Abstract
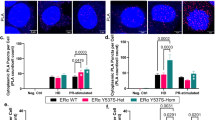
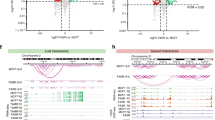
Tamoxifen has shown great success in the treatment of breast cancer; however, long-term treatment can lead to acquired tamoxifen (TOT) resistance and relapse. TOT classically antagonizes estradiol (E2) -dependent breast cancer cell growth, but exerts partial agonist/antagonist behavior on gene expression. Although both E2 and TOT treatment of breast cancer cells results in recruitment of the estrogen receptor (ER) to common and distinct genomic sites, the mechanisms and proteins underlying TOT preferential recruitment of the ER remains poorly defined. To this end, we performed in silico motif-enrichment analyses within the ER-binding peaks in response to E2 or TOT, to identify factors that would specifically recruit ER to genomic binding sites in the presence of TOT as compared to E2. Intriguingly, we found Nkx3-1 and Oct-transcription factor homodimer motifs to be enriched in TOT preferential binding sites and confirmed the critical role of Oct-3/4 (aka Oct-4) in directing ER recruitment to TOT preferential genomic binding sites, by chromatin immunoprecipitation (ChIP) analyses. Further investigation revealed Oct-4 expression to be basally repressed by Nkx3-1 in MCF-7 cells and TOT treatment appeared to elevate Nkx3-1 degradation through a p38MAPK-dependent phosphorylation of the E3 ligase, Skp2 at serine-64 residue, as observed by quantitative mass-spectrometry analyses. Consistently, Oct-4 upon induction by phospho-Ser64-Skp2-mediated proteasomal degradation of Nkx3-1, participated in ER transcriptional complexes along with p38MAPK and Skp2 in a tamoxifen-dependent manner leading to TOT-dependent gene activation and cell proliferation of the TOT-resistant MCF-7-tamr breast cancer cells. Notably, Oct-4 levels were highly elevated in MCF-7-tamr cells, and appeared critical for their TOT sensitivity in cell proliferation assays. Furthermore, overexpression of Oct-4 enhanced tumor growth in the presence of tamoxifen in mice in vivo. Collectively, our work presents a novel mechanism for tamoxifen-specific gene activation by ER, secondary to its TOT preferential recruitment to genomic sites by specific activation of Oct-4, a phenomenon that appears to underlie tamoxifen resistance in breast cancer cells and in xenograft tumor models, and could be useful in designing therapeutic interventions to improve treatment outcome.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Abbreviations
- CoIP:
-
Co-immunoprecipitation
- Ub:
-
ubiquitin
- E2:
-
estradiol
- TOT:
-
tamoxifen
- ER:
-
estrogen receptor
- Skp2:
-
S-phase Kinase-associated Protein-2
- p38MAPK:
-
p38-Mitogen Activated Protein Kinase
- NFATc4:
-
Nuclear Factor Activated T-cell 4
- siRNA:
-
short interfering RNA
- Ad:
-
Adenovirus
- ERE:
-
estrogen response element.
References
Shang Y, Hu X, DiRenzo J, Lazar MA, Brown M . Cofactor dynamics and sufficiency in estrogen receptor-regulated transcription. Cell 2000; 103: 843–852.
Green KA, Carroll JS . Oestrogen-receptor-mediated transcription and the influence of co-factors and chromatin state. Nat Rev Cancer 2007; 7: 713–722.
Fullwood MJ, Liu MH, Pan YF, Liu J, Xu H, Mohamed YB et al. An oestrogen-receptor-alpha-bound human chromatin interactome. Nature 2009; 462: 58–64.
Welboren WJ, Sweep FC, Span PN, Stunnenberg HG . Genomic actions of estrogen receptor alpha: what are the targets and how are they regulated? Endocr Relat Cancer 2009; 16: 1073–1089.
Katzenellenbogen BS, Katzenellenbogen JA . Biomedicine. Defining the "S" in SERMs. Science 2002; 295: 2380–2381.
