Abstract
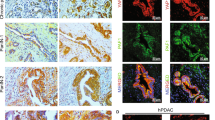
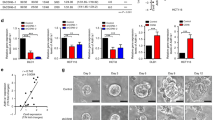
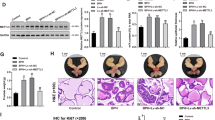
Disruption of glandular architecture associates with poor clinical outcome in high-grade colorectal cancer (CRC). Phosphatase and tensin homolog deleted on chromosome ten (PTEN) regulates morphogenic growth of benign MDCK (Madin Darby Canine Kidney) cells through effects on the Rho-like GTPase cdc42 (cell division cycle 42). This study investigates PTEN-dependent morphogenesis in a CRC model. Stable short hairpin RNA knockdown of PTEN in Caco-2 cells influenced expression or localization of cdc42 guanine nucleotide exchange factors and inhibited cdc42 activation. Parental Caco-2 cells formed regular hollow gland-like structures (glands) with a single central lumen, in three-dimensional (3D) cultures. Conversely, PTEN-deficient Caco-2 ShPTEN cells formed irregular glands with multiple abnormal lumens as well as intra- and/or intercellular vacuoles evocative of the high-grade CRC phenotype. Effects of targeted treatment were investigated. Phosphatidinylinositol 3-kinase (PI3K) modulating treatment did not affect gland morphogenesis but did influence gland number, gland size and/or cell size within glands. As PTEN may be regulated by the nuclear receptor peroxisome proliferator-activated receptor-γ (PPARγ), cultures were treated with the PPARγ ligand rosiglitazone. This treatment enhanced PTEN expression, cdc42 activation and rescued dysmorphogenesis by restoring single lumen formation in Caco-2 ShPTEN glands. Rosiglitazone effects on cdc42 activation and Caco-2 ShPTEN gland development were attenuated by cotreatment with GW9662, a PPARγ antagonist. Taken together, these studies show PTEN–cdc42 regulation of lumen formation in a 3D model of human CRC glandular morphogenesis. Treatment by the PPARγ ligand rosiglitazone, but not PI3K modulators, rescued colorectal glandular dysmorphogenesis of PTEN deficiency.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Abbreviations
- aPKC:
-
atypical protein kinase C
- CA:
-
constitutively active
- cdc42:
-
Cell division cycle 42
- DN:
-
dominant negative
- EGF:
-
epidermal growth factor
- EV:
-
empty vector
- GEFs:
-
Guanine nucleotide exchange factors
- IGF:
-
insulin-like growth factor
- PI3K:
-
Phosphatidylinositol 3-kinase
- PtdIns:
-
Phosphatidylinositol
- PTEN:
-
phosphatase and tensin homolog deleted on chromosome ten
- Ser:
-
serine
- ShRNA:
-
Short hairpin RNA
- siRNA:
-
small interfering RNA
- Thr:
-
threonine
- wt:
-
wild type.
References
Martin-Belmonte F, Gassama A, Datta A, Yu W, Rescher U, Gerke V et al. PTEN-mediated apical segregation of phosphoinositides controls epithelial morphogenesis through Cdc42. Cell 2007; 128: 383–397.
Michaelson D, Silletti J, Murphy G, D'Eustachio P, Rush M, Philips MR . Differential localization of Rho GTPases in live cells: regulation by hypervariable regions and RhoGDI binding. J Cell Biol 2001; 152: 111–126.
Pertz O . Spatio-temporal Rho GTPase signaling—where are we now? J Cell Sci 2010; 123 (Part 11): 1841–1850.
Qin Y, Meisen WH, Hao Y, Macara IG . Tuba, a Cdc42 GEF, is required for polarized spindle orientation during epithelial cyst formation. J Cell Biol 2010; 189: 661–669.
Rodriguez-Fraticelli AE, Vergarajauregui S, Eastburn DJ, Datta A, Alonso MA, Mostov K et al. The Cdc42 GEF Intersectin 2 controls mitotic spindle orientation to form the lumen during epithelial morphogenesis. J Cell Biol 2010; 189: 725–738.
Noda Y, Takeya R, Ohno S, Naito S, Ito T, Sumimoto H . Human homologues of the Caenorhabditis elegans cell polarity protein PAR6 as an adaptor that links the small GTPases Rac and Cdc42 to atypical protein kinase C. Genes Cells 2001; 6: 107–119.
Joberty G, Petersen C, Gao L, Macara IG . The cell-polarity protein Par6 links Par3 and atypical protein kinase C to Cdc42. Nat Cell Biol 2000; 2: 531–539.
Etienne-Manneville S, Hall A . Cell polarity: Par6, aPKC and cytoskeletal crosstalk. Curr Opin Cell Biol 2003; 15: 67–72.
