Abstract
Background
The pathogenesis of renal disease in the context of overweight/obesity, metabolic syndrome, and insulin resistance is not completely understood. This may be due to the lack of a definitive animal model of disease, which limits our understanding of obesity-induced renal damage. We evaluated the changes in renal histology and lipid deposits induced by obesity in a model of insulin resistance: the Iberian swine fed with fat-enriched food.
Methods
Twenty-eight female sows were randomized to standard (SD) or high-fat diet (HFD: 6.8% of saturated fat) for 100 days. Weight, adiposity, analytics, oral glucose tolerance tests, and measured renal function were determined. Renal histology and lipid deposits in renal tissue were analyzed.
Results
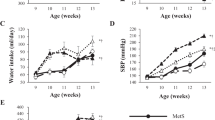
Animals on HFD developed obesity, hypertension, high levels of LDL cholesterol, triglycerides, insulin resistance, and glomerular hyperfiltration. No animal developed overt diabetes. Animals on HFD showed “diabetoid changes”, including mesangial expansion [21.40% ± 4 vs.13.20% ± 4.0, p < 0.0001], nodular glomerulosclerosis [7.40% ± 7, 0.75 vs. 2.40% ± 4.7, p = 0.02], and glomerulomegaly (18% vs. 10%, p = 0.010) than those on SD. Tubular atrophy, interstitial fibrosis, inflammation, arteriolar hyalinosis, or fibrointimal thickening were mild and similar between groups. Triglyceride content in renal tissue was higher in animals on HFD than in SD (15.4% ± 0.5 vs. 12.7% ± 0.7; p < 0.01).
Conclusions
Iberian pigs fed with fat-enriched food showed diabetoid changes and glomerulomegaly as observed in obese humans making this model suitable to study obesity-induced renal disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
http://www.who.int/mediacentre/factsheets/fs311/en/. Accessed January 2018.
Kahn R, Buse J, Ferrannini E, Stern M. The metabolic syndrome: time for a critical appraisal. Joint statement from the American Diabetes Association and the European Association for the Study of Diabetes. Diabetologia. 2005;48:1684–99.
Thomas G, Sehgal AR, Kashyap SR, Srinivas TR, Kirwan JP, Navaneethan SD. Metabolic syndrome and kidney disease: a systematic review and meta-analysis. Clin J Am Soc Nephrol. 2011;6:2364–73.
Hsu Cy, Mc Culloch ChE, Iribarren C, Darbinian J, Go AS. Body mass index and risk for end-stage renal disease. Ann Intern Med. 2006;144:21–8.
Hashimoto Y, Tanaka M, Okada H, Senmaru T, Hamaguchi M, Asano M, et al. Metabolically healthy obesity and risk of incident CKD. Clin J Am Soc Nephrol. 2015;10:578–83.
Stefansson V, Schei J, Solbu MD, Jenssen T, Melsom T, Eriksen B. Metabolic syndrome but not obesity measures are risk factors for accelerated age-related glomerular filtration rate decline in the general population. Kidney Int. 2018;93:1183–90
Kurella M, Lo JC, Chertow GM. Metabolic syndrome and the risk for chronic kidney disease among nondiabetic adults. J Am Soc Nephrol. 2005;16:2134–40.
Alexander MP, Patel TV, Farag YM, Florez A, Rennke A, Singh AK. Kidney pathological changes in metabolic syndrome: a cross-sectional study. Am J Kidney Dis. 2009;53:751–9.
Kambham N, Markowitz GS, Valeri AM, Lin J, D’Agati VD. Obesity-related glomerulopathy: an emerging epidemic. Kidney Int. 2001;59:1498–509.
D’Agati VD, Chagnac A, de Vries AP, Lev M, Porrini E, Herman-Edelstein M, et al. Obesity-related glomerulopathy: clinical and pathologic characteristics and pathogenesis. Nat Rev Nephrol. 2016;12:453–71.
