12b045f8b184/fd6c9085-0722-46d7-b166-bd68f09c43d5/fd6c9085-0722-46d7-b166-bd68f09c43d5_viewable_rendition__v.pdf.
Crawford ED, Andriole G, Freedland SJ, Garnick M, Gomella LG, Henderson J, et al. Evolving understanding and categorization of prostate cancer: preventing progression to metastatic castration-resistant prostate cancer: RADAR IV. Can J Urol. 2020;27:10352–62.
Howard LE, Moreira DM, De Hoedt A, Aronson WJ, Kane CJ, Amling CL, et al. Thresholds for PSA doubling time in men with non-metastatic castration-resistant prostate cancer. BJU Int. 2017;120:E80–E6.
Bryce AH, Alumkal JJ, Armstrong A, Higano CS, Iversen P, Sternberg CN, et al. Radiographic progression with nonrising PSA in metastatic castration-resistant prostate cancer: post hoc analysis of PREVAIL. Prostate Cancer Prostatic Dis. 2017;20:221–7.
Morgans AK, Sweeney C, Wallis CJD, Halabi S, Armstrong AJ, Verholen F, et al. Progression patterns by types of metastatic spread, prostate-specific antigen (PSA), and clinical symptoms: post-hoc analyses of ARAMIS. J Clin Oncol. 2022;40:5044.
Saad F, Sternberg CN, Efstathiou E, Fizazi K, Modelska K, Lin X, et al. Prostate-specific antigen progression in enzalutamide-treated men with nonmetastatic castration-resistant prostate cancer: any rise in prostate-specific antigen may require closer monitoring. Eur Urol. 2020;78:847–53.
Crawford ED, Albala DM, Harris RG, Slovin SF, Bryce AH, Carroll PR, et al. A clinician’s guide to targeted precision imaging in patients with prostate cancer (RADAR VI). JU Open. 2023;1:e00003.
Crawford ED, Koo PJ, Shore N, Slovin SF, Concepcion RS, Freedland SJ, et al. A clinician’s guide to next generation imaging in patients with advanced prostate cancer (RADAR III). J Urol. 2019;201:682–92.
Esen B, Seymen H, Tarim K, Koseoglu E, Bolukbasi Y, Falay O, et al. Diagnostic performance of (68)Ga-PSMA-11 positron emission tomography/computed tomography to monitor treatment response in patients with metastatic prostate cancer: the concordance between biochemical response and prostate-specific membrane antigen results. Eur Urol Focus. 2023;9:832–7.
Fendler WP, Weber M, Iravani A, Hofman MS, Calais J, Czernin J, et al. Prostate-specific membrane antigen ligand positron emission tomography in men with nonmetastatic castration-resistant prostate cancer. Clin Cancer Res. 2019;25:7448–54.
Hofman MS, Lawrentschuk N, Francis RJ, Tang C, Vela I, Thomas P, et al. Prostate-specific membrane antigen PET-CT in patients with high-risk prostate cancer before curative-intent surgery or radiotherapy (proPSMA): a prospective, randomised, multicentre study. Lancet. 2020;395:1208–16.
Crawford ED, Harris RG, Slovin SF, Concepcion RS, Albala DM, Gomella LG, et al. Synthesizing and applying molecular targeted imaging results in patients with prostate cancer (RADAR VII). JU Open. 2023;1:e00011.
ClinicalTrials.gov. Darolutamide + consolidation radiotherapy in advanced prostate cancer detected by PSMA (DECREASE). ClinicalTrials.gov Identifier: NCT04319783. https://clinicaltrials.gov/ct2/show/NCT04319783.
ClinicalTrials.gov. A phase II study of AAA617 alone and AAA617 in combination with ARPI in patients with PSMA PET scan positive CRPC (PSMACare). ClinicalTrials.gov Identifier: NCT05849298. https://clinicaltrials.gov/ct2/show/NCT05849298?term=NCT05849298&draw=2&rank=1.
ClinicalTrials.gov. A study to compare darolutamide given with androgen deprivation therapy (ADT) with ADT in men with nonmetastatic prostate cancer and raise of prostate specific antigen (PSA) levels after local therapies (ARASTEP). ClinicalTrials.gov Identifier: NCT05794906. https://clinicaltrials.gov/ct2/show/NCT05794906?term=NCT05794906&draw=2&rank=1.
