Abstract
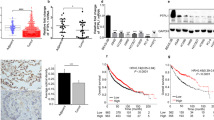
We integrated four gene expression profile data sets, namely two different pair-matched stage I lung adenocarcinoma data sets, secondary metastatic tumors vs benign tumors and lung tumor metastasizes to the brain, and we identified one kinase, T-LAK Cell-Originated Protein Kinase (TOPK), as a putative gene that promotes metastasis. To delineate the role of TOPK in lung cancer, we showed that overexpression of TOPK, but not a catalytically inactive form of TOPK, can enhance the migration and invasion of lung fibroblasts or cells with low TOPK expression. In addition, TOPK-induced cell migration was shown to be a PI3K/AKT-dependent event. TOPK concurrently promoted AKT phosphorylation at Ser473 and decreased the phosphatase and tensin homolog (PTEN) levels, whereas TOPK knockdown had the reverse effects. LY294002, a PI3K inhibitor, did not inhibit the TOPK-induced decrease in PTEN, and co-expression of PTEN significantly reduced TOPK-induced AKT phosphorylation in a dose-dependent manner; these results indicate that the TOPK-mediated PTEN decrease has an upstream role in regulating PI3K/AKT-stimulated migration. Using immunohistochemical analysis of lung cancer tissue samples, we showed that a high TOPK expression level correlates strongly with reduced overall and disease-free survivals. Moreover, an inverse correlation between TOPK and PTEN expression was present and is consistent with the biochemical findings. Finally, a combination of high TOPK and low PTEN expression was inversely correlated with overall and disease-free survivals, independent of other pathologic staging factors. Our results suggest that TOPK is a potential therapeutic target in lung cancer that promotes cell migration by modulating a PI3K/PTEN/AKT-dependent signaling pathway; they also suggest that high TOPK expression, either alone or in combination with a low level of PTEN, may serve as a prognostic marker for lung cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Abe Y, Matsumoto S, Kito K, Ueda N . (2000). Cloning and expression of a novel MAPKK-like protein kinase, lymphokine-activated killer T-cell-originated protein kinase, specifically expressed in the testis and activated lymphoid cells. J Biol Chem 275: 21525–21531.
Abe Y, Takeuchi T, Kagawa-Miki L, Ueda N, Shigemoto K, Yasukawa M et al. (2007). A mitotic kinase TOPK enhances Cdk1/cyclin B1-dependent phosphorylation of PRC1 and promotes cytokinesis. J Mol Biol 370: 231–245.
Akca H, Demiray A, Tokgun O, Yokota J . (2011). Invasiveness and anchorage independent growth ability augmented by PTEN inactivation through the PI3K/AKT/NFkB pathway in lung cancer cells. Lung Cancer 73: 302–309.
al-Kattan K, Sepsas E, Fountain SW, Townsend ER . (1997). Disease recurrence after resection for stage I lung cancer. Eur J Cardiothorac Surg 12: 380–384.
Arriagada R, Auperin A, Burdett S, Higgins JP, Johnson DH, Le Chevalier T et al. (2010). Adjuvant chemotherapy, with or without postoperative radiotherapy, in operable non-small-cell lung cancer: two meta-analyses of individual patient data. Lancet 375: 1267–1277.
Arriagada R, Bergman B, Dunant A, Le Chevalier T, Pignon JP, Vansteenkiste J . (2004). Cisplatin-based adjuvant chemotherapy in patients with completely resected non-small-cell lung cancer. N Engl J Med 350: 351–360.
Ayllon V, O'Connor R . (2007). PBK/TOPK promotes tumour cell proliferation through p38 MAPK activity and regulation of the DNA damage response. Oncogene 26: 3451–3461.
Chalhoub N, Baker SJ . (2009). PTEN and the PI3-kinase pathway in cancer. Annu Rev Pathol 4: 127–150.