Etienne-Manneville S . Cdc42--the centre of polarity. J Cell Sci 2004; 117 (Part 8): 1291–1300.
Etienne-Manneville S, Hall A . Cdc42 regulates GSK-3beta and adenomatous polyposis coli to control cell polarity. Nature 2003; 421: 753–756.
Tan I, Yong J, Dong JM, Lim L, Leung T . A tripartite complex containing MRCK modulates lamellar actomyosin retrograde flow. Cell 2008; 135: 123–136.
Mitsushima M, Toyoshima F, Nishida E . Dual role of Cdc42 in spindle orientation control of adherent cells. Mol Cell Biol 2009; 29: 2816–2827.
Jaffe AB, Kaji N, Durgan J, Hall A . Cdc42 controls spindle orientation to position the apical surface during epithelial morphogenesis. J Cell Biol 2008; 183: 625–633.
Durgan J, Kaji N, Jin D, Hall A . Par6B and atypical PKC (aPKC) regulate mitotic spindle orientation during epithelial morphogenesis. J Biol Chem 2011; 286: 12461–12467.
Shao J, Washington MK, Saxena R, Sheng H . Heterozygous disruption of the PTEN promotes intestinal neoplasia in APCmin/+ mouse: roles of osteopontin. Carcinogenesis 2007; 28: 2476–2483.
Sawai H, Yasuda A, Ochi N, Ma J, Matsuo Y, Wakasugi T et al. Loss of PTEN expression is associated with colorectal cancer liver metastasis and poor patient survival. BMC Gastroenterol 2008; 8: 56.
Nassif NT, Lobo GP, Wu X, Henderson CJ, Morrison CD, Eng C et al. PTEN mutations are common in sporadic microsatellite stable colorectal cancer. Oncogene 2004; 23: 617–628.
Wong KK, Engelman JA, Cantley LC . Targeting the PI3K signaling pathway in cancer. Curr Opin Genet Dev 2010; 20: 87–90.
Grant S . Cotargeting survival signaling pathways in cancer. J Clin Invest 2008; 118: 3003–3006.
Song MS, Carracedo A, Salmena L, Song SJ, Egia A, Malumbres M et al. Nuclear PTEN regulates the APC-CDH1 tumor-suppressive complex in a phosphatase-independent manner. Cell 2011; 144: 187–199.
Wang H, Karikomi M, Naidu S, Rajmohan R, Caserta E, Chen HZ et al. Allele-specific tumor spectrum in pten knockin mice. Proc Natl Acad Sci USA. 2010; 107: 5142–5147.
Gunaratne J, Goh MX, Swa HL, Lee FY, Sanford E, Wong LM et al. Protein interactions of phosphatase and tensin homologue (PTEN) and its cancer-associated G20E mutant compared by using stable isotope labeling by amino acids in cell culture-based parallel affinity purification. J Biol Chem 2011; 286: 18093–18103.
Patel L, Pass I, Coxon P, Downes CP, Smith SA, Macphee CH . Tumor suppressor and anti-inflammatory actions of PPARgamma agonists are mediated via upregulation of PTEN. Curr Biol 2001; 11: 764–768.
Teresi RE, Planchon SM, Waite KA, Eng C . Regulation of the PTEN promoter by statins and SREBP. Hum Mol Genet 2008; 17: 919–928.
Poliseno L, Salmena L, Zhang J, Carver B, Haveman WJ, Pandolfi PP . A coding-independent function of gene and pseudogene mRNAs regulates tumour biology. Nature 2010; 465: 1033–1038.
Seargent JM, Yates EA, Gill JH . GW9662, a potent antagonist of PPARgamma, inhibits growth of breast tumour cells and promotes the anticancer effects of the PPARgamma agonist rosiglitazone, independently of PPARgamma activation. Br J Pharmacol 2004; 143: 933–937.
Lee C, Kim JS, Waldman T . PTEN gene targeting reveals a radiation-induced size checkpoint in human cancer cells. Cancer Res 2004; 64: 6906–6914.
Boehm JS, Hession MT, Bulmer SE, Hahn WC . Transformation of human and murine fibroblasts without viral oncoproteins. Mol Cellular Biol 2005; 25: 6464–6474.
Alessi DR, Andjelkovic M, Caudwell B, Cron P, Morrice N, Cohen P et al. Mechanism of activation of protein kinase B by insulin and IGF-1. EMBO J. 1996; 15: 6541–6551.
Stokoe D, Stephens LR, Copeland T, Gaffney PR, Reese CB, Painter GF et al. Dual role of phosphatidylinositol-3,4,5-trisphosphate in the activation of protein kinase B. Science (New York, NY) 1997; 277: 567–570.