ClinicalTrials.gov. Combined PSMA PET/MRI for detection of lymph node metastases in high-risk prostate cancer patients. ClinicalTrials.gov Identifier: NCT04319783. https://classic.clinicaltrials.gov/ct2/show/NCT04790968.
ClinicalTrials.gov. PSMA response in metastasized hormone sensitive prostate cancer (PET-MaN). ClinicalTrials.gov Identifier: NCT05161728. https://classic.clinicaltrials.gov/ct2/show/NCT05161728.
ClinicalTrials.gov. PSMA-PET imaging for detecting early metastatic prostate cancer in men w/ high decipher test scores. ClinicalTrials.gov Identifier: NCT03495427. https://classic.clinicaltrials.gov/ct2/show/NCT03495427.
ClinicalTrials.gov. PSMA PET/CT guided intensification of therapy in patients at risk of advanced prostate cancer (PATRON). ClinicalTrials.gov Identifier: NCT04557501. https://classic.clinicaltrials.gov/ct2/show/NCT04557501.
Shore ND, Morgans AK, El-Haddad G, Srinivas S, Abramowitz M. Addressing challenges and controversies in the management of prostate cancer with multidisciplinary teams. Target Oncol. 2022;17:709–25.
Giri VN, Knudsen KE, Kelly WK, Cheng HH, Cooney KA, Cookson MS, et al. Implementation of germline testing for prostate cancer: Philadelphia Prostate Cancer Consensus Conference 2019. J Clin Oncol. 2020;38:2798–811.
Freedland SJ, Ramaswamy K, Huang A, Sandin R, Mardekian J, Schultz NM, et al. Survival and economic impact of rapid prostate-specific antigen doubling time in patients with nonmetastatic castration-resistant prostate cancer. Clin Genitourin Cancer. 2023;21:419–29.
Freedland SJ, Appukkuttan S, Yao J, Du Y, Parkin J, Partridge J, et al. Real-world clinical adverse events (AEs) among nonmetastatic castration-resistant prostate cancer (nmCRPC) patients treated with novel anti-androgen (AA) therapies: a retrospective database study. J Clin Oncol. 2021;39:e18675-e.
Yu EY. DARolutamide ObservationaL (DAROL) study in patients with nonmetastatic castration-resistant prostate cancer (nmCRPC). J Clin Oncol. 2022;40:abstr e17029.
George DJ, Khan N, Constantinovici N, Khan J, Chen G, Xu J, et al. Real-world use of darolutamide, enzalutamide, and apalutamide for non-metastatic castration-resistant prostate cancer (DEAR). J Clin Oncol. 2023;41:332.
George DJ, Sartor O, Miller K, Saad F, Tombal B, Kalinovsky J, et al. Treatment patterns and outcomes in patients with metastatic castration-resistant prostate cancer in a real-world clinical practice setting in the United States. Clin Genitourin Cancer. 2020;18:284–94.
Halwani AS, Patil V, Morreall D, Li C, Yong C, Appukkutan S, et al. Real-world treatment patterns among veterans with nonmetastatic castration-resistant prostate cancer (nmCRPC). J Clin Oncol. 2022;40:e17042-e.
Joyce DD, Sharma V, Zganjar A, Kwon ED, Jeffrey KR. MP27-18 Practice patterns in management of hormone sensitive metastatic and castrate-resistant non-metastatic prostate cancer. J Urol. 2022;207:e457.
Shore N, Jiang S, Garcia-Horton V, Terasawa E, Steffen D, Chin A, et al. The hospitalization-related costs of adverse events for novel androgen receptor inhibitors in non-metastatic castration-resistant prostate cancer: an indirect comparison. Adv Ther. 2022;39:5025–42.
Schultz NM, O’Day K, Sugarman R, Ramaswamy K. Budget impact of enzalutamide for nonmetastatic castration-resistant prostate cancer. J Manag Care Spec Pharm. 2020;26:538–49.