Chen CH, Lai JM, Chou TY, Chen CY, Su LJ, Lee YC et al. (2009a). VEGFA upregulates FLJ10540 and modulates migration and invasion of lung cancer via PI3K/AKT pathway. PLoS ONE 4: e5052.
Chen TC, Lee SA, Hong TM, Shih JY, Lai JM, Chiou HY et al. (2009b). From midbody protein-protein interaction network construction to novel regulators in cytokinesis. J Proteome Res 8: 4943–4953.
Friedl P, Wolf K . (2003). Tumour-cell invasion and migration: diversity and escape mechanisms. Nat Rev Cancer 3: 362–374.
Furnari FB, Lin H, Huang HS, Cavenee WK . (1997). Growth suppression of glioma cells by PTEN requires a functional phosphatase catalytic domain. Proc Natl Acad Sci USA 94: 12479–12484.
Gaudet S, Branton D, Lue RA . (2000). Characterization of PDZ-binding kinase, a mitotic kinase. Proc Natl Acad Sci USA 97: 5167–5172.
Herrero-Martin D, Osuna D, Ordonez JL, Sevillano V, Martins AS, Mackintosh C et al. (2009). Stable interference of EWS-FLI1 in an Ewing sarcoma cell line impairs IGF-1/IGF-1R signalling and reveals TOPK as a new target. Br J Cancer 101: 80–90.
Hu F, Gartenhaus RB, Eichberg D, Liu Z, Fang HB, Rapoport AP . (2010). PBK/TOPK interacts with the DBD domain of tumor suppressor p53 and modulates expression of transcriptional targets including p21. Oncogene 29: 5464–5474.
Hung JJ, Hsu WH, Hsieh CC, Huang BS, Huang MH, Liu JS et al. (2009). Post-recurrence survival in completely resected stage I non-small cell lung cancer with local recurrence. Thorax 64: 192–196.
Kratz JR, Jablons DM . (2009). Genomic prognostic models in early-stage lung cancer. Clin Lung Cancer 10: 151–157.
Matsumoto S, Abe Y, Fujibuchi T, Takeuchi T, Kito K, Ueda N et al. (2004). Characterization of a MAPKK-like protein kinase TOPK. Biochem Biophys Res Commun 325: 997–1004.
Nandi A, Tidwell M, Karp J, Rapoport AP . (2004). Protein expression of PDZ-binding kinase is up-regulated in hematologic malignancies and strongly down-regulated during terminal differentiation of HL-60 leukemic cells. Blood Cells Mol Dis 32: 240–245.
Nandi AK, Ford T, Fleksher D, Neuman B, Rapoport AP . (2007). Attenuation of DNA damage checkpoint by PBK, a novel mitotic kinase, involves protein-protein interaction with tumor suppressor p53. Biochem Biophys Res Commun 358: 181–188.
Oh SM, Zhu F, Cho YY, Lee KW, Kang BS, Kim HG et al. (2007). T-lymphokine-activated killer cell-originated protein kinase functions as a positive regulator of c-Jun-NH2-kinase 1 signaling and H-Ras-induced cell transformation. Cancer Res 67: 5186–5194.
Omuro AM, Kris MG, Miller VA, Franceschi E, Shah N, Milton DT et al. (2005). High incidence of disease recurrence in the brain and leptomeninges in patients with nonsmall cell lung carcinoma after response to gefitinib. Cancer 103: 2344–2348.
Park JH, Lin ML, Nishidate T, Nakamura Y, Katagiri T . (2006). PDZ-binding kinase/T-LAK cell-originated protein kinase, a putative cancer/testis antigen with an oncogenic activity in breast cancer. Cancer Res 66: 9186–9195.
Simons-Evelyn M, Bailey-Dell K, Toretsky JA, Ross DD, Fenton R, Kalvakolanu D et al. (2001). PBK/TOPK is a novel mitotic kinase which is upregulated in Burkitt's lymphoma and other highly proliferative malignant cells. Blood Cells Mol Dis 27: 825–829.
