Abstract
Introduction
Magnetic resonance imaging is being widely adopted in the clinical management of prostate cancer. The correlation of the Prostate Imaging Reporting and Data System (PIRADS) to the presence of cancer has been established but studies have primarily evaluated this in a single clinical setting. This study aims to characterize the correlation of PIRADS score to the diagnosis of cancer on fusion biopsy among men who are undergoing primary biopsy, those who have had a previous negative biopsy or men on active surveillance.
Materials & Methods
A consecutive sample of men undergoing US-MR biopsy at a single academic institution from 2014 to 2017 were included in this retrospective study. Men were stratified into groups according to their clinical history: biopsy-naïve, previous negative transrectal ultrasound (TRUS) biopsy or on active surveillance. The correlation of PIRADS score to the diagnosis of any and clinically significant cancer (Gleason score ≥ 3 + 4) was determined.
Results
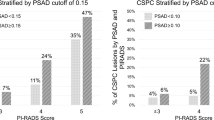
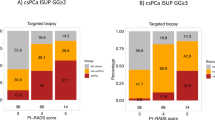
A total of 255 patients with 365 discrete lesions were analyzed. PIRADS score 1–2, 3, 4 and 5 yielded any prostate cancer in 7.7, 29.7, 42.3 and 82.4% of the cases, respectively, across all indications while clinically significant cancer was found in 0, 8.9, 21.4 and 62.7%, respectively. The area under the receiver operative curves for the diagnosis of any and significant cancer was 0.69 (95%CI: 0.64–0.74) and 0.74 (95%CI: 0.69–0.79) respectively. Men who have had a previous negative biopsy had lower detection rates for any prostate cancer for PIRADS 3 and 4 lesions compared to those that were biopsy-naïve or on active surveillance.
Conclusion
Cancer detection rates are significantly associated with PIRADS score. Biopsy yields differ across biopsy indications which should be considered when selecting a PIRADS score threshold for biopsy. Biopsy of PIRADS 3 lesions could potentially be avoided in men who have previously undergone a negative TRUS biopsy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Ahmed HU, El-Shater Bosaily A, Brown LC, Gabe R, Kaplan R, Parmar MK, et al. Diagnostic accuracy of multi-parametric MRI and TRUS biopsy in prostate cancer (PROMIS): a paired validating confirmatory study. Lancet. 2017;389:815–22.
Siddiqui MM, Rais-Bahrami S, Turkbey B, George AK, Rothwax J, Shakir N, et al. Comparison of MR/ultrasound fusion-guided biopsy with ultrasound-guided biopsy for the diagnosis of prostate cancer. JAMA. 2015;313:390–7.
Verma S, Choyke PL, Eberhardt SC, Oto A, Tempany CM, Turkbey B, et al. The Current state of MR imaging–targeted biopsy techniques for detection of prostate cancer. Radiology. 2017;285:343–56.
Weinreb JC, Barentsz JO, Choyke PL, Cornud F, Haider MA, Macura KJ, et al. PI-RADS Prostate imaging - reporting and data system: 2015, version 2. Eur Urol. 2016;69:16–40.
Woo S, Suh CH, Kim SY, Cho JY, Kim SH. Diagnostic performance of prostate imaging reporting and data system version 2 for detection of prostate cancer: a systematic review and diagnostic meta-analysis. Eur Urol. 2017;72:177–88.
Liddell H, Jyoti R, Haxhimolla HZ. mp-MRI prostate characterised PIRADS 3 lesions are associated with a low risk of clinically significant prostate cancer - a retrospective review of 92 biopsied PIRADS 3 lesions. Curr Urol. 2014;8:96–100.
Pokorny MR, de Rooij M, Duncan E, Schröder FH, Parkinson R, Barentsz JO, et al. Prospective study of diagnostic accuracy comparing prostate cancer detection by transrectal ultrasound– guided biopsy versus magnetic resonance (MR) imaging with subsequent mr-guided biopsy in men without previous prostate biopsies. Eur Urol. 2014;66:22–9.
Ting F, Van Leeuwen PJ, Thompson J, Shnier R, Moses D, Delprado W, et al. Assessment of the performance of magnetic resonance imaging/ultrasound fusion guided prostate biopsy against a combined targeted plus systematic biopsy approach using 24-core transperineal template saturation mapping prostate biopsy. Prostate Cancer. 2016;2016:3794738.
Hansen NL, Kesch C, Barrett T, Koo B, Radtke JP, Bonekamp D, et al. Multicentre evaluation of targeted and systematic biopsies using magnetic resonance and ultrasound image-fusion guided transperineal prostate biopsy in patients with a previous negative biopsy. BJU Int. 2016;120:631–8.
NiMhurchu E, O’Kelly F, Murphy IG, Lavelle LP, Collins CD, Lennon G, et al. Predictive value of PI-RADS classification in MRI-directed transrectal ultrasound guided prostate biopsy. Clin Radiol. 2016;71:375–80.