Deji N, Kume S, Araki S, Soumura M, Sugimoto T, Isshiki K, et al. Structural and functional changes in the kidneys of high-fat diet-induced obese mice. Am J Physiol Renal Physiol. 2009;296:F118–26.
Kume S, Uzu T, Araki S, Sugimoto T, Isshiki K, Chin-Kanasaki M, et al. Role of altered renal lipid metabolism in the development of renal injury induced by a high-fat diet. J Am Soc Nephrol. 2007;18:2715–23.
Wei P, Lane PH, Lane JT, Padanilam BJ, Sansom SC. Glomerular structural and functional changes in a high-fat diet mouse model of early-stage Type 2 diabetes. Diabetologia. 2004;47:1541–9.
Jiang T, Wang Z, Proctor G, Moskowitz S, Liebman SE, Rogers T, et al. Diet-induced obesity in C57BL/6J mice causes increased renal lipid accumulation and glomerulosclerosis via a sterol regulatory element-binding protein-1c-dependent pathway. J Biol Chem. 2005;280:32317–25.
Dominguez J, Wu P, Packer CS, Temm C, Kelly KJ. Lipotoxic and inflammatory phenotypes in rats with uncontrolled metabolic syndrome and nephropathy. Am J Physiol Renal Physiol. 2007;293:F670–9.
Henegar JR, Bigler SA, Henegar LK, Tyagi SC, Hall JE. Functional and structural changes in the kidney in the early stages of obesity. J Am Soc Nephrol. 2001;12:1211–7.
Bloor ID, Sebert SP, Mahajan RP, Symonds ME. The influence of sex on early stage markers of kidney dysfunction in response to juvenile obesity. Hypertension. 2012;60:991–7.
Yokota SD, Benyajati S, Dantzler WH. Comparative aspects of glomerular filtration in vertebrates. Ren Physiol. 1985;8:193–221. PMID: 3906795
Davies B, Morris T. Physiological parameters in laboratory animals and humans. Pharm Res. 1993;10:1093–5. PMID: 8378254
Sachs DH. The pig as a potential xenograft donor. Vet Immunol Immunopathol. 1994;43:185–91. Review.
Tumbleson ME, Schook LB. In Advances in swine in biomedical research. Tumbleson ME, Schook LB, editors. Vol. 1. New York, USA: Plenum Press. p. 1–4.
Torres-Rovira L., Astiz S., Caro A., Lopez-Bote C., Ovilo C., Pallares P., et al. Diet-Induced SwineModel with obesity/leptin resistance for the study of metabolic syndrome and type 2 diabetes. Sci World J. 2012; p. 8. 510149.
Luis-Lima S, García-Contreras C, Vázquez-Gómez M, Astiz S, Carrara F, Gaspari F, et al. A simple method to measure renal function in swine by the plasma clearance of iohexol. Int J Mol Sci. 2018;19:pii: E232 https://doi.org/10.3390/ijms19010232
Racusen LC, Solez K, Colvin RB, Bonsib SM, Castro MC, Cavallo T, et al. The Banff 97 working classification of renal allograft pathology. Kidney Int. 1999;55:713–23.
Barbero A, Astiz S, Lopez-Bote CJ, Perez-Solana ML, Ayuso M, Garcia-Real I, et al. Maternal malnutrition and offspring sex determine juvenile obesity and metabolic disorders in a swine model of leptin resistance. PLoS One. 2013;8:e78424 https://doi.org/10.1371/journal.pone.0078424. eCollection 2013.
Nieto R, Miranda A, Garcia M, Aguilera J. The effect of dietary protein content and feeding level on the rate of protein deposition and energy utilization in growing Iberian pigs from15 to 50 kg body weight. Br J Nutr. 2002;88(no. 1):39–49.
