Abstract
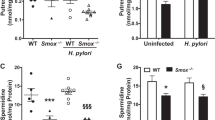
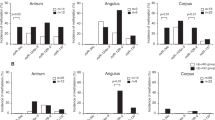
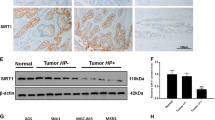
Chronic inflammation contributes to the development of various forms of cancer. The polyamine catabolic enzyme spermine oxidase (SMOX) is induced in chronic inflammatory conditions, including Helicobacter pylori-associated gastritis, where its production of hydrogen peroxide contributes to DNA damage and subsequent tumorigenesis. MicroRNA expression levels are also altered in inflammatory conditions; specifically, the tumor suppressor miR-124 becomes silenced by DNA methylation. We sought to determine if this repression of miR-124 is associated with elevated SMOX activity and concluded that miR-124 is indeed a negative regulator of SMOX. In gastric adenocarcinoma cells harboring highly methylated and silenced mir-124 gene loci, 5-azacytidine treatment allowed miR-124 re-expression and decreased SMOX expression. Overexpression of an exogenous miR-124-3p mimic repressed SMOX mRNA and protein expression as well as H2O2 production by >50% within 24 h. Reporter assays indicated that direct interaction of miR-124 with the 3′-untranslated region of SMOX mRNA contributes to this negative regulation. Importantly, overexpression of miR-124 before infection with H. pylori prevented the induction of SMOX believed to contribute to inflammation-associated tumorigenesis. Compelling human in vivo data from H. pylori-positive gastritis tissues indicated that the mir-124 gene loci are more heavily methylated in a Colombian population characterized by elevated SMOX expression and a high risk for gastric cancer. Furthermore, the degree of mir-124 methylation significantly correlated with SMOX expression throughout the population. These results indicate a protective role for miR-124 through the inhibition of SMOX-mediated DNA damage in the etiology of H. pylori-associated gastric cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Wang Y, Devereux W, Woster PM, Stewart TM, Hacker A, Casero RA Jr . Cloning and characterization of a human polyamine oxidase that is inducible by polyamine analogue exposure. Cancer Res 2001; 61: 5370–5373.
Murray-Stewart T, Wang Y, Goodwin A, Hacker A, Meeker A, Casero RA Jr . Nuclear localization of human spermine oxidase isoforms—possible implications in drug response and disease etiology. FEBS J 2008; 275: 2795–2806.
Cervelli M, Bellini A, Bianchi M, Marcocci L, Nocera S, Polticelli F et al. Mouse spermine oxidase gene splice variants. Nuclear subcellular localization of a novel active isoform. Eur J Biochem 2004; 271: 760–770.
Ha HC, Sirisoma NS, Kuppusamy P, Zweier JL, Woster PM, Casero RA Jr . The natural polyamine spermine functions directly as a free radical scavenger. Proc Natl Acad Sci USA 1998; 95: 11140–11145.
Chaturvedi R, Cheng Y, Asim M, Bussiere FI, Xu H, Gobert AP et al. Induction of polyamine oxidase 1 by Helicobacter pylori causes macrophage apoptosis by hydrogen peroxide release and mitochondrial membrane depolarization. J Biol Chem 2004; 279: 40161–40173.
Xu H, Chaturvedi R, Cheng Y, Bussiere FI, Asim M, Yao MD et al. Spermine oxidation induced by Helicobacter pylori results in apoptosis and DNA damage: implications for gastric carcinogenesis. Cancer Res 2004; 64: 8521–8525.
Goodwin AC, Destefano Shields CE, Wu S, Huso DL, Wu X, Murray-Stewart TR et al. Polyamine catabolism contributes to enterotoxigenic Bacteroides fragilis-induced colon tumorigenesis. Proc Natl Acad Sci USA 2011; 108: 15354–15359.
Babbar N, Casero RA Jr . Tumor necrosis factor-alpha increases reactive oxygen species by inducing spermine oxidase in human lung epithelial cells: a potential mechanism for inflammation-induced carcinogenesis. Cancer Res 2006; 66: 11125–11130.
