Abstract
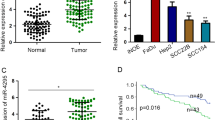
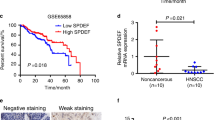
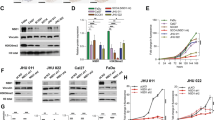
Head and neck squamous cell carcinoma (HNSCC) is the sixth most prevalent cancer worldwide with about 600 000 new cases diagnosed each year. Understanding the molecular pathways that lead to HNSCC is crucial to identify new targets for anti-cancer drug development. Protein kinase Cɛ (PKCɛ) is elevated in HNSCC and regulates the activation of Akt, Stat3 and Rho GTPases. To date, the molecular mechanism of PKCɛ dysregulation in HNSCC remains to be elucidated. In silico analysis identified three putative microRNA-107 (miR-107) binding sites in the 3′-untranslated region (UTR) of PKCɛ. An inverse relationship was revealed between miR-107 and PKCɛ in HNSCC cell lines. Delivery of miR-107 reduced PKCɛ levels in SCC15, SCC25 and CAL27, three HNSCC cell lines with high PKCɛ and low miR-107. The activity of a luciferase reporter construct containing the 3′-UTR of PKCɛ was downregulated by miR-107 and mutations in the three cognate miR-107 binding sites completely ablated the regulation by miR-107. Treatment with miR-107 significantly blocked cell proliferation, DNA replication, colony formation and invasion in SCC25 and CAL27 cells. Ectopic expression of miR-resistant PKCɛ was sufficient to partially rescue the loss-of-function phenotype in miR-107-overexpressing SCC25 cells. Tumor growth in nude mice was retarded by 93±7% in CAL27/miR-107 cells compared with CAL27/miR-control cells. Last, human primary HNSCC tumors with elevated PKCɛ had reduced miR-107 expression. Our results demonstrate that PKCɛ is directly regulated by miR-107 and, moreover, suggest that miR-107 may be a potential anti-cancer therapeutic for HNSCC.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Avissar M, Christensen BC, Kelsey KT, Marsit CJ . (2009). MicroRNA expression ratio is predictive of head and neck squamous cell carcinoma. Clin Cancer Res 15: 2850–2855.
Ayala de la Pena F, Kanasaki K, Kanasaki M, Tangirala N, Maeda G, Kalluri R . (2011). Loss of p53 and acquisition of angiogenic microRNA profile is insufficient to facilitate progression of bladder urothelial carcinoma in situ to invasive carcinoma. J Biol Chem 286: 20778–20787.
Aziz MH, Hafeez BB, Sand JM, Pierce DB, Aziz SW, Dreckschmidt NE et al. (2010). Protein kinase C varepsilon mediates Stat3Ser727 phosphorylation, Stat3-regulated gene expression, and cell invasion in various human cancer cell lines through integration with MAPK cascade (RAF-1, MEK1/2, and ERK1/2). Oncogene 29: 3100–3109.
Bonner JA, Harari PM, Giralt J, Cohen RB, Jones CU, Sur RK et al. (2010). Radiotherapy plus cetuximab for locoregionally advanced head and neck cancer: 5-year survival data from a phase 3 randomised trial, and relation between cetuximab-induced rash and survival. Lancet Oncol 11: 21–28.
Calin GA, Croce CM . (2006). MicroRNA signatures in human cancers. Nat Rev Cancer 6: 857–866.
Cohen EE, Lingen MW, Zhu B, Zhu H, Straza MW, Pierce C et al. (2006). Protein kinase C zeta mediates epidermal growth factor-induced growth of head and neck tumor cells by regulating mitogen-activated protein kinase. Cancer Res 66: 6296–6303.
Cohen EE, Zhu H, Lingen MW, Martin LE, Kuo WL, Choi EA et al. (2009). A feed-forward loop involving protein kinase Calpha and microRNAs regulates tumor cell cycle. Cancer Res 69: 65–74.
Croce CM . (2009). Causes and consequences of microRNA dysregulation in cancer. Nat Rev Genet 10: 704–714.
Datta J, Kutay H, Nasser MW, Nuovo GJ, Wang B, Majumder S et al. (2008). Methylation mediated silencing of MicroRNA-1 gene and its role in hepatocellular carcinogenesis. Cancer Res 68: 5049–5058.
Dempsey EC, Newton AC, Mochly-Rosen D, Fields AP, Reyland ME, Insel PA et al. (2000). Protein kinase C isozymes and the regulation of diverse cell responses. Am J Physiol Lung Cell Mol Physiol 279: L429–L438.
Feng L, Xie Y, Zhang H, Wu Y . (2011). miR-107 targets cyclin-dependent kinase 6 expression, induces cell cycle G1 arrest and inhibits invasion in gastric cancer cells. Med Oncol (doi:10.1007/s12032-011-9823-1).
Filipowicz W, Bhattacharyya SN, Sonenberg N . (2008). Mechanisms of post-transcriptional regulation by microRNAs: are the answers in sight? Nat Rev Genet 9: 102–114.
Gutcher I, Webb PR, Anderson NG . (2003). The isoform-specific regulation of apoptosis by protein kinase C. Cell Mol Life Sci 60: 1061–1070.
