Abstract
Background/objectives
A number of meta-analyses suggest an association between any maternal smoking in pregnancy and offspring overweight obesity. Whether there is a dose–response relationship across number of cigarettes and whether this differs by sex remains unclear.
Subject/methods
Studies reporting number of cigarettes smoked during pregnancy and offspring BMI published up to May 2015 were searched. An individual patient data meta-analysis of association between the number of cigarettes smoked during pregnancy and offspring overweight (defined according to the International Obesity Task Force reference) was computed using a generalized additive mixed model with non-linear effects and adjustment for confounders (maternal weight status, breastfeeding, and maternal education) and stratification for sex.
Results
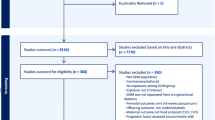
Of 26 identified studies, 16 authors provided data on a total of 238,340 mother–child-pairs. A linear positive association was observed between the number of cigarettes smoked and offspring overweight for up to 15 cigarettes per day with an OR increase per cigarette of 1.03, 95% CI = [1.02–1.03]. The OR flattened with higher cigarette use. Associations were similar in males and females. Sensitivity analyses supported these results.
Conclusions
A linear dose–response relationship of maternal smoking was observed in the range of 1–15 cigarettes per day equally in boys and girls with no further risk increase for doses above 15 cigarettes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Rayfield S, Plugge E. Systematic review and meta-analysis of the association between maternal smoking in pregnancy and childhood overweight and obesity. J Epidemiol Community Health. 2016;71:162–73. https://doi.org/10.1136/jech-2016-207376
Riedel C, Schönberger K, Yang S, Koshy G, Chen Y-C, Gopinath B, et al. Parental smoking and childhood obesity: higher effect estimates for maternal smoking in pregnancy compared to paternal smoking - a meta-analysis. Int J Epidemiol. 2014;43:1593–606.
Ino T. Maternal smoking during pregnancy and offspring obesity: meta-analysis. Pediatr Int. 2010;52:94–99.
Oken E, Levitan EB, Gillman MW. Maternal smoking during pregnancy and child overweight: systematic review and meta-analysis. Int J Obes. 2008;32:201–10.
Zinkhan EK, Lang BY, Yu B, Wang Y, Jiang C, Fitzhugh M, et al. Maternal tobacco smoke increased visceral adiposity and serum corticosterone levels in adult male rat offspring. Pediatr Res. 2014;76:17–23.
Gao Y-J, Holloway AC, Zeng Z, Lim GE, Petrik JJ, Foster WG, et al. Prenatal exposure to nicotine causes postnatal obesity and altered perivascular adipose tissue function. Obes Res. 2005;13:687–92.
Somm E, Schwitzgebel VM, Vauthay DM, Camm EJ, Chen CY, Giacobino J-P, et al. Prenatal nicotine exposure alters early pancreatic islet and adipose tissue development with consequences on the control of body weight and glucose metabolism later in life. Endocrinology. 2008;149:6289–99.
Oliveira E, Moura EG, Santos-Silva AP, Fagundes ATS, Rios AS, Abreu-Villaça Y, et al. Short- and long-term effects of maternal nicotine exposure during lactation on body adiposity, lipid profile, and thyroid function of rat offspring. J Endocrinol. 2009;202:397–405.
Holloway AC, Lim GE, Petrik JJ, Foster WG, Morrison KM, Gerstein HC. Fetal and neonatal exposure to nicotine in Wistar rats results in increased beta cell apoptosis at birth and postnatal endocrine and metabolic changes associated with type 2 diabetes. Diabetologia. 2005;48:2661–6.
Yousefi M, Karmaus W, Zhang H, Ewart S, Arshad H, Holloway JW. The methylation of the LEPR/LEPROT genotype at the promoter and body regions influence concentrations of leptin in girls and BMI at age 18 years if their mother smoked during pregnancy. Int J Mol Epidemiol Genet. 2013;4:86–100.
Rhee KE, Phelan S, McCaffery J. Early determinants of obesity: genetic, epigenetic, and in utero influences. Int J Pediatr. 2012;2012:1–9.
Joubert BR, Felix JF, Yousefi P, Bakulski KM, Just AC, Breton C, et al. DNA methylation in newborns and maternal smoking in pregnancy: genome-wide consortium meta-analysis. Am J Hum Genet. 2016;98:680–96.