Chaturvedi R, Asim M, Piazuelo MB, Yan F, Barry DP, Sierra JC et al. Activation of EGFR and ERBB2 by Helicobacter pylori results in survival of gastric epithelial cells with DNA damage. Gastroenterology 2014; 146: 1739–1751, 1714.
Goodwin AC, Jadallah S, Toubaji A, Lecksell K, Hicks JL, Kowalski J et al. Increased spermine oxidase expression in human prostate cancer and prostatic intraepithelial neoplasia tissues. Prostate 2008; 68: 766–772.
Hong SK, Chaturvedi R, Piazuelo MB, Coburn LA, Williams CS, Delgado AG et al. Increased expression and cellular localization of spermine oxidase in ulcerative colitis and relationship to disease activity. Inflamm Bowel Dis 2010; 16: 1557–1566.
Chaturvedi R, Asim M, Romero-Gallo J, Barry DP, Hoge S, de Sablet T et al. Spermine oxidase mediates the gastric cancer risk associated with Helicobacter pylori CagA. Gastroenterology 2011; 141: 1696–1708, 1691 and 1692.
Chaturvedi R, de Sablet T, Asim M, Piazuelo MB, Barry DP, Verriere TG et al. Increased Helicobacter pylori-associated gastric cancer risk in the Andean region of Colombia is mediated by spermine oxidase. Oncogene 2015; 34: 3429–3440.
Goodwin CS . Helicobacter pylori gastritis, peptic ulcer, and gastric cancer: clinical and molecular aspects. Clin Infect Dis 1997; 25: 1017–1019.
Plummer M, Franceschi S, Vignat J, Forman D, de Martel C . Global burden of gastric cancer attributable to Helicobacter pylori. Int J Cancer 2015; 136: 487–490.
Peek RM Jr, Blaser MJ . Helicobacter pylori and gastrointestinal tract adenocarcinomas. Nat Rev Cancer 2002; 2: 28–37.
Malfertheiner P, Sipponen P, Naumann M, Moayyedi P, Megraud F, Xiao SD et al. Helicobacter pylori eradication has the potential to prevent gastric cancer: a state-of-the-art critique. Am J Gastroenterol 2005; 100: 2100–2115.
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A . Global cancer statistics, 2012. CA Cancer J Clin 2015; 65: 87–108.
Camargo MC, Yepez MC, Ceron C, Guerrero N, Bravo LE, Correa P et al. Age at acquisition of Helicobacter pylori infection: comparison of two areas with contrasting risk of gastric cancer. Helicobacter 2004; 9: 262–270.
Correa P, Cuello C, Duque E, Burbano LC, Garcia FT, Bolanos O et al. Gastric cancer in Colombia. III. Natural history of precursor lesions. J Natl Cancer Inst 1976; 57: 1027–1035.
Ando T, Yoshida T, Enomoto S, Asada K, Tatematsu M, Ichinose M et al. DNA methylation of microRNA genes in gastric mucosae of gastric cancer patients: its possible involvement in the formation of epigenetic field defect. Int J Cancer 2009; 124: 2367–2374.
Ueda Y, Ando T, Nanjo S, Ushijima T, Sugiyama T . DNA methylation of microRNA-124a is a potential risk marker of colitis-associated cancer in patients with ulcerative colitis. Dig Dis Sci 2014; 59: 2444–2451.
Shi XB, Xue L, Ma AH, Tepper CG, Gandour-Edwards R, Kung HJ et al. Tumor suppressive miR-124 targets androgen receptor and inhibits proliferation of prostate cancer cells. Oncogene 2013; 32: 4130–4138.
Lujambio A, Ropero S, Ballestar E, Fraga MF, Cerrato C, Setién F et al. Genetic unmasking of an epigenetically silenced microRNA in human cancer cells. Cancer Res 2007; 67: 1424–1429.
Ushijima T, Hattori N . Molecular pathways: involvement of Helicobacter pylori-triggered inflammation in the formation of an epigenetic field defect, and its usefulness as cancer risk and exposure markers. Clin Cancer Res 2012; 18: 923–929.