Etienne-Manneville S, Hall A . Integrin-mediated activation of Cdc42 controls cell polarity in migrating astrocytes through PKCzeta. Cell 2001; 106: 489–498.
Sun H, Lesche R, Li DM, Liliental J, Zhang H, Gao J et al. PTEN modulates cell cycle progression and cell survival by regulating phosphatidylinositol 3,4,5,-trisphosphate and Akt/protein kinase B signaling pathway. Proc Natl Acad Sci USA 1999; 96: 6199–6204.
Aoki K, Nakamura T, Fujikawa K, Matsuda M . Local phosphatidylinositol 3,4,5-trisphosphate accumulation recruits Vav2 and Vav3 to activate Rac1/Cdc42 and initiate neurite outgrowth in nerve growth factor-stimulated PC12 cells. Mol Biol Cell 2005; 16: 2207–2217.
Cho YB, Chun HK, Yun HR, Kim HC, Yun SH, Lee WY . Histological grade predicts survival time associated with recurrence after resection for colorectal cancer. Hepato-Gastroenterol 2009; 56: 1335–1340.
Liliental J, Moon SY, Lesche R, Mamillapalli R, Li D, Zheng Y et al. Genetic deletion of the Pten tumor suppressor gene promotes cell motility by activation of Rac1 and Cdc42 GTPases. Curr Biol 2000; 10: 401–404.
Cross DA, Alessi DR, Cohen P, Andjelkovich M, Hemmings BA . Inhibition of glycogen synthase kinase-3 by insulin mediated by protein kinase B. Nature 1995; 378: 785–789.
Al-Khouri AM, Ma Y, Togo SH, Williams S, Mustelin T . Cooperative phosphorylation of the tumor suppressor phosphatase and tensin homologue (PTEN) by casein kinases and glycogen synthase kinase 3beta. J Biol Chem 2005; 280: 35195–35202.
ter Haar E, Coll JT, Austen DA, Hsiao HM, Swenson L, Jain J . Structure of GSK3beta reveals a primed phosphorylation mechanism. Nat Struct Biol 2001; 8: 593–596.
Maccario H, Perera NM, Davidson L, Downes CP, Leslie NR . PTEN is destabilized by phosphorylation on Thr366. Biochem J 2007; 405: 439–444.
Deevi R, Fatehullah A, Jagan I, Nagaraju M, Bingham V, Campbell FC . PTEN regulates colorectal epithelial apoptosis through Cdc42 signalling. Br J Cancer 2011; 105: 1313–1321.
Orena SJ, Torchia AJ, Garofalo RS . Inhibition of glycogen-synthase kinase 3 stimulates glycogen synthase and glucose transport by distinct mechanisms in 3T3-L1 adipocytes. J Biol Chem 2000; 275: 15765–15772.
Summers SA, Kao AW, Kohn AD, Backus GS, Roth RA, Pessin JE et al. The role of glycogen synthase kinase 3beta in insulin-stimulated glucose metabolism. J Biol Chem 1999; 274: 17934–17940.
De Sarno P, Li X, Jope RS . Regulation of Akt and glycogen synthase kinase-3 beta phosphorylation by sodium valproate and lithium. Neuropharmacology 2002; 43: 1158–1164.
Kong M, Mounier C, Wu J, Posner BI . Epidermal growth factor-induced phosphatidylinositol 3-kinase activation and DNA synthesis. Identification of Grb2-associated binder 2 as the major mediator in rat hepatocytes. J Biol Chem 2000; 275: 36035–36042.
Sinha S, Yang W . Cellular signaling for activation of Rho GTPase Cdc42. Cell Signal 2008; 20: 1927–1934.
Fuentes EJ, Karnoub AE, Booden MA, Der CJ, Campbell SL . Critical role of the pleckstrin homology domain in Dbs signaling and growth regulation. J Biol Chem 2003; 278: 21188–21196.
Schmidt A, Hall A . Guanine nucleotide exchange factors for Rho GTPases: turning on the switch. Genes Dev 2002; 16: 1587–1609.
Han J, Luby-Phelps K, Das B, Shu X, Xia Y, Mosteller RD et al. Role of substrates and products of PI 3-kinase in regulating activation of Rac-related guanosine triphosphatases by Vav. Science 1998; 279: 558–560.
Whitehead IP, Campbell S, Rossman KL, Der CJ . Dbl family proteins. Biochim Biophys Acta. 1997; 1332: F1–23.
Hussain NK, Jenna S, Glogauer M, Quinn CC, Wasiak S, Guipponi M et al. Endocytic protein intersectin-l regulates actin assembly via Cdc42 and N-WASP. Nat Cell Biol 2001; 3: 927–932.
Pruitt WM, Karnoub AE, Rakauskas AC, Guipponi M, Antonarakis SE, Kurakin A et al. Role of the pleckstrin homology domain in intersectin-L Dbl homology domain activation of Cdc42 and signaling. Biochim Biophys Acta 2003; 1640: 61–68.