Lee KWK, Richmond R, Hu P, French L, Shin J, Bourdon C, et al. Prenatal exposure to maternal cigarette smoking and DNA methylation: epigenome-wide association in a discovery sample of adolescents and replication in an independent cohort at birth through 17 years of age. Environ Health Perspect. 2015;123:193–9.
Leary SD, Smith GD, Rogers IS, Reilly JJ, Wells JC, Ness AR. Smoking during pregnancy and offspring fat and lean mass in childhood. Obes Silver Spring. 2006;14:2284–93.
Kleiser C, Schaffrath Rosario A, Mensink GB, Prinz-Langenohl R, Kurth BM. Potential determinants of obesity among children and adolescents in Germany: results from the cross-sectional KiGGS Study. BMC Public Health. 2009;9:46.
Plachta-Danielzik S, Kehden B, Landsberg B, Schaffrath Rosario A, Kurth BM, Arnold C, et al. Attributable risks for childhood overweight: evidence for limited effectiveness of prevention. Pediatrics. 2012;130:e865–e871.
Harris HR, Willett WC, Michels KB. Parental smoking during pregnancy and risk of overweight and obesity in the daughter. Int J Obes. 2005;2013:1356–63.
Iliadou AN, Koupil I, Villamor E, Altman D, Hultman C, Långström N, et al. Familial factors confound the association between maternal smoking during pregnancy and young adult offspring overweight. Int J Epidemiol. 2010;39:1193–202.
Gilman SE, Gardener H, Buka SL. Maternal smoking during pregnancy and children’s cognitive and physical development: a causal risk factor? Am J Epidemiol. 2008;168:522–31.
Eliopoulos C, Klein J, Phan MK, Knie B, Greenwald M, Chitayat D, et al. Hair concentrations of nicotine and cotinine in women and their newborn infants. JAMA. 1994;271:621–3.
Eliopoulos C, Klein J, Chitayat D, Greenwald M, Koren G. Nicotine and cotinine in maternal and neonatal hair as markers of gestational smoking. Clin Investig Med Médecine Clin Exp. 1996;19:231–42.
Møller SE, Ajslev TA, Andersen CS, Dalgård C, Sørensen TIA. Risk of childhood overweight after exposure to tobacco smoking in prenatal and early postnatal life. PLoS ONE. 2014;9:e109184.
Grzeskowiak LE, Hodyl NA, Stark MJ, Morrison JL, Clifton VL. Association of early and late maternal smoking during pregnancy with offspring body mass index at 4 to 5 years of age. J Dev Orig Health Dis. 2015;6:485–92.
Howe LD, Matijasevich A, Tilling K, Brion MJ, Leary SD, Smith GD, et al. Maternal smoking during pregnancy and offspring trajectories of height and adiposity: comparing maternal and paternal associations. Int J Epidemiol. 2012;41:722–32.
Koshy G, Delpisheh A, Brabin BJ. Dose response association of pregnancy cigarette smoke exposure, childhood stature, overweight and obesity. Eur J Public Health. 2011;21:286–91.
Sharma AJ, Cogswell ME, Li R. Dose-response associations between maternal smoking during pregnancy and subsequent childhood obesity: effect modification by maternal race/ethnicity in a low-income US cohort. Am J Epidemiol. 2008;168:995–1007.
Widerøe M, Vik T, Jacobsen G, Bakketeig LS. Does maternal smoking during pregnancy cause childhood overweight? Paediatr Perinat Epidemiol. 2003;17:171–9.
von Kries R, Bolte G, Baghi L, Toschke AM, Group GMES. Parental smoking and childhood obesity--is maternal smoking in pregnancy the critical exposure? Int J Epidemiol. 2008;37:210–6.
Hill SY, Shen S, Locke Wellman J, Rickin E, Lowers L. Offspring from families at high risk for alcohol dependence: increased body mass index in association with prenatal exposure to cigarettes but not alcohol. Psychiatry Res. 2005;135:203–16.
Chen A, Pennell ML, Klebanoff MA, Rogan WJ, Longnecker MP. Maternal smoking during pregnancy in relation to child overweight: follow-up to age 8 years. Int J Epidemiol. 2006;35:121–30.
Reilly JJ, Armstrong J, Dorosty AR, Emmett PM, Ness A, Rogers I, et al. Early life risk factors for obesity in childhood: cohort study. BMJ. 2005;330:1357.