Koukos G, Polytarchou C, Kaplan JL, Morley-Fletcher A, Gras-Miralles B, Kokkotou E et al. MicroRNA-124 regulates STAT3 expression and is down-regulated in colon tissues of pediatric patients with ulcerative colitis. Gastroenterology 2013; 145: 842–852, 842.
Wilting SM, van Boerdonk RA, Henken FE, Meijer CJ, Diosdado B, Meijer GA et al. Methylation-mediated silencing and tumour suppressive function of hsa-miR-124 in cervical cancer. Mol Cancer 2010; 9: 167–181.
Rider JE, Hacker A, Mackintosh CA, Pegg AE, Woster PM, Casero RA Jr . Spermine and spermidine mediate protection against oxidative damage caused by hydrogen peroxide. Amino Acids 2007; 33: 231–240.
Pegg AE, Xiong H, Feith DJ, Shantz LM . S-adenosylmethionine decarboxylase: structure, function and regulation by polyamines. Biochem Soc Trans 1998; 26: 580–586.
Hardbower DM, de Sablet T, Chaturvedi R, Wilson KT . Chronic inflammation and oxidative stress: the smoking gun for Helicobacter pylori-induced gastric cancer? Gut Microbes 2013; 4: 475–481.
Maekita T, Nakazawa K, Mihara M, Nakajima T, Yanaoka K, Iguchi M et al. High levels of aberrant DNA methylation in Helicobacter pylori-infected gastric mucosae and its possible association with gastric cancer risk. Clin Cancer Res 2006; 12: 989–995.
Nakajima T, Maekita T, Oda I, Gotoda T, Yamamoto S, Umemura S et al. Higher methylation levels in gastric mucosae significantly correlate with higher risk of gastric cancers. Cancer Epidemiol Biomarkers Prev 2006; 15: 2317–2321.
Niwa T, Tsukamoto T, Toyoda T, Mori A, Tanaka H, Maekita T et al. Inflammatory processes triggered by Helicobacter pylori infection cause aberrant DNA methylation in gastric epithelial cells. Cancer Res 2010; 70: 1430–1440.
Schneider BG, Piazuelo MB, Sicinschi LA, Mera R, Peng DF, Roa JC et al. Virulence of infecting Helicobacter pylori strains and intensity of mononuclear cell infiltration are associated with levels of DNA hypermethylation in gastric mucosae. Epigenetics 2013; 8: 1153–1161.
Schneider BG, Peng DF, Camargo MC, Piazuelo MB, Sicinschi LA, Mera R et al. Promoter DNA hypermethylation in gastric biopsies from subjects at high and low risk for gastric cancer. Int J Cancer 2010; 127: 2588–2597.
Kodaman N, Pazos A, Schneider BG, Piazuelo MB, Mera R, Sobota RS et al. Human and Helicobacter pylori coevolution shapes the risk of gastric disease. Proc Natl Acad Sci USA 2014; 111: 1455–1460.
Xie L, Zhang Z, Tan Z, He R, Zeng X, Xie Y et al. MicroRNA-124 inhibits proliferation and induces apoptosis by directly repressing EZH2 in gastric cancer. Mol Cell Biochem 2014; 392: 153–159.
Dalal RS, Moss SF . At the bedside: Helicobacter pylori, dysregulated host responses, DNA damage, and gastric cancer. J Leukoc Biol 2014; 96: 213–224.
Ford AC, Forman D, Hunt RH, Yuan Y, Moayyedi P . Helicobacter pylori eradication therapy to prevent gastric cancer in healthy asymptomatic infected individuals: systematic review and meta-analysis of randomised controlled trials. BMJ 2014; 348: 3174–3187.
Asada K, Nakajima T, Shimazu T, Yamamichi N, Maekita T, Yokoi C et al. Demonstration of the usefulness of epigenetic cancer risk prediction by a multicentre prospective cohort study. Gut 2015; 64: 388–396.
Tsai H-C, Li H, Van Neste L, Cai Y, Robert C, Rassool FV et al. Transient low doses of DNA demethylating agents exert durable anti-tumor effects on hematological and epithelial tumor cells. Cancer Cell 2012; 21: 430–446.
