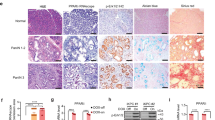
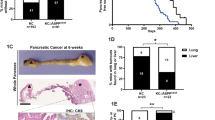
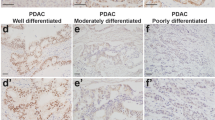
Abstract
Smoking is an established risk factor for pancreatic cancer (PC), but late diagnosis limits the evaluation of its mechanistic role in the progression of PC. We used a well-established genetically engineered mouse model (LSL-K-rasG12D) of PC to elucidate the role of smoking during initiation and development of pancreatic intraepithelial neoplasia (PanIN). The 10-week-old floxed mice (K-rasG12D; Pdx-1cre) and their control unfloxed (LSL-K-rasG12D) littermates were exposed to cigarette smoke (total suspended particles: 150 mg/m3) for 20 weeks. Smoke exposure significantly accelerated the development of PanIN lesions in the floxed mice, which correlated with tenfold increase in the expression of cytokeratin19. The systemic accumulation of myeloid-derived suppressor cells (MDSCs) decreased significantly in floxed mice compared with unfloxed controls (P<0.01) after the smoke exposure with the concurrent increase in the macrophage (P<0.05) and dendritic cell (DCs) (P<0.01) population. Further, smoking-induced inflammation (IFN-γ, CXCL2; P<0.05) was accompanied by enhanced activation of pancreatic stellate cells and elevated levels of serum retinoic acid-binding protein 4, indicating increased bioavailability of retinoic acid which contributes to differentiation of MDSCs to tumor-associated macrophages (TAMs) and DCs. TAMs predominantly contribute to the increased expression of heparin-binding epidermal growth factor-like growth factor (EGFR ligand) in pre-neoplastic lesions in smoke-exposed floxed mice that facilitate acinar-to-ductal metaplasia (ADM). Further, smoke exposure also resulted in partial suppression of the immune system early during PC progression. Overall, the present study provides a novel mechanism of smoking-induced increase in ADM in the presence of constitutively active K-ras mutation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Braat H, Bruno M, Kuipers EJ, Peppelenbosch MP . Pancreatic cancer: promise for personalised medicine? Cancer Lett 2012; 318: 1–8.
Siegel R, Naishadham D, Jemal A . Cancer statistics, 2012. CA Cancer J Clin 2012; 62: 10–29.
Maitra A, Hruban RH . Pancreatic cancer. Annu Rev Pathol 2008; 3: 157–188.
Gabrilovich DI, Nagaraj S . Myeloid-derived suppressor cells as regulators of the immune system. Nat Rev Immunol 2009; 9: 162–174.
Toh B, Wang X, Keeble J, Sim WJ, Khoo K, Wong WC et al. Mesenchymal transition and dissemination of cancer cells is driven by myeloid-derived suppressor cells infiltrating the primary tumor. PLoS Biol 2011; 9: e1001162.
Zhao F, Obermann S, von WR, Haile L, Manns MP, Korangy F et al. Increase in frequency of myeloid-derived suppressor cells in mice with spontaneous pancreatic carcinoma. Immunology 2009; 128: 141–149.
Huang B, Pan PY, Li Q, Sato AI, Levy DE, Bromberg J et al. Gr-1+CD115+ immature myeloid suppressor cells mediate the development of tumor-induced T regulatory cells and T-cell anergy in tumor-bearing host. Cancer Res 2006; 66: 1123–1131.
Bracci PM . Obesity and pancreatic cancer: overview of epidemiologic evidence and biologic mechanisms. Mol Carcinog 2012; 51: 53–63.
Duell EJ, Holly EA, Bracci PM, Liu M, Wiencke JK, Kelsey KT . A population-based, case-control study of polymorphisms in carcinogen-metabolizing genes, smoking, and pancreatic adenocarcinoma risk. J Natl Cancer Inst 2002; 94: 297–306.