Ovilo C, Fernandez A, Noguera JL, Barragán C, Letón R, Rodríguez C, et al. Fine mapping of porcine chromosome 6 QTL and LEPR effects on body composition in multiple generations of an Iberian by Landrace intercross. Genet Res. 2005;85(no. 1):57–67.
Muñoz G, Ovilo C, Silio L, Tomas A, Noguera J, Rodriguez M. Single- and joint-population analyses of two experimental pig crosses to confirm quantitative trait loci on Sus scrofa chromosome 6 and leptin receptor effects on fatness and growth traits. J Anim Sci. 2009;87(2):459–68.
Spurlock ME, Gabler NK. The development of porcine models of obesity and the metabolic syndrome. J Nutr. 2008;138(2):397–402.
Gonzalez-Bulnes A, Astiz S, Vazquez-Gomez M, Garcia-Contreras C. Developmental origins of metabolic disorders: The need for biomarker candidates and therapeutic targets from adequate preclinical models A. EuPA Open Proteomics. 2016;10:50–55.
Koopmans SJ, Schuurman T. Considerations on pig models for appetite, metabolic syndrome and obese type 2 diabetes: from food intake to metabolic disease. Eur J Pharmacol. 2015;759:231–9.
Kasiske BL, Crosson JT. Renal disease in patients with massive. Arch Intern Med. 1986;146:1105–9.
Griffin K, Kramer H, Bidani A. Adverse renal consequences of obesity. Am J Physiol Renal Physiol. 2008;294:F685–F696.
Palatini P, et al. Glomerular hyperfiltration predicts the development of microalbuminuria in stage 1 hypertension. Kidney Int. 2006;70:578–84.
Wiseman M, Saunders A, Keen H, Viberti F. Effect of blood glucose control on increased glomerular filtration rate and kidney size in insulin-dependent diabetes. N Engl J Med. 1985;312:617–21.
Sarafidis P, Ruilope L. Insulin resistance, hyperinsulinemia, and renal injury. Am J Nephrol. 2006;26:232–44.
Hostetter TH, Olson JL, Rennke HG, Venkatachalam MA, Brenner BM. Hyperfiltration in remnant nephrons. Am J Physiol. 1981;241:F85–93.
Chagnac A, Weinstein T, Korzets A, Ramadan E, Hirsch J, Gafter U. Glomerular hemodynamics in severe obesity. Am J Physiol Renal Physiol. 2000;278:F817–22.
Kriz W, Lemley KV. A potential role for mechanical forces in the detachment of podocytes and the progression of CKD. J Am Soc Nephrol. 2015;26:258–69.
Berfield AK, Andress DL, Abrass CK. IGF-1-induced lipid accumulation impairs mesangial cell migration and contractile function. Kidney Int. 2002;62:1229–37.
Singh AK, Gudehithlu KP, Pegoraro AA, Singh GK, Basheerudin K, Robey RB, et al. Vascular factors altered in glucose-treated mesangial cells and diabetic glomeruli. Changes in vascular factors impair endothelial cell growth and matrix. Lab. Invest. 2004;84:597–606.
de Vries AP, Ruggenenti P, Ruan XZ, Praga M, Cruzado JM, Bajema IM, et al. ERA-EDTA Working Group Diabesity. Fatty kidney: emerging role of ectopic lipid in obesity-related renal disease. Lancet Diabetes Endocrinol. 2014;2:417–26.
Bobulescu I, Lotan Y, Zhan J, Rosenthal T, Rogers J, Adams-Huet B, et al. Triglycerides in the human kidney cortex: relationship with body size. PLoS ONE 9: e101285.
Roden M, Price TB, Perseghin G, Petersen KF, Rothman DL, Cline GW, et al. Mechanism of free fatty acid-induced insulin resistance in humans. J Clin Invest. 1996;97:2859–65.
Poudyal H, Brown L. Stearoyl-CoA desaturase: a vital checkpoint in the development and progression of obesity. Endocr Metab Immune Disord Drug Targets. 2011;11:217–31.