Power C, Jefferis BJ. Fetal environment and subsequent obesity: a study of maternal smoking. Int J Epidemiol. 2002;31:413–9.
Dior UP, Lawrence GM, Sitlani C, Enquobahrie D, Manor O, Siscovick DS, et al. Parental smoking during pregnancy and offspring cardio-metabolic risk factors at ages 17 and 32. Atherosclerosis. 2014;235:430–7.
Durmuş B, Ay L, Hokken-Koelega ACS, Raat H, Hofman A, Steegers EAP, et al. Maternal smoking during pregnancy and subcutaneous fat mass in early childhood. The Generation R Study. Eur J Epidemiol. 2011;26:295–304.
Gorog K, Pattenden S, Antova T, Niciu E, Rudnai P, Scholtens S, et al. Maternal smoking during pregnancy and childhood obesity: results from the CESAR Study. Matern Child Health J. 2011;15:985–92.
Huang DYC, Lanza HI, Anglin MD. Trajectory of adolescent obesity: exploring the impact of prenatal to childhood experiences. J Child Fam Stud. 2014;23:1090–101.
Dahlhoff M, Pfister S, Blutke A, Rozman J, Klingenspor M, Deutsch MJ, et al. Peri-conceptional obesogenic exposure induces sex-specific programming of disease susceptibilities in adult mouse offspring. Biochim Biophys Acta. 2014;1842:304–17.
Stroup DF, Berlin JA, Morton SC, Olkin I, Williamson GD, Rennie D, et al. Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA. 2000;283:2008–12.
Cole TJ. Establishing a standard definition for child overweight and obesity worldwide: international survey. BMJ. 2000;320:1240
Sachdev HS, Fall CHD, Osmond C, Lakshmy R, Dey Biswas SK, Leary SD, et al. Anthropometric indicators of body composition in young adults: relation to size at birth and serial measurements of body mass index in childhood in the New Delhi birth cohort. Am J Clin Nutr. 2005;82:456–66.
Ylihärsilä H, Kajantie E, Osmond C, Forsén T, Barker DJ, Eriksson JG. Body mass index during childhood and adult body composition in men and women aged 56-70 y. Am J Clin Nutr. 2008;87:1769–75.
Bayer O, Krüger H, von Kries R, Toschke AM. Factors associated with tracking of BMI: a meta-regression analysis on BMI tracking. Obes Silver Spring Md. 2011;19:1069–76.
VanderWeele TJ, Mumford SL, Schisterman EF. Conditioning on intermediates in perinatal epidemiology. Epidemiol Camb Mass. 2012;23:1–9.
Rubin DB. Multiple imputation for nonresponse in surveys. Hoboken, NJ: Wiley-Interscience; 2004.
Lin X, Zhang D. Inference in generalized additive mixed modelsby using smoothing splines. J R Stat Soc Ser B Stat Methodol. 1999;61:381–400.
Jones G, Riley M, Dwyer T. Maternal smoking during pregnancy, growth, and bone mass in prepubertal children. J Bone Miner Res J Am Soc Bone Miner Res. 1999;14:146–51.
Suzuki K, Kondo N, Sato M, Tanaka T, Ando D, Yamagata Z. Gender differences in the association between maternal smoking during pregnancy and childhood growth trajectories: multilevel analysis. Int J Obes. 2011;35:53–59.
Suzuki K, Kondo N, Sato M, Tanaka T, Ando D, Yamagata Z. Maternal smoking during pregnancy and childhood growth trajectory: a random effects regression analysis. J Epidemiol. 2012;22:175–8.
Riedel C, Fenske N, Muller MJ, Plachta-Danielzik S, Keil T, Grabenhenrich L, et al. Differences in BMI z-scores between offspring of smoking and nonsmoking mothers: a longitudinal study of German children from birth through 14 years of age. Env Health Perspect. 2014;122:761–7.
Silva AA, Barbieri MA, Cardoso VC, Batista RF, Simões VM, Vianna EO, et al. Prevalence of non-communicable diseases in Brazilian children: follow-up at school age of two Brazilian birth cohorts of the 1990’s. BMC Public Health. 2011;11:486.
Boerschmann H, Pflüger M, Henneberger L, Ziegler A-G, Hummel S. Prevalence and predictors of overweight and insulin resistance in offspring of mothers with gestational diabetes mellitus. Diabetes Care. 2010;33:1845–9.