Duell EJ . Epidemiology and potential mechanisms of tobacco smoking and heavy alcohol consumption in pancreatic cancer. Mol Carcinog 2012; 51: 40–52.
Sopori M . Effects of cigarette smoke on the immune system. Nat Rev Immunol 2002; 2: 372–377.
Sopori ML, Kozak W . Immunomodulatory effects of cigarette smoke. J Neuroimmunol 1998; 83: 148–156.
Zeidel A, Beilin B, Yardeni I, Mayburd E, Smirnov G, Bessler H . Immune response in asymptomatic smokers. Acta Anaesthesiol Scand 2002; 46: 959–964.
Blackford A, Parmigiani G, Kensler TW, Wolfgang C, Jones S, Zhang X et al. Genetic mutations associated with cigarette smoking in pancreatic cancer. Cancer Res 2009; 69: 3681–3688.
Malfertheiner P, Schutte K . Smoking—a trigger for chronic inflammation and cancer development in the pancreas. Am J Gastroenterol 2006; 101: 160–162.
Nouri-Shirazi M, Guinet E . Evidence for the immunosuppressive role of nicotine on human dendritic cell functions. Immunology 2003; 109: 365–373.
Hingorani SR, Petricoin EF, Maitra A, Rajapakse V, King C, Jacobetz MA et al. Preinvasive and invasive ductal pancreatic cancer and its early detection in the mouse. Cancer Cell 2003; 4: 437–450.
Hingorani SR, Wang L, Multani AS, Combs C, Deramaudt TB, Hruban RH et al. Trp53R172H and KrasG12D cooperate to promote chromosomal instability and widely metastatic pancreatic ductal adenocarcinoma in mice. Cancer Cell 2005; 7: 469–483.
Elliott MK, Sisson JH, West WW, Wyatt TA . Differential in vivo effects of whole cigarette smoke exposure versus cigarette smoke extract on mouse ciliated tracheal epithelium. Exp Lung Res 2006; 32: 99–118.
Hengesbach LM, Hoag KA . Physiological concentrations of retinoic acid favor myeloid dendritic cell development over granulocyte development in cultures of bone marrow cells from mice. J Nutr 2004; 134: 2653–2659.
Kusmartsev S, Cheng F, Yu B, Nefedova Y, Sotomayor E, Lush R et al. All-trans-retinoic acid eliminates immature myeloid cells from tumor-bearing mice and improves the effect of vaccination. Cancer Res 2003; 63: 4441–4449.
Nefedova Y, Fishman M, Sherman S, Wang X, Beg AA, Gabrilovich DI . Mechanism of all-trans retinoic acid effect on tumor-associated myeloid-derived suppressor cells. Cancer Res 2007; 67: 11021–11028.
Apte MV, Haber PS, Applegate TL, Norton ID, McCaughan GW, Korsten MA et al. Periacinar stellate shaped cells in rat pancreas: identification, isolation, and culture. Gut 1998; 43: 128–133.
Bachem MG, Schneider E, Gross H, Weidenbach H, Schmid RM, Menke A et al. Identification, culture, and characterization of pancreatic stellate cells in rats and humans. Gastroenterology 1998; 115: 421–432.
McCarroll JA, Phillips PA, Santucci N, Pirola RC, Wilson JS, Apte MV . Vitamin A inhibits pancreatic stellate cell activation: implications for treatment of pancreatic fibrosis. Gut 2006; 55: 79–89.
McCarroll JA, Phillips PA, Park S, Doherty E, Pirola RC, Wilson JS et al. Pancreatic stellate cell activation by ethanol and acetaldehyde: is it mediated by the mitogen-activated protein kinase signaling pathway? Pancreas 2003; 27: 150–160.
Habisch H, Zhou S, Siech M, Bachem MG . Interaction of stellate cells with pancreatic carcinoma cells. Cancers (Basel) 2010; 2: 1661–1682.