Soria JC, Lee HY, Lee JI, Wang L, Issa JP, Kemp BL et al. (2002). Lack of PTEN expression in non-small cell lung cancer could be related to promoter methylation. Clin Cancer Res 8: 1178–1184.
Strauss GM, Herndon 2nd JE, Maddaus MA, Johnstone DW, Johnson EA, Harpole DH et al. (2008). Adjuvant paclitaxel plus carboplatin compared with observation in stage IB non-small-cell lung cancer: CALGB 9633 with the Cancer and Leukemia Group B, Radiation Therapy Oncology Group, and North Central Cancer Treatment Group Study Groups. J Clin Oncol 26: 5043–5051.
Su LJ, Chang CW, Wu YC, Chen KC, Lin CJ, Liang SC et al. (2007). Selection of DDX5 as a novel internal control for Q-RT-PCR from microarray data using a block bootstrap re-sampling scheme. BMC Genomics 8: 140.
Sugimura H, Nichols FC, Yang P, Allen MS, Cassivi SD, Deschamps C et al. (2007). Survival after recurrent nonsmall-cell lung cancer after complete pulmonary resection. Ann Thorac Surg 83: 409–417; discussion 417–408.
Vivanco I, Sawyers CL . (2002). The phosphatidylinositol 3-kinase AKT pathway in human cancer. Nat Rev Cancer 2: 489–501.
Wang X, Trotman LC, Koppie T, Alimonti A, Chen Z, Gao Z et al. (2007). NEDD4-1 is a proto-oncogenic ubiquitin ligase for PTEN. Cell 128: 129–139.
Zhu F, Zykova TA, Kang BS, Wang Z, Ebeling MC, Abe Y et al. (2007). Bidirectional signals transduced by TOPK-ERK interaction increase tumorigenesis of HCT116 colorectal cancer cells. Gastroenterology 133: 219–231.
Zlobec I, Molinari F, Kovac M, Bihl MP, Altermatt HJ, Diebold J et al. (2010). Prognostic and predictive value of TOPK stratified by KRAS and BRAF gene alterations in sporadic, hereditary and metastatic colorectal cancer patients. Br J Cancer 102: 151–161.
Zykova TA, Zhu F, Lu C, Higgins L, Tatsumi Y, Abe Y et al. (2006). Lymphokine-activated killer T-cell-originated protein kinase phosphorylation of histone H2AX prevents arsenite-induced apoptosis in RPMI7951 melanoma cells. Clin Cancer Res 12: 6884–6893.
Acknowledgements
We thank Dr Chi-Ying F Huang (Yang-Ming University, Taipei, Taiwan) for providing lung cancer microarray data. This work was supported by grants from National Science Council (Taiwan) (NSC95-2311-B-030-002-MY3 and NSC99-2627-B-030-001) to JM Lai and Center of Excellence for Cancer Research at Taipei Veterans General Hospital (DOH100-TD-C-111-007) to YC Wu.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Shih, MC., Chen, JY., Wu, YC. et al. TOPK/PBK promotes cell migration via modulation of the PI3K/PTEN/AKT pathway and is associated with poor prognosis in lung cancer. Oncogene 31, 2389–2400 (2012). https://doi.org/10.1038/onc.2011.419
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.419
Keywords
This article is cited by
-
TOPK/PBK is phosphorylated by ERK2 at serine 32, promotes tumorigenesis and is involved in sorafenib resistance in RCC
Cell Death & Disease (2022)
-
TOPK Activation Exerts Protective Effects on Cisplatin-induced Acute Kidney Injury
Current Medical Science (2022)
-
The role of T-LAK cell-originated protein kinase in targeted cancer therapy
Molecular and Cellular Biochemistry (2022)
-
TOPKi-NBD: a fluorescent small molecule for tumor imaging
European Journal of Nuclear Medicine and Molecular Imaging (2020)
-
TOPK promotes metastasis of esophageal squamous cell carcinoma by activating the Src/GSK3β/STAT3 signaling pathway via γ-catenin
BMC Cancer (2019)