Wang H, Jiang Y, Peng H, Chen Y, Zhu P, Huang Y . Recent progress in microRNA delivery for cancer therapy by non-viral synthetic vectors. Adv Drug Deliv Rev 2015; 81: 142–160.
O'Hagan HM, Wang W, Sen S, Destefano Shields C, Lee SS, Zhang YW et al. Oxidative damage targets complexes containing DNA methyltransferases, SIRT1, and polycomb members to promoter CpG Islands. Cancer Cell 2011; 20: 606–619.
Wang Y, Murray-Stewart T, Devereux W, Hacker A, Frydman B, Woster PM et al. Properties of purified recombinant human polyamine oxidase, PAOh1/SMO. Biochem Biophys Res Commun 2003; 304: 605–611.
Kabra PM, Lee HK, Lubich WP, Marton LJ . Solid-phase extraction and determination of dansyl derivatives of unconjugated and acetylated polyamines by reversed-phase liquid chromatography: improved separation systems for polyamines in cerebrospinal fluid, urine and tissue. J Chromatogr 1986; 380: 19–32.
Bradford MM . A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 1976; 72: 248–254.
Betel D, Wilson M, Gabow A, Marks DS, Sander C . The microRNA.org resource: targets and expression. Nucleic Acids Res 2008; 36: D149–D153.
Lewis BP, Burge CB, Bartel DP . Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets. Cell 2005; 120: 15–20.
de Sablet T, Piazuelo MB, Shaffer CL, Schneider BG, Asim M, Chaturvedi R et al. Phylogeographic origin of Helicobacter pylori is a determinant of gastric cancer risk. Gut 2011; 60: 1189–1195.
Shimizu T, Suzuki H, Nojima M, Kitamura H, Yamamoto E, Maruyama R et al. Methylation of a panel of microRNA genes is a novel biomarker for detection of bladder cancer. Eur Urol 2013; 63: 1091–1100.
Acknowledgements
We thank Alberto Delgado for his technical expertise and performance of SMOX immunohistochemistry studies. This study was supported by National Institutes of Health Grants R01CA051085 and R01CA098454 (to RAC), R01DK053620 and R01CA190612 (to KTW), P01CA028842 (to PC and KTW), P01CA116087 (to KTW), K01AT007324 (to RC), the Samuel Waxman Cancer Research Foundation (to RAC), the Vanderbilt Digestive Disease Research Center grant (P30DK058404), UL1RR024975 (Vanderbilt CTSA, Pilot Project to KTW) and Merit Review Grant 1I01BX001453 from the Office of Medical Research, Department of Veterans Affairs (to KTW).
Author contributions
TMS, KTW and RAC designed the research; TMS, JCS, MBP, RC and BGS performed the research; LEB and PC provided unique human samples; TMS, MBP, RMM, KTW and RAC analyzed the data; and TMS, KTW and RAC wrote the paper. All authors revised and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Murray-Stewart, T., Sierra, J., Piazuelo, M. et al. Epigenetic silencing of miR-124 prevents spermine oxidase regulation: implications for Helicobacter pylori-induced gastric cancer. Oncogene 35, 5480–5488 (2016). https://doi.org/10.1038/onc.2016.91
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2016.91
This article is cited by
-
Early Leishmania infectivity depends on miR-372/373/520d family-mediated reprogramming of polyamines metabolism in THP-1-derived macrophages
Scientific Reports (2024)
-
HRCT1, negatively regulated by miR-124-3p, promotes tumor metastasis and the growth of gastric cancer by activating the ERBB2-MAPK pathway
Gastric Cancer (2023)
-
The role of polyamines in gastric cancer
Oncogene (2021)
-
SP1-activated long noncoding RNA lncRNA GCMA functions as a competing endogenous RNA to promote tumor metastasis by sponging miR-124 and miR-34a in gastric cancer
Oncogene (2020)
-
Helicobacter pylori infection induced genome instability and gastric cancer
Genome Instability & Disease (2020)