Abstract
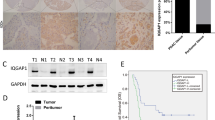
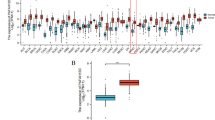
Pancreatic ductal adenocarcinoma (PDAC) is the eighth largest cause of cancer-related mortality across the world, with a median 5-year survival rate of less than 3.5%. This is partly because the molecules and the molecular mechanisms that contribute to PDAC are not well understood. Our goal is to understand the role of p21-activated kinase 1 (Pak1) signaling axis in the progression of PDAC. Pak1, a serine/threonine kinase, is a well-known regulator of cytoskeletal remodeling, cell motility, cell proliferation and cell survival. Recent reports suggest that Pak1 by itself can have an oncogenic role in a wide variety of cancers. In this study, we analyzed the expression of Pak1 in human pancreatic cancer tissues and found that Pak1 levels are significantly upregulated in PDAC samples as compared with adjacent normals. Further, to study the functional role of Pak1 in pancreatic cancer model systems, we developed stable overexpression and lentiviral short hairpin RNA-mediated knockdown (KD) clones of Pak1 and studied the changes in transforming properties of the cells. We also observed that Pak1 KD clones failed to form tumors in nude mice. By adopting a quantitative PCR array-based approach, we identified fibronectin, a component of the extracellular matrix and a mesenchymal marker, as a transcriptional target of Pak1 signaling. The underlying molecular mechanism of Pak1-mediated transformation includes its nuclear import and recruitment to the fibronectin promoter via interaction with nuclear factor-κB (NF-κB)–p65 complex. To our knowledge, this is the first study illustrating Pak1–NF-κB–p65-mediated fibronectin regulation as a potent tumor-promoting mechanism in KRAS intact model.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
World Cancer Research Fund/American Institute for Cancer Research. Continuous Update Project Summary. Food, Nutrition, Physical Activity, and the Prevention of Pancreatic Cancer 2012.
Hidalgo M, Von Hoff DD . Translational therapeutic opportunities in ductal adenocarcinoma of the pancreas. Clin Cancer Res 2012; 18: 4249–4256.
Jemal A, Siegel R, Xu J, Ward E . Cancer statistics, 2010. CA Cancer J Clin. 2010; 60: 277–300.
Vincent A, Herman J, Schulick R, Hruban RH, Goggins M . Pancreatic cancer. Lancet 2011; 378: 607–620.
Maitra A, Hurban RH . Pancreatic cancer. Annu Rev Pathol 2008; 3: 157–188.
Costello E, Greenhalf W, Neoptolemos JP . New biomarkers and targets in pancreatic cancer and their application to treatment. Nat Rev Gastroenterol Hepatol 2012; 9: 435–444.
Yeh JJ, Der CJ . Targeting signal transduction in pancreatic cancer treatment. Expert Opin Ther Targets 2007; 11: 673–694.
Krautz C, Rückert F, Saeger HD, Pilarsky C, Grützmann R . An update on molecular research of pancreatic adenocarcinoma. Anticancer Agents Med Chem 2011; 11: 411–417.
Falasca M, Selvaggi F, Buus R, Sulpizio S, Edling CE . Targeting phosphoinositide 3-kinase pathways in pancreatic cancer—from molecular signalling to clinical trials. Anticancer Agents Med Chem 2011; 11: 455–463.
Almoguera C, Shibata D, Forrester K, Martin J, Arnheim N, Perucho M . Most human carcinomas of the exocrine pancreas contain mutant c-K-ras genes. Cell 1988; 53: 549–554.
Morris JP 4th, Wang SC, Hebrok M . KRAS, Hedgehog, Wnt and the twisted developmental biology of pancreatic ductal adenocarcinoma. Nat Rev Cancer 2010; 10: 683–695.
Bokoch GM . Biology of the p21-activated kinases. Annu Rev Biochem 2003; 72: 743–781.
Kumar R, Gururaj AE, Barnes CJ . p21-activated kinases in cancer. Nat Rev Cancer 2006; 6: 459–471.
Arias-Romero LE, Chernoff J . A tale of two Paks. Biol Cell 2008; 100: 97–108.
Balasenthil S, Sahin AA, Barnes CJ, Wang RA, Pestell RG, Vadlamudi RK et al. p21-activated kinase-1 signaling mediates cyclin D1 expression in mammary epithelial and cancer cells. J Biol Chem 2004; 279: 1422–1428.
Fernandez-Zapico ME, Gonzalez-Paz NC, Weiss E, Savoy DN, Molina JR, Fonseca R et al. Ectopic expression of VAV1 reveals an unexpected role in pancreatic cancer tumorigenesis. Cancer Cell 2005; 7: 39–49.
Crnogorac-Jurcevic T, Efthimiou E, Capelli P, Blaveri E, Baron A, Terris B et al. Gene expression profiles of pancreatic cancer and stromal desmoplasia. Oncogene 2001; 20: 7437–7446.
Kimmelman AC, Hezel AF, Aguirre AJ, Zheng H, Paik JH, Ying H et al. Genomic alterations link Rho family of GTPases to the highly invasive phenotype of pancreas cancer. Proc Natl Acad Sci USA 2008; 105: 19372–19377.
