Abstract
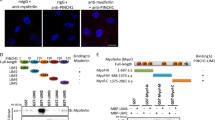
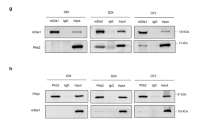
Myoferlin, a member of ferlin family of proteins, was first discovered as a candidate gene for muscular dystrophy and cardiomyopathy. Recently, myoferlin was shown to be also expressed in endothelial and cancer cells where it was shown to modulate vascular endothelial growth factor (VEGFR)-2 and epidermal growth factor receptor (EGFR) signaling by enhancing their stability and recycling. Based on these reports, we hypothesized that myoferlin might be regulating IL-6 signaling by modulating IL-6R stabilization and recycling. However, in our immunoprecipitation (IP) experiments, we did not observe myoferlin binding with IL-6R. Instead, we made a novel discovery that in resting cells myoferlin was bound to EHD2 protein and when cells were treated with IL-6, myoferlin dissociated from EHD2 and binds to activated STAT3. Interestingly, myoferlin depletion did not affect STAT3 phosphorylation, but completely blocked STAT3 translocation to nucleus. In addition, inhibition of STAT3 phosphorylation by phosphorylation-defective STAT3 mutants or JAK inhibitor blocked STAT3 binding to myoferlin and nuclear translocation. Myoferlin knockdown significantly decreased IL-6-mediated tumor cell migration, tumorsphere formation and ALDH-positive cancer stem cell population, in vitro. Furthermore, myoferlin knockdown significantly decreased IL-6-meditated tumor growth and tumor metastasis. Based on these results, we have proposed a novel model for the role of myoferlin in chaperoning phosphorylated STAT3 to the nucleus.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Kishimoto T . Interleukin-6: discovery of a pleiotropic cytokine. Arthritis Res Ther 2006; 8: S2.
Yadav A, Kumar B, Datta J, Teknos TN, Kumar P . IL-6 promotes head and neck tumor metastasis by inducing epithelial-mesenchymal transition via the JAK-STAT3-SNAIL signaling pathway. Mol Cancer Res 2011; 9: 1658–1667.
Lederle W, Depner S, Schnur S, Obermueller E, Catone N, Just A et al. IL-6 promotes malignant growth of skin SCCs by regulating a network of autocrine and paracrine cytokines. Int J Cancer 2011; 128: 2803–2814.
Lesina M, Kurkowski Magdalena U, Ludes K, Rose-John S, Treiber M, Klöppel G et al. Stat3/Socs3 activation by IL-6 transsignaling promotes progression of pancreatic intraepithelial neoplasia and development of pancreatic cancer. Cancer Cell 2011; 19: 456–469.
Nakashima J, Tachibana M, Horiguchi Y, Oya M, Ohigashi T, Asakura H et al. Serum interleukin 6 as a prognostic factor in patients with prostate cancer. Clin Cancer Res 2000; 6: 2702–2706.
Dethlefsen C, Højfeldt G, Hojman P . The role of intratumoral and systemic IL-6 in breast cancer. Breast Cancer Res Treat 2013; 138: 657–664.
Duffy SA, Taylor JM, Terrell JE, Islam M, Li Y, Fowler KE et al. Interleukin-6 predicts recurrence and survival among head and neck cancer patients. Cancer 2008; 113: 750–757.
Riedel F, Zaiss I, Herzog D, Gotte K, Naim R, Hormann K . Serum levels of interleukin-6 in patients with primary head and neck squamous cell carcinoma. Anticancer Res 2005; 25: 2761–2765.
Yamasaki K, Taga T, Hirata Y, Yawata H, Kawanishi Y, Seed B et al. Cloning and expression of the human interleukin-6 (BSF-2/IFN beta 2) receptor. Science 1988; 241: 825–828.
Hibi M, Murakami M, Saito M, Hirano T, Taga T, Kishimoto T . Molecular cloning and expression of an IL-6 signal transducer, gp130. Cell 1990; 63: 1149–1157.