Oken E, Huh SY, Taveras EM, Rich-Edwards JW, Gillman MW. Associations of maternal prenatal smoking with child adiposity and blood pressure. Obes Res. 2005;13:2021–8.
Syme C, Abrahamowicz M, Mahboubi A, Leonard GT, Perron M, Richer L, et al. Prenatal exposure to maternal cigarette smoking and accumulation of intra-abdominal fat during adolescence. Obes Silver Spring Md. 2010;18:1021–5.
Pausova Z, Paus T, Abrahamowicz M, Bernard M, Gaudet D, Leonard G, et al. Cohort profile: the Saguenay Youth Study (SYS). Int J Epidemiol. 2016;46:e19 https://doi.org/10.1093/ije/dyw023
Thiering E, Brüske I, Kratzsch J, Thiery J, Sausenthaler S, Meisinger C, et al. Prenatal and postnatal tobacco smoke exposure and development of insulin resistance in 10 year old children. Int J Hyg Environ Health. 2011;214:361–8.
Prabhu N, Smith N, Campbell D, Craig LC, Seaton A, Helms PJ, et al. First trimester maternal tobacco smoking habits and fetal growth. Thorax. 2010;65:235–40.
Williams RL. Intrauterine growth curves: intra- and international comparisons with different ethnic groups in California. Prev Med. 1975;4:163–72.
Fried PA, Watkinson B, Gray R. Growth from birth to early adolescence in offspring prenatally exposed to cigarettes and marijuana. Neurotoxicol Teratol. 1999;21:513–25.
Kuhle S, Allen AC, Veugelers PJ. Prevention potential of risk factors for childhood overweight. Can J Public Health Rev Can St Publique. 2010;101:365–8.
Cavlek T, Cavlek M, Bozikov J, Sturz B, Grsic K. Influende of parental smoking on children’s growth and weight at birth and age 6. Paediatr Croat. 2010;54:117–23.
Taylor AE, Davey Smith G, Bares CB, Edwards AC, Munafo MR. Partner smoking and maternal cotinine during pregnancy: implications for negative control methods. Drug Alcohol Depend. 2014;139:159–63.
Mansi G, Raimondi F, Pichini S, Capasso L, Sarno M, Zuccaro P, et al. Neonatal urinary cotinine correlates with behavioral alterations in newborns prenatally exposed to tobacco smoke. Pediatr Res. 2007;61:257–61.
Laubenthal J, Zlobinskaya O, Poterlowicz K, Baumgartner A, Gdula MR, Fthenou E, et al. Cigarette smoke-induced transgenerational alterations in genome stability in cord blood of human F1 offspring. FASEB J. 2012;26:3946–56.
Kenny PJ. Common cellular and molecular mechanisms in obesity and drug addiction. Nat Rev Neurosci. 2011;12:638–51.
Haghighi A, Schwartz DH, Abrahamowicz M, Leonard GT, Perron M, Richer L, et al. Prenatal exposure to maternal cigarette smoking, amygdala volume, and fat intake in adolescence. JAMA Psychiatry. 2013;70:98–105.
Rolls ET. Taste, olfactory and food texture reward processing in the brain and obesity. Int J Obes. 2005;2011:550–61.
Bray GA, Popkin BM. Dietary fat intake does affect obesity! Am J Clin Nutr. 1998;68:1157–73.
Tappy L. Thermic effect of food and sympathetic nervous system activity in humans. Reprod Nutr Dev. 1996;36:391–7.
Wrase J, Makris N, Braus DF, Mann K, Smolka MN, Kennedy DN, et al. Amygdala volume associated with alcohol abuse relapse and craving. Am J Psychiatry. 2008;165:1179–84.
Primeaux SD, York DA, Bray GA. Neuropeptide Y administration into the amygdala alters high fat food intake. Peptides. 2006;27:1644–51.
Boghossian S, Park M, York DA. Melanocortin activity in the amygdala controls appetite for dietary fat. Am J Physiol Regul Integr Comp Physiol. 2010;298:R385–R393.
Grabenhorst F, Rolls ET, Parris BA, d’Souza AA. How the brain represents the reward value of fat in the mouth. Cereb Cortex. 2010;20:1082–91. (N Y N 1991).
Trygg K, Lund-Larsen K, Sandstad B, Hoffman HJ, Jacobsen G, Bakketeig LS. Do pregnant smokers eat differently from pregnant non-smokers? Paediatr Perinat Epidemiol. 1995;9:307–19.