Shang Y, Brown M . Molecular determinants for the tissue specificity of SERMs. Science 2002; 295: 2465–2468.
Katzenellenbogen BS, Frasor J . Therapeutic targeting in the estrogen receptor hormonal pathway. Semin Oncol 2004; 31: 28–38.
Brueggemeier RW, Hackett JC, Diaz-Cruz ES . Aromatase inhibitors in the treatment of breast cancer. Endocr Rev 2005; 26: 331–345.
Jordan VC, O'Malley BW . Selective estrogen-receptor modulators and antihormonal resistance in breast cancer. J Clin Oncol 2007; 25: 5815–5824.
Jaiyesimi IA, Buzdar AU, Decker DA, Hortobagyi GN . Use of tamoxifen for breast cancer: twenty-eight years later. J Clin Oncol 1995; 13: 513–529.
Herman ME, Katzenellenbogen BS . Response-specific antiestrogen resistance in a newly characterized MCF-7 human breast cancer cell line resulting from long-term exposure to trans-hydroxytamoxifen. J Steroid Biochem Mol Biol 1996; 59: 121–134.
Connor CE, Norris JD, Broadwater G, Willson TM, Gottardis MM, Dewhirst MW et al. Circumventing tamoxifen resistance in breast cancers using antiestrogens that induce unique conformational changes in the estrogen receptor. Cancer Res 2001; 61: 2917–2922.
Osborne CK . Tamoxifen in the treatment of breast cancer. N Engl J Med 1998; 339: 1609–1618.
Bianco S, Gevry N . Endocrine resistance in breast cancer: from cellular signaling pathways to epigenetic mechanisms. Transcription 2012; 3: 165–170.
Huber-Keener KJ, Liu X, Wang Z, Wang Y, Freeman W, Wu S et al. Differential gene expression in tamoxifen-resistant breast cancer cells revealed by a new analytical model of RNA-Seq data. PLoS One 2012; 7: e41333.
Frasor J, Stossi F, Danes JM, Komm B, Lyttle CR, Katzenellenbogen BS . Selective estrogen receptor modulators: discrimination of agonistic versus antagonistic activities by gene expression profiling in breast cancer cells. Cancer Res 2004; 64: 1522–1533.
Gutierrez MC, Detre S, Johnston S, Mohsin SK, Shou J, Allred DC et al. Molecular changes in tamoxifen-resistant breast cancer: relationship between estrogen receptor, HER-2, and p38 mitogen-activated protein kinase. J Clin Oncol 2005; 23: 2469–2476.
Wang LH, Yang XY, Zhang X, An P, Kim HJ, Huang J et al. Disruption of estrogen receptor DNA-binding domain and related intramolecular communication restores tamoxifen sensitivity in resistant breast cancer. Cancer Cell 2006; 10: 487–499.
Bayliss J, Hilger A, Vishnu P, Diehl K, El-Ashry D . Reversal of the estrogen receptor negative phenotype in breast cancer and restoration of antiestrogen response. Clin Cancer Res 2007; 13: 7029–7036.
Hurtado A, Holmes KA, Geistlinger TR, Hutcheson IR, Nicholson RI, Brown M et al. Regulation of ERBB2 by oestrogen receptor-PAX2 determines response to tamoxifen. Nature 2008; 456: 663–666.
Hurtado A, Holmes KA, Ross-Innes CS, Schmidt D, Carroll JS . FOXA1 is a key determinant of estrogen receptor function and endocrine response. Nat Genet 2011; 43: 27–33.
Iorns E, Turner NC, Elliott R, Syed N, Garrone O, Gasco M et al. Identification of CDK10 as an important determinant of resistance to endocrine therapy for breast cancer. Cancer Cell 2008; 13: 91–104.
Musgrove EA, Sutherland RL . Biological determinants of endocrine resistance in breast cancer. Nat Rev Cancer 2009; 9: 631–643.
Lupien M, Brown M . Cistromics of hormone-dependent cancer. Endocr Relat Cancer 2009; 16: 381–389.