Taga T, Hibi M, Hirata Y, Yamasaki K, Yasukawa K, Matsuda T et al. Interleukin-6 triggers the association of its receptor with a possible signal transducer, gp130. Cell 1989; 58: 573–581.
Murakami M, Hibi M, Nakagawa N, Nakagawa T, Yasukawa K, Yamanishi K et al. IL-6-induced homodimerization of gp130 and associated activation of a tyrosine kinase. Science 1993; 260: 1808–1810.
Heinrich PC, Behrmann I, Muller-Newen G, Schaper F, Graeve L . Interleukin-6-type cytokine signalling through the gp130/Jak/STAT pathway. Biochem J 1998; 334: 297–314.
Zhong Z, Wen Z, Darnell J . Stat3: a STAT family member activated by tyrosine phosphorylation in response to epidermal growth factor and interleukin-6. Science 1994; 264: 95–98.
Wente SR, Rout MP . The nuclear pore complex and nuclear transport. Cold Spring Harb Perspect Biol 2010; 2: a000562.
Bernatchez PN, Sharma A, Kodaman P, Sessa WC . Myoferlin is critical for endocytosis in endothelial cells. Am J Physiol Cell Physiol 2009; 297: C484–C492.
Bernatchez PN, Acevedo L, Fernandez-Hernando C, Murata T, Chalouni C, Kim J et al. Myoferlin regulates vascular endothelial growth factor receptor-2 stability and function. J Biol Chem 2007; 282: 30745–30753.
Davis DB, Delmonte AJ, Ly CT, McNally EM . Myoferlin, a candidate gene and potential modifier of muscular dystrophy. Hum Mol Genet 2000; 9: 217–226.
Jiménez JL, Bashir R . In silico functional and structural characterisation of ferlin proteins by mapping disease-causing mutations and evolutionary information onto three-dimensional models of their C2 domains. J Neurol Sci 2007; 260: 114–123.
Perin MS, Friedt VA, Mignery GA, Jahn R, Südhof TC . Phospholipid binding by a synaptic vesicle protein homologous to the regulatory region of protein kinase C. Nature 1990; 345: 260–263.
Liu J, Aoki M, Illa I, Wu C, Fardeau M, Angelini C et al. Dysferlin, a novel skeletal muscle gene, is mutated in Miyoshi myopathy and limb girdle muscular dystrophy. Nat Genet 1998; 20: 31–36.
Demonbreun AR, Lapidos KA, Heretis K, Levin S, Dale R, Pytel P et al. Myoferlin regulation by NFAT in muscle injury, regeneration and repair. J Cell Sci 2010; 123: 2413–2422.
Doherty KR, Cave A, Davis DB, Delmonte AJ, Posey A, Earley JU et al. Normal myoblast fusion requires myoferlin. Development 2005; 132: 5565–5575.
Doherty KR, Demonbreun AR, Wallace GQ, Cave A, Posey AD, Heretis K et al. The endocytic recycling protein EHD2 interacts with myoferlin to regulate myoblast fusion. J Biol Chem 2008; 283: 20252–20260.
Roux I, Safieddine S, Nouvian R, Grati Mh, Simmler M-C, Bahloul A et al. Otoferlin, defective in a human deafness form, is essential for exocytosis at the auditory ribbon synapse. Cell 2006; 127: 277–289.
Davis DB, Doherty KR, Delmonte AJ, McNally EM . Calcium-sensitive phospholipid binding properties of normal and mutant ferlin C2 domains. J Biol Chem 2002; 277: 22883–22888.
Turtoi A, Blomme A, Bellahcene A, Gilles C, Hennequiere V, Peixoto P et al. Myoferlin is a key regulator of EGFR activity in breast cancer. Cancer Res 2013; 73: 5438–5448.
Li R, Ackerman WEt, Mihai C, Volakis LI, Ghadiali S, Kniss DA . Myoferlin depletion in breast cancer cells promotes mesenchymal to epithelial shape change and stalls invasion. PLoS One 2012; 7: e39766.