Llaquet H, Pichini S, Joya X, Papaseit E, Vall O, Klein J, et al. Biological matrices for the evaluation of exposure to environmental tobacco smoke during prenatal life and childhood. Anal Bioanal Chem. 2010;396:379–99.
Jacqz-Aigrain E, Zhang D, Maillard G, Luton D, André J, Oury JF. Maternal smoking during pregnancy and nicotine and cotinine concentrations in maternal and neonatal hair. BJOG Int J Obstet Gynaecol. 2002;109:909–11.
Toschke AM, Montgomery SM, Pfeiffer U, von Kries R. Early intrauterine exposure to tobacco-inhaled products and obesity. Am J Epidemiol. 2003;158:1068–74.
Venditti CC, Smith GN. Self-reported cigarette smoking status imprecisely quantifies exposure in pregnancy. Open J Obstet Gynecol. 2012;02:56–61.
Pickett KE, Rathouz PJ, Kasza K, Wakschlag LS, Wright R. Self-reported smoking, cotinine levels, and patterns of smoking in pregnancy. Paediatr Perinat Epidemiol. 2005;19:368–76.
Talge NM, Mudd LM, Sikorskii A, Basso O. United States birth weight reference corrected for implausible gestational age estimates. Pediatrics. 2014;133:844–53.
Cole TJ, Freeman JV, Preece MA. British 1990 growth reference centiles for weight, height, body mass index and head circumference fitted by maximum penalized likelihood. Stat Med. 28. 1998;17:407–29.
Voigt M, Schneider K, Jährig K. Analyse des Geburtengutes des Jahrgangs 1992 der Bundesrepublik Deutschland. Geburtshilfe Frauenheilkd. 1996;56:550–8.
Dobbins TA, Sullivan EA, Roberts CL, Simpson JM. Australian national birthweight percentiles by sex and gestational age, 1998-2007. Med J Aust. 3. 2012;197:291–4.
Oken E, Kleinman KP, Rich-Edwards J, Gillman MW. A nearly continuous measure of birth weight for gestational age using a United States national reference. BMC Pediatr. 2003;3:6.
Acknowledgements
We want to thank the funders of the individual studies: the UK Medical Research Council and the Wellcome Trust (Grant ref: 102215/2/13/2) and the University of Bristol, the Danish National Research Foundation, Pharmacy Foundation, the March of Dimes Birth Defects Foundation, the Augustinus Foundation, and the Health Foundation, the US NICHD (contracts no. 1-HD-4-2803 and no. 1-HD-1-3127, R01 HD HD034568), the NHMRC, the CNPq (Portuguese acronym for the National Research Council—grant 523474/96-2) and FAPESP (Portuguese acronym for the São Paulo State Research Council—grant 00/0908-7). We would like to thank the participating families of all studies for the use of data. For the ASPAC study, we want to thank the midwives for their help in recruiting families, and the whole ALSPAC team, which includes interviewers, computer and laboratory technicians, clerical workers, research scientists, volunteers, managers, receptionists, and nurses. This work was supported by the Deutschen Forschungsgesellschaft (German Research Foundation, DFG) [KR 1926/9-1, KU1443/4-1]. Dr. Gilman’s contribution was supported by the Intramural Research Program of the Eunice Kennedy Shriver National Institute of Child Health and Human Development.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Albers, L., Sobotzki, C., Kuß, O. et al. Maternal smoking during pregnancy and offspring overweight: is there a dose–response relationship? An individual patient data meta-analysis. Int J Obes 42, 1249–1264 (2018). https://doi.org/10.1038/s41366-018-0050-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41366-018-0050-0
This article is cited by
-
Maternal smoking, nutritional factors at different life stage, and the risk of incident type 2 diabetes: a prospective study of the UK Biobank
BMC Medicine (2024)
-
Comparing body composition between the sweet-liking phenotypes: experimental data, systematic review and individual participant data meta-analysis
International Journal of Obesity (2024)
-
Smoking during pregnancy is associated with child overweight independent of maternal pre-pregnancy BMI and genetic predisposition to adiposity
Scientific Reports (2022)
-
Early life exposures are associated with appetitive traits in infancy: findings from the BiTwin cohort
European Journal of Nutrition (2022)
-
Racial Disparities in Obesity Treatment Among Children and Adolescents
Current Obesity Reports (2021)