Appukkuttan S, Farej R, Miles L, Purser M, Wen L. Budget impact analysis of darolutamide for treatment of nonmetastatic castration-resistant prostate cancer. J Manag Care Spec Pharm. 2021;27:166–74.
Chapa R, Li CY, Basit A, Thakur A, Ladumor MK, Sharma S, et al. Contribution of uptake and efflux transporters to oral pharmacokinetics of furosemide. ACS Omega. 2020;5:32939–50.
Lexicomp. Drug interactions. Wolters Kluwer clinical drug information. Hudson (Ohio): Lexi-comp, Inc. http://online.lexi.com.
Drugs.com. Drug interactions checker. https://www.drugs.com/drug_interactions.html.
Acknowledgements
We acknowledge the contributions of Carden Jennings Publishing Co., Ltd., and medical writers Aimee Spevak and Caitlin Rothermel in assisting in the preparation of this manuscript. FOCUS: Androgen receptor antagonists; castration-resistant prostate cancer; drug interactions; prostate-specific antigen; quality of life; safety.
Funding
This activity is supported by an independent medical education grant from Bayer for medical writing of this manuscript.
Author information
Authors and Affiliations
Contributions
EAS, CHM, and AER contributed to section 1—Metastatic Free Survival (MFS) Endpoint. KLS, MSC, and AA contributed to section 2—Safety/Tolerability. RRM and MDK contributed to section 3—Drug-Drug Interactions. AHB contributed to section 4—Patient Monitoring. All authors contributed to section 5—Current Challenges and Future Considerations.
Corresponding author
Ethics declarations
Competing interests
NDS: Consulting fees: AbbVie, Alessa Therapeutics, Amgen, Arquer, Asieris, Astellas, Astra Zeneca, Bayer, BMS, Boston Scientific, Clarity, Dendreon, Exact Images, Ferring, FIZE Medical, GConcology, GenesisCare, Genetech, Foundation Medicine, ImmunityBio, Incyte, Invitae, Janssen, Lantheus, Lilly, MDX, Merck, Minomic, Myovant, Myriad, NGM, Nonagen, Novartis, NYMOX, Pacific Edge, Photocure, Pfizer, PlatformQ, Profound, Promaxo, Propella, Protara, Sanofi, Speciality Networks, Telix, Tolmar, Urogen, Vaxiion, Vessi; Payment for expert testimony: Ferring; Other financial or non-financial interests: Alessa, Photocure Board. ESA: Support for present the manuscript: Merck; Grants or contracts: Astellas, AstraZeneca, Bayer, Bristol Myers-Squibb, Celgene, Constellation Pharma, MacroGenics, Merck, Orion, Sanofi; Consulting fees: Aikido Pharma, Corcept Therapeutics, EcoR1, Foundation Medicine, Hookipa Pharma, Invitae, KeyQuest Health, Menarini Silicon Biosystems, z-Alpha; Payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events: UroWebinar; Support for attending meetings and/or travel: MJH Events, Research to Practice, Sanofi; Patents planned, issued or pending: Qiagen; Participation on a Data Safety Monitoring Board or Advisory Board: Aadi Bioscience/CM Propel, Amgen; Board or Advisory Board: AstraZeneca, Bayer, Blue Earth Diagnostics, Ismar, Janssen, Merck, Sanofi. AHB: Support for present manuscript: Bayer; Grants or contracts: Astellas; Payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events: Merck, Pfizer, Janssen, Myovant, Astra Zeneca, Bayer, Astellas, Novartis; Support for attending meetings and/or travel: Pfizer, Bayer. RRM: Consulting fees: AstraZeneca, Aveo, Bayer, Bristol-Myers Squibb, Calithera, Caris, Dendreon, Eisai, Exelixis, Johnson & Johnson, Lilly, Merck, Myovant, Novartis, Pfizer, Sanofi, SeaGen, Sorrento Therapeutics, Telix, Tempus; Institutional research support: AstraZeneca, Bayer, Tempus, Oncternal, BMS, Exelixis. KLS: Grants or contracts: Dendreon Pharmaceuticals, Alliance Foundation Trials, Pfizer-B8011006, TulsaPRO – Perigon, Landmark Services, Janssen-pending, Merck Master Engagement, Medical Director, Surgery Center, Phase IB Trial of Relugolix and Enzalutamide as Neoadjuv; Consulting fees: Merck & Company Advisory-expired; Payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events: Bayer Speaker Bureau 2022, Carden Jennings