Hoskins JM, Carey LA, McLeod HL . CYP2D6 and tamoxifen: DNA matters in breast cancer. Nat Rev Cancer 2009; 9: 576–586.
de Leeuw R, Flach K, Bentin Toaldo C, Alexi X, Canisius S, Neefjes J et al. PKA phosphorylation redirects ERalpha to promoters of a unique gene set to induce tamoxifen resistance. Oncogene 2013; 32: 3543–3551.
Beelen K, Zwart W, Linn SC . Can predictive biomarkers in breast cancer guide adjuvant endocrine therapy? Nat Rev Clin Oncol 2012; 9: 529–541.
Bergamaschi A, Christensen BL, Katzenellenbogen BS . Reversal of endocrine resistance in breast cancer: interrelationships among 14-3-3zeta, FOXM1, and a gene signature associated with mitosis. Breast Cancer Res 2011; 13: R70.
Carroll JS, Meyer CA, Song J, Li W, Geistlinger TR, Eeckhoute J et al. Genome-wide analysis of estrogen receptor binding sites. Nat Genet 2006; 38: 1289–1297.
Welboren WJ, van Driel MA, Janssen-Megens EM, van Heeringen SJ, Sweep FC, Span PN et al. ChIP-Seq of ERalpha and RNA polymerase II defines genes differentially responding to ligands. Embo J 2009; 28: 1418–1428.
Ross-Innes CS, Stark R, Teschendorff AE, Holmes KA, Ali HR, Dunning MJ et al. Differential oestrogen receptor binding is associated with clinical outcome in breast cancer. Nature 2012; 481: 389–393.
Cardozo T, Pagano M . The SCF ubiquitin ligase: insights into a molecular machine. Nat Rev Mol Cell Biol 2004; 5: 739–751.
Lin HK, Wang G, Chen Z, Teruya-Feldstein J, Liu Y, Chan CH et al. Phosphorylation-dependent regulation of cytosolic localization and oncogenic function of Skp2 by Akt/PKB. Nat Cell Biol 2009; 11: 420–432.
Bhatt S, Xiao Z, Meng Z, Katzenellenbogen BS . Phosphorylation by p38 mitogen-activated protein kinase promotes estrogen receptor alpha turnover and functional activity via the SCF(Skp2) proteasomal complex. Mol Cell Biol 2012; 32: 1928–1943.
Jin T, Branch DR, Zhang X, Qi S, Youngson B, Goss PE . Examination of POU homeobox gene expression in human breast cancer cells. Int J Cancer 1999; 81: 104–112.
Pesce M, Scholer HR . Oct-4: gatekeeper in the beginnings of mammalian development. Stem Cells 2001; 19: 271–278.
Tai MH, Chang CC, Kiupel M, Webster JD, Olson LK, Trosko JE . Oct4 expression in adult human stem cells: evidence in support of the stem cell theory of carcinogenesis. Carcinogenesis 2005; 26: 495–502.
Loh YH, Wu Q, Chew JL, Vega VB, Zhang W, Chen X et al. The Oct4 and Nanog transcription network regulates pluripotency in mouse embryonic stem cells. Nat Genet 2006; 38: 431–440.
Monk M, Holding C . Human embryonic genes re-expressed in cancer cells. Oncogene 2001; 20: 8085–8091.
Gidekel S, Pizov G, Bergman Y, Pikarsky E . Oct-3/4 is a dose-dependent oncogenic fate determinant. Cancer Cell 2003; 4: 361–370.
Ramos-Mejia V, Escalante-Alcalde D, Kunath T, Ramirez L, Gertsenstein M, Nagy A et al. Phenotypic analyses of mouse embryos with ubiquitous expression of Oct4: effects on mid-hindbrain patterning and gene expression. Dev Dyn 2005; 232: 180–190.
Nichols J, Zevnik B, Anastassiadis K, Niwa H, Klewe-Nebenius D, Chambers I et al. Formation of pluripotent stem cells in the mammalian embryo depends on the POU transcription factor Oct4. Cell 1998; 95: 379–391.