Arany I, Clark J, Reed D, Juncos L, Dixit M. The role of p66shc in renal toxicity of oleic acid. Am J Nephrol. 2013; 38. https://doi.org/10.1159/000354357
Martínez-García C, Izquierdo-Lahuerta A, Vivas Y, Velasco I, Yeo T-K, Chen S, et al. Renal lipotoxicity-associated inflammation and insulin resistance affects actin cytoskeleton organization in podocytes. PLoS One. 2015;10:e0142291 https://doi.org/10.1371/journal.pone.0142291
Sieber J, Jehle A. Free fatty acids and their metabolism affect function and survival of podocytes. Front Endocrinol. 2014; 5:186.
Caligiuri SP, Blydt-Hansen T, Love K, Grégoire M, Taylor CG, Zahradka P, et al. Evidence for the use of glomerulomegaly as a surrogate marker of glomerular damage and for alpha-linolenic acid-rich oils in the treatment of early obesity-related glomerulopathy in a diet-induced rodent model of obesity. Appl Physiol Nutr Metab. 2014;39:951–9.
Li Z, Woollard JR, Wang S, Korsmo MJ, Ebrahimi B, Grande JP, et al. Increased glomerular filtration rate in early metabolic syndrome is associated with renal adiposity and microvascular proliferation. Am J Physiol Renal Physiol. 2011;301:F1078–87.
Acknowledgements
The authors thank the INIA farm staff for their assistance with animal care and handling. CGC, MVG, SA, and AGB are members of the EU COST-Action BM1308 “Sharing Advances on Large Animal Models (SALAAM)”. The authors would like to acknowledge the DIABESITY working group of the ERA-EDTA. the IMBRAIN (CIBICAN) project (FP7-RE6-POT- 2012-CT2012-31637-IMBRAIN) funded under the 7th Framework Program (capacities); the Instituto de Salud Carlos III (ISCIII) for the following grants: PI13/00342, PI, PI16/01814, the REDINREN RD16/0009 and PI10/02428; funding from the IRSIN (Instituto Reina Sofia de Investigacion), FEDER funds, SLL is a research fellow supported by the Instituto de Salud Carlos III (Grants for Río Hortega specialized healthcare post training contracts), ISCIII CM15/00214, Spain; EP is a Researcher of the Ramón y Cajal Program of the ISCIII. EP and AGB are the guarantors of the study.
Funding
The IMBRAIN (CIBICAN) project (FP7-RE6-POT-2012-CT2012-31637-IMBRAIN) 329 funded under the 7th Framework Program (capacities); the Instituto de Salud Carlos 330 III (ISCIII) for the following grants: PI13/00342, PI, PI16/01814, the REDINREN RD16/0009 and PI10/02428; funding from the IRSIN (Instituto Reina Sofia de Investigacion), FEDER funds, SLL is a research fellow supported by the Instituto de Salud Carlos III (Grants for Río Hortega specialized healthcare post training contracts), ISCIII CM15/00214, Spain; EP is a Researcher of the Ramón y Cajal Program of the ISCIII.
Author information
Authors and Affiliations
Contributions
AGB and EP had the idea of the study; RRR and JG performed the histological analysis, CGC, MVG, JLP, and BI helped in the performance of the study, AERR, ESR, and JDC helped in the histological evaluation, SLL performed the plasma clearance of iohexol.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Rodríguez, R.R., González-Bulnes, A., Garcia-Contreras, C. et al. The Iberian pig fed with high-fat diet: a model of renal disease in obesity and metabolic syndrome. Int J Obes 44, 457–465 (2020). https://doi.org/10.1038/s41366-019-0434-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41366-019-0434-9
This article is cited by
-
Rodent models to study type 1 and type 2 diabetes induced human diabetic nephropathy
Molecular Biology Reports (2023)
-
Dynamic chromatin architecture of the porcine adipose tissues with weight gain and loss
Nature Communications (2023)