Turtoi A, Musmeci D, Wang Y, Dumont B, Somja J, Bevilacqua G et al. Identification of novel accessible proteins bearing diagnostic and therapeutic potential in human pancreatic ductal adenocarcinoma. J Proteome Res 2011; 10: 4302–4313.
Difilippantonio S, Chen Y, Pietas A, Schlüns K, Pacyna-Gengelbach M, Deutschmann N et al. Gene expression profiles in human non-small and small-cell lung cancers. Eur J Cancer 2003; 39: 1936–1947.
Abba MC, Hu Y, Sun H, Drake JA, Gaddis S, Baggerly K et al. Gene expression signature of estrogen receptor α status in breast cancer. BMC Genomics 2005; 6: 37–37.
Yu C, Sharma A, Trane A, Utokaparch S, Leung C, Bernatchez P . Myoferlin gene silencing decreases Tie-2 expression in vitro and angiogenesis in vivo. Vasc Pharmacol 2011; 55: 26–33.
Kumar B, Brown NB, Swanson BJ, Schmitt AC, Old O, Ozer E et al. High expression of myoferlin is associated with poor outcome in oropharyngeal squamous cell carcinoma patients and is inversely associated with HPV-status. Oncotarget 2016; 7: 18665–18677.
Tian SS, Lamb P, Seidel HM, Stein RB, Rosen J . Rapid activation of the STAT3 transcription factor by granulocyte colony-stimulating factor. Blood 1994; 84: 1760–1764.
Li R, Ackerman WE, Mihai C, Volakis LI, Ghadiali S, Kniss DA . Myoferlin depletion in breast cancer cells promotes mesenchymal to epithelial shape change and stalls invasion. PLoS ONE 2012; 7: e39766.
Schreiner SJ, Schiavone AP, Smithgall TE . Activation of STAT3 by the Src family kinase Hck requires a functional SH3 domain. J Biol Chem 2002; 277: 45680–45687.
Villalva C, Martin-Lanneree S, Cortes U, Dkhissi F, Wager M, Le Corf A et al. STAT3 is essential for the maintenance of neurosphere-initiating tumor cells in patients with glioblastomas: a potential for targeted therapy? Int J Cancer 2010; 128: 826–838.
Kumar P, Ning Y, Polverini PJ . Endothelial cells expressing Bcl-2 promotes tumor metastasis by enhancing tumor angiogenesis, blood vessel leakiness and tumor invasion. Lab Invest 2008; 88: 740–749.
Li M, Yang X, Zhang J, Shi H, Hang Q, Huang X et al. Effects of EHD2 interference on migration of esophageal squamous cell carcinoma. Med Oncol 2013; 30: 396.
Yang X, Ren H, Yao L, Chen X, He A . Role of EHD2 in migration and invasion of human breast cancer cells. Tumor Biol 2015; 36: 3717–3726.
Shi Y, Liu X, Sun Y, Wu D, Qiu A, Cheng H et al. Decreased expression and prognostic role of EHD2 in human breast carcinoma: correlation with E-cadherin. J Mol Hist 2015; 46: 221–231.
Lv J, Fan N, Wang Y, Wang X, Gao C . Identification of differentially expressed proteins of normal and cancerous human colorectal tissues by liquid chromatograph-mass spectrometer based on iTRAQ approach. Cancer Invest 2015; 33: 420–428 1-9.
Eisenberg MC, Kim Y, Li R, Ackerman WE, Kniss DA, Friedman A . Mechanistic modeling of the effects of myoferlin on tumor cell invasion. Proc Natl Acad Sci 2011; 108: 20078–20083.
Liu L, McBride KM, Reich NC . STAT3 nuclear import is independent of tyrosine phosphorylation and mediated by importin-α3. Proc Natl Acad Sci USA 2005; 102: 8150–8155.
Yahata Y, Shirakata Y, Tokumaru S, Yamasaki K, Sayama K, Hanakawa Y et al. Nuclear translocation of phosphorylated STAT3 is essential for vascular endothelial growth factor-induced human dermal microvascular endothelial cell migration and tube formation. J Biol Chem 2003; 278: 40026–40031.