Holmes KA, Song JS, Liu XS, Brown M, Carroll JS . Nkx3-1 and LEF-1 function as transcriptional inhibitors of estrogen receptor activity. Cancer Res 2008; 68: 7380–7385.
Bowen C, Bubendorf L, Voeller HJ, Slack R, Willi N, Sauter G et al. Loss of NKX3.1 expression in human prostate cancers correlates with tumor progression. Cancer Res 2000; 60: 6111–6115.
Kim MJ, Bhatia-Gaur R, Banach-Petrosky WA, Desai N, Wang Y, Hayward SW et al. Nkx3.1 mutant mice recapitulate early stages of prostate carcinogenesis. Cancer Res 2002; 62: 2999–3004.
Kim MJ, Cardiff RD, Desai N, Banach-Petrosky WA, Parsons R, Shen MM et al. Cooperativity of Nkx3.1 and Pten loss of function in a mouse model of prostate carcinogenesis. Proc Natl Acad Sci USA 2002; 99: 2884–2889.
Massarweh S, Osborne CK, Creighton CJ, Qin L, Tsimelzon A, Huang S et al. Tamoxifen resistance in breast tumors is driven by growth factor receptor signaling with repression of classic estrogen receptor genomic function. Cancer Res 2008; 68: 826–833.
Coser KR, Wittner BS, Rosenthal NF, Collins SC, Melas A, Smith SL et al. Antiestrogen-resistant subclones of MCF-7 human breast cancer cells are derived from a common monoclonal drug-resistant progenitor. Proc Natl Acad Sci USA 2009; 106: 14536–14541.
Allgood VE, Zhang Y, O'Malley BW, Weigel NL . Analysis of chicken progesterone receptor function and phosphorylation using an adenovirus-mediated procedure for high-efficiency DNA transfer. Biochemistry 1997; 36: 224–232.
Metivier R, Penot G, Hubner MR, Reid G, Brand H, Kos M et al. Estrogen receptor-alpha directs ordered, cyclical, and combinatorial recruitment of cofactors on a natural target promoter. Cell 2003; 115: 751–763.
Ogawa S, Lozach J, Benner C, Pascual G, Tangirala RK, Westin S et al. Molecular determinants of crosstalk between nuclear receptors and toll-like receptors. Cell 2005; 122: 707–721.
Acknowledgements
We thank Dr Peter Yau, Biotechnology Core, UIUC, for contributions to the mass-spectrometry analyses. This work was supported by grants from the NIH 5RO1 CA18119 (BSK), The Breast Cancer Research Foundation (BSK) and Juvenile Diabetes Research Foundation (GW). SB conceived of, designed and executed the study, performed experiments, data analyses and interpretation and wrote the manuscript. JDS conceived the study, performed computational analysis and contributed to experimental data. GW and SJ contributed to experimental data. BSK conceived and designed the study, performed data analyses and interpretation and wrote the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Bhatt, S., Stender, J., Joshi, S. et al. OCT-4: a novel estrogen receptor-α collaborator that promotes tamoxifen resistance in breast cancer cells. Oncogene 35, 5722–5734 (2016). https://doi.org/10.1038/onc.2016.105
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2016.105
This article is cited by
-
One-Pot Biopreparation of Trimetallic ZnO–MgO–CuO Nanoparticles: Enhanced Cytotoxicity, Antibacterial Activities and Molecular Docking Studies
Chemistry Africa (2024)
-
circRNA-SFMBT2 orchestrates ERα activation to drive tamoxifen resistance in breast cancer cells
Cell Death & Disease (2023)
-
USP22 positively modulates ERα action via its deubiquitinase activity in breast cancer
Cell Death & Differentiation (2020)
-
Oncoprotein HBXIP induces PKM2 via transcription factor E2F1 to promote cell proliferation in ER-positive breast cancer
Acta Pharmacologica Sinica (2019)
-
Oncoprotein HBXIP enhances HOXB13 acetylation and co-activates HOXB13 to confer tamoxifen resistance in breast cancer
Journal of Hematology & Oncology (2018)