Hwang HW, Mendell JT . (2006). MicroRNAs in cell proliferation, cell death, and tumorigenesis. Br J Cancer 94: 776–780.
Jaken S, Parker PJ . (2000). Protein kinase C binding partners. Bioessays 22: 245–254.
Kamangar F, Dores GM, Anderson WF . (2006). Patterns of cancer incidence, mortality, and prevalence across five continents: defining priorities to reduce cancer disparities in different geographic regions of the world. J Clin Oncol 24: 2137–2150.
Kozaki K, Imoto I, Mogi S, Omura K, Inazawa J . (2008). Exploration of tumor-suppressive microRNAs silenced by DNA hypermethylation in oral cancer. Cancer Res 68: 2094–2105.
Laramore GE, Scott CB, al-Sarraf M, Haselow RE, Ervin TJ, Wheeler R et al. (1992). Adjuvant chemotherapy for resectable squamous cell carcinomas of the head and neck: report on Intergroup Study 0034. Int J Radiat Oncol Biol Phys 23: 705–713.
Lee KH, Lotterman C, Karikari C, Omura N, Feldmann G, Habbe N et al. (2009). Epigenetic silencing of microRNA miR-107 regulates cyclin-dependent kinase 6 expression in pancreatic cancer. Pancreatology 9: 293–301.
Leemans CR, Braakhuis BJ, Brakenhoff RH . (2011). The molecular biology of head and neck cancer. Nat Rev Cancer 11: 9–22.
Li X, Zhang Y, Shi Y, Dong G, Liang J, Han Y et al. (2010). MicroRNA-107, an oncogene microRNA that regulates tumor invasion and metastasis by targeting DICER1 in gastric cancer: MiR-107 promotes gastric cancer invasion and metastasis. J Cell Mol Med 15: 1887–1895.
Liu X, Jiang L, Wang A, Yu J, Shi F, Zhou X . (2009). MicroRNA-138 suppresses invasion and promotes apoptosis in head and neck squamous cell carcinoma cell lines. Cancer Lett 286: 217–222.
Martello G, Rosato A, Ferrari F, Manfrin A, Cordenonsi M, Dupont S et al. (2010). A microRNA targeting dicer for metastasis control. Cell 141: 1195–1207.
Martinez-Gimeno C, Diaz-Meco MT, Dominguez I, Moscat J . (1995). Alterations in levels of different protein kinase C isotypes and their influence on behavior of squamous cell carcinoma of the oral cavity: epsilon PKC, a novel prognostic factor for relapse and survival. Head Neck 17: 516–525.
Nasser MW, Datta J, Nuovo G, Kutay H, Motiwala T, Majumder S et al. (2008). Down-regulation of micro-RNA-1 (miR-1) in lung cancer. Suppression of tumorigenic property of lung cancer cells and their sensitization to doxorubicin-induced apoptosis by miR-1. J Biol Chem 283: 33394–33405.
Pan Q, Bao LW, Teknos TN, Merajver SD . (2006). Targeted disruption of protein kinase C epsilon reduces cell invasion and motility through inactivation of RhoA and RhoC GTPases in head and neck squamous cell carcinoma. Cancer Res 66: 9379–9384.
Parkin DM . (2001). Global cancer statistics in the year 2000. Lancet Oncol 2: 533–543.
Wong TS, Liu XB, Wong BY, Ng RW, Yuen AP, Wei WI . (2008). Mature miR-184 as potential oncogenic microRNA of squamous cell carcinoma of tongue. Clin Cancer Res 14: 2588–2592.
Yamakuchi M, Lotterman CD, Bao C, Hruban RH, Karim B, Mendell JT et al. (2010). P53-induced microRNA-107 inhibits HIF-1 and tumor angiogenesis. Proc Natl Acad Sci USA 107: 6334–6339.
Acknowledgements
This work was supported in part by the National Cancer Institute at the National Institutes of Health (R01CA135096); American Cancer Society (RSG0821901); the Michelle Theado Memorial Grant and the John Young Memorial Grant from the Joan Bisesi Fund for Head and Neck Oncology Research and the Arthur G. James Cancer Hospital and Richard J. Solove Research Institute, The Ohio State University Comprehensive Cancer Center.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Datta, J., Smith, A., Lang, J. et al. microRNA-107 functions as a candidate tumor-suppressor gene in head and neck squamous cell carcinoma by downregulation of protein kinase Cɛ. Oncogene 31, 4045–4053 (2012). https://doi.org/10.1038/onc.2011.565
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.565
Keywords
This article is cited by
-
Depleted tumor suppressor miR-107 in plasma relates to tumor progression and is a novel therapeutic target in pancreatic cancer
Scientific Reports (2017)
-
Long noncoding RNA NEAT1 promotes laryngeal squamous cell cancer through regulating miR-107/CDK6 pathway
Journal of Experimental & Clinical Cancer Research (2016)
-
Subgroup J avian leukosis virus infection of chicken dendritic cells induces apoptosis via the aberrant expression of microRNAs
Scientific Reports (2016)
-
Decreased expression of microRNA-107 predicts poorer prognosis in glioma
Tumor Biology (2015)
-
Identification and characterization of Dicer1e, a Dicer1 protein variant, in oral cancer cells
Molecular Cancer (2014)