Publishing, Elzevier, Ebix Oakstone, National Comprehensive Cancer Network, Society of Urologic Nurses & Associates, ARK Prostate Cancer Symposium; Support for attending meetings and/or travel: Tulsa Pro Perigon – Onsite Case Observation; American Urological Association; National Comprehensive Cancer Network; Participation on a Data Safety Monitoring Board or Advisory Board: Merck; Leadership or fiduciary role in other board, society, committee or advocacy group: Society of Urologic Oncology Trials Consortium – Board of Directors, Prostate Organ Site Comm; Johns Hopkins Medicine/Oakstone Publishing Practical Reviews in Urology; Society of Urologic Oncology-Clinical Trials Consortium Board Of Directors; Society of Urologic Oncology Young Urologic Oncologist. AA: Nothing to disclose. MSC: Grants or contracts: MDXHealth, Clinical Trial Agreement; Royalties or licenses: Springer Nature Group; Consulting fees: Pfizer, Nonagen Bioscience Corp., Propella, Consulting JW, Myovant Sciences, Inc., TesoRx Pharma, LLC, Merck & Co., Inc.; Payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events: Remedica Medical Education & Publishing, MJH LifeSciences Prostate Cancer Roundtable, ASCO Symposium, Clinical Education Alliance, BioPharm Communications, Abstract Reviewer, LynxDX; Payment for expert testimony: Walters, Stanley, Natrajan; Rhodes & Hieryonymus; Williams, Hall, Latherow; Moscarino & Treu; Support for attending meetings and/or travel: American Urological Association (AUA), Society of Urologic Oncology, South Central Section of the AUA, UCSF Grand Rounds, Desai Sethi Urology Institute, American Board of Urology, Onclive (USPROST8CON), American Society of Clinical Oncologists (ASCO), Astellas, Vanderbilt University, Thomas Jefferson University, VP, Southeastern Section of the AUA, University of Wisconsin School of Medicine, VP; Participation on a Data Safety Monitoring Board or Advisory Board: Lantheus Advisory Board, Janssen Pharmaceuticals, Inc. Advisory Board, Pacific Edge Diagnostics USA Ltd., Advisory Board, Janssen Biotech, Academic Urology Advisory Board, Signatera GU Cancers Advisory Board, Bayer Prostate Cancer Advisory Council, Prokarium Ltd Bladder Cancer Advisory Board, Janssen Scientific Affairs, LLC Advisory Board, Bayer, Fallon Medica Advisory Board; Leadership or fiduciary role in other board, society, committee or advocacy group, paid or unpaid: American Urological Association (AUA), South Central Section of the AUA, Society of Urological Oncology, Urology Times, American Board of Urology. MDK: Nothing to disclose. CHM: Support for present the manuscript: NCI - P30 CA006973; Grants or contracts: V Foundation; Winn Career Development Award; Prostate Cancer Foundation; Royalties or licenses: McGraw Hill; Consulting fees: Astellas Pharmaceuticals, Obseva, Bayer; Payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events: Mashup MD; Participation on a Data Safety Monitoring Board or Advisory Board: Dendreon. AER: Grants or contracts: Site PI clinical trials with Bayer; Consulting fees: Astellas/Pfizer, Bayer, Blue Earth, Janssen, Lantheus; Payment or honoraria for lectures, presentations, speakers bureaus: Astellas/Pfizer, Blue Earth, Janssen, Lantheus.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: All authors have requested a correction to include the middle initials for all authors.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shore, N.D., Antonarakis, E.S., Ross, A.E. et al. A multidisciplinary approach to address unmet needs in the management of patients with non-metastatic castration-resistant prostate cancer. Prostate Cancer Prostatic Dis (2024). https://doi.org/10.1038/s41391-024-00803-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41391-024-00803-5
This article is cited by
-
Prostatectomy in oligometastatic prostate cancer: a call for high-quality evidence
Prostate Cancer and Prostatic Diseases (2024)