Salazar MA, Kwiatkowski AV, Pellegrini L, Cestra G, Butler MH, Rossman KL et al. Tuba, a novel protein containing bin/amphiphysin/Rvs and Dbl homology domains, links dynamin to regulation of the actin cytoskeleton. J Biol Chem 2003; 278: 49031–49043.
Itoh T, De Camilli P . BAR, F-BAR (EFC) and ENTH/ANTH domains in the regulation of membrane-cytosol interfaces and membrane curvature. Biochim Biophys Acta 2006; 1761: 897–912.
Lemmon MA . Membrane recognition by phospholipid-binding domains. Nat Rev Mol Cell Biol 2008; 9: 99–111.
Guruswamy S, Swamy MV, Choi CI, Steele VE, Rao CV . S-adenosyl L-methionine inhibits azoxymethane-induced colonic aberrant crypt foci in F344 rats and suppresses human colon cancer Caco-2 cell growth in 3D culture. Int J Cancer 2008; 122: 25–30.
Johnson K, Wodarz A . A genetic hierarchy controlling cell polarity. Nat Cell Biol 2003; 5: 12–14.
Goberdhan DC, Paricio N, Goodman EC, Mlodzik M, Wilson C . Drosophila tumor suppressor PTEN controls cell size and number by antagonizing the Chico/PI3-kinase signaling pathway. Genes Dev 1999; 13: 3244–3258.
Backman S, Stambolic V, Mak T . PTEN function in mammalian cell size regulation. Curr Opin Neurobiol 2002; 12: 516–522.
Donev IS, Wang W, Yamada T, Li Q, Takeuchi S, Matsumoto K et al. Transient PI3K inhibition induces apoptosis and overcomes HGF-mediated resistance to EGFR-TKIs in EGFR mutant lung cancer. Clin Cancer Res 2011; 17: 2260–2269.
Balciunaite E, Kazlauskas A . Early phosphoinositide 3-kinase activity is required for late activation of protein kinase Cepsilon in platelet-derived-growth-factor-stimulated cells: evidence for signalling across a large temporal gap. Biochem J. 2001; 358 (Pt 2): 281–285.
Raynaud FI, Eccles SA, Patel S, Alix S, Box G, Chuckowree I et al. Biological properties of potent inhibitors of class I phosphatidylinositide 3-kinases: from PI-103 through PI-540, PI-620 to the oral agent GDC-0941. Mol Cancer Ther 2009; 8: 1725–1738.
Carracedo A, Alimonti A, Pandolfi PP . PTEN level in tumor suppression: how much is too little? Cancer Res 2011; 71: 629–633.
Trotman LC, Niki M, Dotan ZA, Koutcher JA, Di Cristofano A, Xiao A et al. Pten dose dictates cancer progression in the prostate. PLoS Biol 2003; 1: E59.
Pelz C, Matsumoto T, Molski TF, Becker EL, Sha'afi RI . Characterization of the membrane-associated GTPase activity: effects of chemotactic factors and toxins. J Cellular Biochem 1989; 39: 197–206.
Debnath J, Muthuswamy SK, Brugge JS . Morphogenesis and oncogenesis of MCF-10A mammary epithelial acini grown in three-dimensional basement membrane cultures. Methods (San Diego, California) 2003; 30: 256–268.
Acknowledgements
We gratefully acknowledge the support of the Wellcome Trust (WT081232MA) and the Department for Education and Learning, Northern Ireland for financial support. We are greatly indebted to Dr T Waldman, Georgetown University, USA, for supply of PTEN+/+ HCT116 and PTEN −/−HCT116 cells, to Dr Pietro DiCamilli, Howard Hughes Medical Institute, Yale, for his generous provision of anti-human Tuba antibody, to Dr Estelle McLean for technical help, to Dr James Murray for the gift of PI-103 and to S Church for assistance with all imaging.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Jagan, I., Fatehullah, A., Deevi, R. et al. Rescue of glandular dysmorphogenesis in PTEN-deficient colorectal cancer epithelium by PPARγ-targeted therapy. Oncogene 32, 1305–1315 (2013). https://doi.org/10.1038/onc.2012.140
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2012.140
Keywords
This article is cited by
-
Integrative multi-omics analysis of a colon cancer cell line with heterogeneous Wnt activity revealed RUNX2 as an epigenetic regulator of EMT
Oncogene (2020)
-
The cis-acting replication element of the Hepatitis C virus genome recruits host factors that influence viral replication and translation
Scientific Reports (2016)
-
p53 regulates cytoskeleton remodeling to suppress tumor progression
Cellular and Molecular Life Sciences (2015)