Ungefroren H, Groth S, Sebens S, Lehnert H, Gieseler F, Fändrich F . Differential roles of Smad2 and Smad3 in the regulation of TGF-β1-mediated growth inhibition and cell migration in pancreatic ductal adenocarcinoma cells: control by Rac1. Mol Cancer 2011; 10: 67.
Menard RE, Mattingly RR . Cell surface receptors activate p21-activated kinase 1 via multiple Ras and PI3-kinase-dependent pathways. Cell Signal 2003; 15: 1099–1109.
Wu R, Abramson AL, Symons MH, Steinberg BM . Pak1 and Pak2 are activated in recurrent respiratory papillomas, contributing to one pathway of Rac1-mediated COX-2 expression. Int J Cancer 2010; 127: 2230–2237.
He H, Shulkes A, Baldwin GS . PAK1 interacts with beta-catenin and is required for the regulation of the beta-catenin signalling pathway by gastrins. Biochim Biophys Acta 2008; 1783: 1943–1954.
Singh RR, Song C, Yang Z, Kumar R . Nuclear localization and chromatin targets of p21-activated kinase 1. J Biol Chem 2005; 280: 18130–18137.
Sánchez-Solana B, Motwani M, Li DQ, Eswaran J, Kumar R . p21-activated kinase-1 signaling regulates transcription of tissue factor and tissue factor pathway inhibitor. J Biol Chem 2012; 287: 39291–39302.
Frost JA, Swantek JL, Stippec S, Yin MJ, Gaynor R, Cobb MH . Stimulation of NF-κB activity by multiple signaling pathways requires PAK1. J Biol Chem 2000; 275: 19693–19699.
Stanisavljevic J, Porta-de-la-Riva M, Batlle R, de Herreros AG, Baulida J . The p65 subunit of NF-κB and PARP1 assist Snail1 in activating fibronectin transcription. J Cell Sci 2011; 124: 4161–4171.
Navas C, Hernández-Porras I, Schuhmacher AJ, Sibilia M, Guerra C, Barbacid M . EGF receptor signaling is essential for k-ras oncogene-driven pancreatic ductal adenocarcinoma. Cancer Cell 2012; 22: 318–330.
Kichina JV, Goc A, Al-Husein B, Somanath PR, Kandel ES . PAK1 as a therapeutic target. Expert Opin Ther Targets 2010; 14: 703–725.
Ong CC, Jubb AM, Zhou W, Haverty PM, Harris AL, Belvin M et al. p21-activated kinase 1: PAK'ed with potential. Oncotarget 2011; 2: 491–496.
Ong CC, Jubb AM, Haverty PM, Zhou W, Tran V, Truong T et al. Targeting p21-activated kinase 1 (PAK1) to induce apoptosis of tumor cells. Proc Natl Acad Sci USA 2011; 108: 7177–7182.
Ye DZ, Field J . PAK signaling in cancer. Cell Logist 2012; 2: 105–116.
Linder S, Castaños-Velez E, von Rosen A, Biberfeld P . Immunohistochemical expression of extracellular matrix proteins and adhesion molecules in pancreatic carcinoma. Hepatogastroenterology 2001; 48: 1321–1327.
Mao Y, Schwarzbauer JE . Stimulatory effects of a three-dimensional microenvironment on cell-mediated fibronectin fibrillogenesis. J Cell Sci 2005; 118: 4427–4436.
Holm C, Rayala S, Jirström K, Stål O, Kumar R, Landberg G . Association between Pak1 expression and subcellular localization and tamoxifen resistance in breast cancer patients. J Natl Cancer Inst 2006; 98: 671–680.
Acknowledgements
We thank Silpa for help in mutagenesis and cloning and Swarnalatha for help with Pak1 lentiviral clones. Thanks to Sekar Sathiya, Rohan Prasad, Hemadev and the entire team of the Centre for Toxicology and Developmental Research (CEFT), Sri Ramachandra University, Chennai, for help with animal experiments. We thank the Department of Biotechnology (DBT), Government of India for the financial support to SKR (grant no.: BT/PR13559/Med/30/283/2010) and Indian Institute of Technology Madras (IITM) for all other facilities.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Jagadeeshan, S., Krishnamoorthy, Y., Singhal, M. et al. Transcriptional regulation of fibronectin by p21-activated kinase-1 modulates pancreatic tumorigenesis. Oncogene 34, 455–464 (2015). https://doi.org/10.1038/onc.2013.576
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2013.576
Keywords
This article is cited by
-
Mechanisms of obesity- and diabetes mellitus-related pancreatic carcinogenesis: a comprehensive and systematic review
Signal Transduction and Targeted Therapy (2023)
-
In Vitro Modeling of the Tumor Microenvironment in Tumor Organoids
Tissue Engineering and Regenerative Medicine (2020)
-
KIBRA attains oncogenic activity by repressing RASSF1A
British Journal of Cancer (2017)
-
Signaling coupled epigenomic regulation of gene expression
Oncogene (2017)
-
Transcriptional regulation of ataxia–telangiectasia and Rad3-related protein by activated p21-activated kinase-1 protects keratinocytes in UV-B-induced premalignant skin lesions
Oncogene (2017)