Hutchison SK, Harrison C, Stepto N, Meyer C, Teede HJ . Retinol-binding protein 4 and insulin resistance in polycystic ovary syndrome. Diabetes Care 2008; 31: 1427–1432.
Manicassamy S, Pulendran B . Retinoic acid-dependent regulation of immune responses by dendritic cells and macrophages. Semin Immunol 2009; 21: 22–27.
Mirza N, Fishman M, Fricke I, Dunn M, Neuger AM, Frost TJ et al. All-trans-retinoic acid improves differentiation of myeloid cells and immune response in cancer patients. Cancer Res 2006; 66: 9299–9307.
Ding Q, Jin T, Wang Z, Chen Y . Catalase potentiates retinoic acid-induced THP-1 monocyte differentiation into macrophage through inhibition of peroxisome proliferator-activated receptor gamma. J Leukoc Biol 2007; 81: 1568–1576.
Zhang QW, Liu L, Gong CY, Shi HS, Zeng YH, Wang XZ et al. Prognostic significance of tumor-associated macrophages in solid tumor: a meta-analysis of the literature. PLoS One 2012; 7: e50946.
Ray KC, Moss ME, Franklin JL, Weaver CJ, Higginbotham J, Song Y et al. Heparin-binding epidermal growth factor-like growth factor eliminates constraints on activated Kras to promote rapid onset of pancreatic neoplasia. Oncogene 2013; 33: 823–831.
Means AL, Ray KC, Singh AB, Washington MK, Whitehead RH, Harris RC Jr et al. Overexpression of heparin-binding EGF-like growth factor in mouse pancreas results in fibrosis and epithelial metaplasia. Gastroenterology 2003; 124: 1020–1036.
Navas C, Hernandez-Porras I, Schuhmacher AJ, Sibilia M, Guerra C, Barbacid M . EGF receptor signaling is essential for k-ras oncogene-driven pancreatic ductal adenocarcinoma. Cancer Cell 2012; 22: 318–330.
Kroening PR, Barnes TW, Pease L, Limper A, Kita H, Vassallo R . Cigarette smoke-induced oxidative stress suppresses generation of dendritic cell IL-12 and IL-23 through ERK-dependent pathways. J Immunol 2008; 181: 1536–1547.
Zhang S, Petro TM . The effect of nicotine on murine CD4 T cell responses. Int J Immunopharmacol 1996; 18: 467–478.
Clark CE, Hingorani SR, Mick R, Combs C, Tuveson DA, Vonderheide RH . Dynamics of the immune reaction to pancreatic cancer from inception to invasion. Cancer Res 2007; 67: 9518–9527.
Colby JK, Klein RD, McArthur MJ, Conti CJ, Kiguchi K, Kawamoto T et al. Progressive metaplastic and dysplastic changes in mouse pancreas induced by cyclooxygenase-2 overexpression. Neoplasia 2008; 10: 782–796.
Bayne LJ, Beatty GL, Jhala N, Clark CE, Rhim AD, Stanger BZ et al. Tumor-derived granulocyte-macrophage colony-stimulating factor regulates myeloid inflammation and T cell immunity in pancreatic cancer. Cancer Cell 2012; 21: 822–835.
Pylayeva-Gupta Y, Lee KE, Hajdu CH, Miller G, Bar-Sagi D . Oncogenic Kras-induced GM-CSF production promotes the development of pancreatic neoplasia. Cancer Cell 2012; 21: 836–847.
Breitman TR, Collins SJ, Keene BR . Terminal differentiation of human promyelocytic leukemic cells in primary culture in response to retinoic acid. Blood 1981; 57: 1000–1004.
van Bockstaele DR, Lenjou M, Snoeck HW, Lardon F, Stryckmans P, Peetermans ME . Direct effects of 13-cis and all-trans retinoic acid on normal bone marrow (BM) progenitors: comparative study on BM mononuclear cells and on isolated CD34+ BM cells. Ann Hematol 1993; 66: 61–66.