Liu Y-p, Tan Y-n, Wang Z-l, Zeng L, Lu Z-x, Li L-l et al. Phosphorylation and nuclear translocation of STAT3 regulated by the Epstein-Barr virus latent membrane protein 1 in nasopharyngeal carcinoma. Int J Mol Med 2008; 21: 153–162.
DeFranco DB . Role of molecular chaperones in subnuclear trafficking of glucocorticoid receptors. Kidney Int 2000; 57: 1241–1249.
Amatschek S, Koenig U, Auer H, Steinlein P, Pacher M, Gruenfelder A et al. Tissue-wide expression profiling using cDNA subtraction and microarrays to identify tumor-specific genes. Cancer Res 2004; 64: 844–856.
Adam PJ, Boyd R, Tyson KL, Fletcher GC, Stamps A, Hudson L et al. Comprehensive proteomic analysis of breast cancer cell membranes reveals unique proteins with potential roles in clinical cancer. J Biol Chem 2003; 278: 6482–6489.
van 't Veer LJ, Dai H, van de Vijver MJ, He YD, Hart AAM, Mao M et al. Gene expression profiling predicts clinical outcome of breast cancer. Nature 2002; 415: 530–536.
Bourguignon LY, Earle C, Wong G, Spevak CC, Krueger K . Stem cell marker (Nanog) and Stat-3 signaling promote MicroRNA-21 expression and chemoresistance in hyaluronan/CD44-activated head and neck squamous cell carcinoma cells. Oncogene 2012; 31: 149–160.
Real PJ, Sierra A, De Juan A, Segovia JC, Lopez-Vega JM, Fernandez-Luna JL . Resistance to chemotherapy via Stat3-dependent overexpression of Bcl-2 in metastatic breast cancer cells. Oncogene 2002; 21: 7611–7618.
Brenner JC, Graham MP, Kumar B, Saunders LM, Kupfer R, Lyons RH et al. Genotyping of 73 UM-SCC head and neck squamous cell carcinoma cell lines. Head Neck 2010; 32: 417–426.
Al Moustafa A-E, Foulkes WD, Benlimame N, Wong A, Yen L, Bergeron J et al. E6//E7 proteins of HPV type 16 and ErbB-2 cooperate to induce neoplastic transformation of primary normal oral epithelial cells. Oncogene 2004; 23: 350–358.
Livak KJ, Schmittgen TD . Analysis of relative gene expression data using real-time quantitative PCR and the 2-[Delta][Delta]CT method. Methods 2001; 25: 402–408.
Kumar B, Yadav A, Lang JC, Teknos TN, Kumar P . Suberoylanilide hydroxamic acid (SAHA) reverses chemoresistance in head and neck cancer cells by targeting cancer stem cells via the downregulation of nanog. Genes Cancer 2015; 6: 169–181.
Kumar P, Benedict R, Urzua F, Fischbach C, Mooney D, Polverini P . Combination treatment significantly enhances the efficacy of antitumor therapy by preferentially targeting angiogenesis. Lab Invest 2005; 85: 756–767.
Harrell MI, Iritani BM, Ruddell A . Lymph node mapping in the mouse. J Immunol Methods 2008; 332: 170–174.
Acknowledgements
This work was supported in part by funding from NIH/NCI (CA178649 to PK) and The Ohio State University Comprehensive Cancer Center (PK and TNT). NIH/NCI-CA178649 (P Kumar) and The Ohio State University Comprehensive Cancer Center.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Yadav, A., Kumar, B., Lang, J. et al. A muscle-specific protein ‘myoferlin’ modulates IL-6/STAT3 signaling by chaperoning activated STAT3 to nucleus. Oncogene 36, 6374–6382 (2017). https://doi.org/10.1038/onc.2017.245
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2017.245
This article is cited by
-
Identification of myoferlin as a mitochondria-associated membranes component required for calcium signaling in PDAC cell lines
Cell Communication and Signaling (2024)
-
Human colon cancer cells highly express myoferlin to maintain a fit mitochondrial network and escape p53-driven apoptosis
Oncogenesis (2019)