Wegelin O, van Melick HHE, Hooft L, Bosch J, Reitsma HB, Barentsz JO, et al. Comparing three different techniques for magnetic resonance imaging-targeted prostate biopsies: a systematic review of in-bore versus magnetic resonance imaging-transrectal ultrasound fusion versus cognitive registration. is there a preferred technique? Eur Urol. 2017;71:517–31.
Metzger GJ, Kalavagunta C, Spilseth B, Bolan PJ, Li X, Hutter D, et al. Detection of prostate cancer: quantitative multiparametric mr imaging models developed using registered correlative histopathology. Radiology. 2016;279:805–16.
DeLong ER, DeLong DM, Clarke-Pearson DL. Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics. 1988;44:837–45.
Venderink W, van Luijtelaar A, Bomers JG, van der Leest M, Hulsbergen-van de Kaa C, Barentsz JO, et al. Results of targeted biopsy in men with magnetic resonance imaging lesions classified equivocal, likely or highly likely to be clinically significant prostate cancer. Eur Urol. 2017;73;3:353–36
Rosenkrantz AB, Verma S, Choyke P, Eberhardt SC, Eggener SE, Gaitonde K, et al. Prostate magnetic resonance imaging and magnetic resonance imaging targeted biopsy in patients with a prior negative biopsy: a consensus statement by AUA and SAR. J Urol. 2016;196:1613–8.
Loeb S, Bjurlin M, Nicholson J, Tammela TL, Penson D, Carter HB, et al. Overdiagnosis and overtreatment of prostate cancer. Eur Urol. 2014;65:1046–55.
Pokorny MR, de Rooij M, Duncan E, Schroder FH, Parkinson R, Barentsz JO, et al. Prospective study of diagnostic accuracy comparing prostate cancer detection by transrectal ultrasound-guided biopsy versus magnetic resonance (MR) imaging with subsequent MR-guided biopsy in men without previous prostate biopsies. Eur Urol. 2014;66:22–9.
Siddiqui MM, George AK, Rubin R, Rais-Bahrami S, Parnes HL,Merino MJ, et al. Efficiency of prostate cancer diagnosis by mr/ultrasound fusion-guided biopsy vs standard extended-sextant biopsy for MR-visible lesions. J Natl Cancer Inst. 2016;108:djw039
Recabal P, Assel M, Sjoberg DD, Lee D, Laudone VP, Touijer K, et al. The efficacy of multiparametric magnetic resonance imaging and magnetic resonance imaging targeted biopsy in risk classification for patients with prostate cancer on active surveillance. J Urol. 2016;196:374–81.
Gaziev G, Wadhwa K, Barrett T, Koo BC, Gallagher FA, Serrao E, et al. Defining the learning curve for multiparametric magnetic resonance imaging (MRI) of the prostate using MRI-transrectal ultrasonography (TRUS) fusion-guided transperineal prostate biopsies as a validation tool. BJU Int. 2016;117:80–6.
Polanec S, Helbich TH, Bickel H, Pinker-Domenig K, Georg D, Shariat SF, et al. Head-to-head comparison of PI-RADS v2 and PI-RADS v1. Eur J Radiol. 2016;85:1125–31.
Kasel-Seibert M, Lehmann T, Aschenbach R, Guettler FV, Abubrig M, Grimm M-O, et al. Assessment of PI-RADS v2 for the Detection of Prostate Cancer. Eur J Radiol. 2016;85:726–31.
Auer T, Edlinger M, Bektic J, Nagele U, Herrmann T, Schäfer G, et al. Performance of PI-RADS version 1 versus version 2 regarding the relation with histopathological results. World J Urol. 2017;35:687–93.
Pessoa RR, Viana PC, Mattedi RL, Guglielmetti GB, Cordeiro MD, Coelho RF, et al. Value of 3-Tesla multiparametric magnetic resonance imaging and targeted biopsy for improved risk stratification in patients considered for active surveillance. BJU Int. 2017;119:535–42.
Acknowledgements
NJS has received support from the Cloverfields Foundation and The Institute for Prostate and Urologic Cancers (University of Minnesota).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Sathianathen, N., Konety, B., Soubra, A. et al. Which scores need a core? An evaluation of MR-targeted biopsy yield by PIRADS score across different biopsy indications. Prostate Cancer Prostatic Dis 21, 573–578 (2018). https://doi.org/10.1038/s41391-018-0065-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41391-018-0065-6
This article is cited by
-
PI-RADS upgrading as the strongest predictor for the presence of clinically significant prostate cancer in patients with initial PI-RADS-3 lesions
World Journal of Urology (2024)
-
Usefulness of grayscale values of hypoechoic lesions matched with target lesions observed on magnetic resonance imaging for the prediction of clinically significant prostate cancer
BMC Urology (2022)
-
Role of the elastography strain ratio using transrectal ultrasonography in the diagnosis of prostate cancer and clinically significant prostate cancer
Scientific Reports (2022)
-
Diagnostic value of combining PI-RADS v2.1 with PSAD in clinically significant prostate cancer
Abdominal Radiology (2022)
-
Evidence-based approach to active surveillance of prostate cancer
World Journal of Urology (2020)