Kuwata T, Wang IM, Tamura T, Ponnamperuma RM, Levine R, Holmes KL et al. Vitamin A deficiency in mice causes a systemic expansion of myeloid cells. Blood 2000; 95: 3349–3356.
Froeling FE, Feig C, Chelala C, Dobson R, Mein CE, Tuveson DA et al. Retinoic acid-induced pancreatic stellate cell quiescence reduces paracrine Wnt-beta-catenin signaling to slow tumor progression. Gastroenterology 2011; 141: 1486–1497.
van Geenen EJ, Smits MM, Schreuder TC, van der Peet DL, Bloemena E, Mulder CJ . Smoking is related to pancreatic fibrosis in humans. Am J Gastroenterol 2011; 106: 1161–1166.
Mohty M, Morbelli S, Isnardon D, Sainty D, Arnoulet C, Gaugler B et al. All-trans retinoic acid skews monocyte differentiation into interleukin-12-secreting dendritic-like cells. Br J Haematol 2003; 122: 829–836.
Goswami S, Sahai E, Wyckoff JB, Cammer M, Cox D, Pixley FJ et al. Macrophages promote the invasion of breast carcinoma cells via a colony-stimulating factor-1/epidermal growth factor paracrine loop. Cancer Res 2005; 65: 5278–5283.
Edwards JP, Zhang X, Frauwirth KA, Mosser DM . Biochemical and functional characterization of three activated macrophage populations. J Leukoc Biol 2006; 80: 1298–1307.
Moniaux N, Chakraborty S, Yalniz M, Gonzalez J, Shostrom VK, Standop J et al. Early diagnosis of pancreatic cancer: neutrophil gelatinase-associated lipocalin as a marker of pancreatic intraepithelial neoplasia. Br J Cancer 2008; 98: 1540–1547.
Singh AP, Moniaux N, Chauhan SC, Meza JL, Batra SK . Inhibition of MUC4 expression suppresses pancreatic tumor cell growth and metastasis. Cancer Res 2004; 64: 622–630.
Momi N, Ponnusamy MP, Kaur S, Rachagani S, Kunigal SS, Chellappan S et al. Nicotine/cigarette smoke promotes metastasis of pancreatic cancer through alpha7nAChR-mediated MUC4 upregulation. Oncogene 2013; 32: 1384–1395.
Mathison A, Liebl A, Bharucha J, Mukhopadhyay D, Lomberk G, Shah V et al. Pancreatic stellate cell models for transcriptional studies of desmoplasia-associated genes. Pancreatology 2010; 10: 505–516.
Acknowledgements
We are grateful to Dr Patrick C Swanson, Creighton University, Omaha, for suggestions. We appreciate the UNMC Cell Analysis Facility for analysis and graduate students and technician Erik Moore from Dr Batra’s lab for help in sample preparation. This work was supported in part by the grant from National Institute of Health (R01CA78590, EDRN U01 CA111294, R01 CA133774, R01 CA131944, SPORE P50 CA127297, T32 CA009476, P20 GM103480, R21 CA156037 and U54 TMEN CA163120).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Kumar, S., Torres, M., Kaur, S. et al. Smoking accelerates pancreatic cancer progression by promoting differentiation of MDSCs and inducing HB-EGF expression in macrophages. Oncogene 34, 2052–2060 (2015). https://doi.org/10.1038/onc.2014.154
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2014.154
This article is cited by
-
TRAF3/STAT6 axis regulates macrophage polarization and tumor progression
Cell Death & Differentiation (2023)
-
Experimental models of pancreatic cancer desmoplasia
Laboratory Investigation (2018)
-
Variation in KRAS driver substitution distributions between tumor types is determined by both mutation and natural selection
Scientific Reports (2016)
-
Conditional survival of pancreatic ductal adenocarcinoma in surgical and nonsurgical patients: a retrospective analysis report from a single institution in China
World Journal of Surgical Oncology (2015)