Abstract
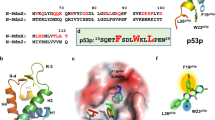
The p53 tumor suppressor gene encodes a transcription factor that is commonly mutated in cancer. Tumors arise when premalignant cells are unable to undergo p53-dependent apoptosis, cell cycle arrest or DNA repair. The p53-signaling pathway affects not only tumor development, but also the response of tumors to chemotherapeutic drugs. In this study, we use cell penetrating peptide conjugates of phosphorodiamidate morpholino oligomers (PPMOs) to inhibit p53 expression. We examine the functional properties of endogenous p53 isoforms that are produced upon PPMO-mediated inhibition of p53 translation and splicing, and report that loss of N-terminal or C-terminal sequences interferes with the transcriptional activity of p53. Importantly, we report that PPMO-mediated inhibition of p53 expression sensitizes human cancer cells with wild-type p53 to chemotherapeutic drugs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Abes R, Arzumanov A, Moulton H, Abes S, Ivanova G, Gait MJ et al. (2008). Arginine-rich cell penetrating peptides: design, structure-activity, and applications to alter pre-mRNA splicing by steric-block oligonucleotides. J Pept Sci 14: 455–460.
Adorno M, Cordenonsi M, Montagner M, Dupont S, Wong C, Hann B et al. (2009). A mutant-p53/Smad complex opposes p63 to empower TGFβ-induced metastasis. Cell 137: 87–98.
Banks L, Matlashewski G, Crawford L . (1986). Isolation of human-p53-specific monoclonal antibodies and their use in the studies of human p53 expression. Eur J Biochem 159: 529–534.
Bertheau P, Espie M, Turpin E, Lehmann J, Plassa LF, Varna M et al. (2008). TP53 status and response to chemotherapy in breast cancer. Pathobiology 75: 132–139.
Bourdon JC, Fernandes K, Murray-Zmijewski F, Liu G, Diot A, Xirodimas DP et al. (2005). P53 isoforms can regulate P53 transcriptional activity. Genes Dev 19: 2122–2137.
Bunz F, Hwang PM, Torrance C, Waldman T, Zhang Y, Dillehay L et al. (1999). Disruption of p53 in human cancer cells alters the responses to therapeutic agents. J Clin Invest 104: 263–269.
Courtois S, Verhaegh G, North S, Luciani MG, Lassus P, Hibner U et al. (2002). ΔN-p53, a natural isoform of p53 lacking the first transactivation domain, counteracts growth suppression by wild-type p53. Oncogene 21: 6722–6728.
El-Deiry WS . (2003). The role of p53 in chemosensitivity and radiosensitivity. Oncogene 22: 7486–7495.
Fei P, Bernhard EJ, El-Deiry WS . (2002). Tissue-specific induction of p53 targets in vivo. Cancer Res 62: 7316–7327.
Flaman JM, Waridel F, Estreicher A, Vannier A, Limacher JM, Gilbert D et al. (1996). The human tumour suppressor gene p53 is alternatively spliced in normal cells. Oncogene 12: 813–818.
Fujita K, Mondal AM, Horikawa I, Nguyen GH, Kumamoto K, Sohn JJ et al. (2009). p53 isoforms Δ133p53 and p53β are endogenous regulators of replicative cellular senescence. Nat Cell Biol 11: 1135–1142.
Ghosh A, Stewart D, Matlashewski G . (2004). Regulation of human p53 activity and cell localization by alternative splicing. Mol Cell Biol 24: 7987–7997.
Gudkov AV, Komarova EA . (2003). The role of p53 in determining sensitivity to radiotherapy. Nat Rev Cancer 3: 117–129.
Harlow E, Crawford LV, Pim DC, Williamson NM . (1981). Monoclonal antibodies specific for simian virus 40 tumor antigens. J Virol 39: 861–869.
Harms KL, Chen X . (2006). The functional domains in p53 family proteins exhibit both common and distinct properties. Cell Death Differ 13: 890–897.
Horikawa I, Suzuki M, Oshimura M . (1995). An amino-terminally truncated p53 protein expressed in a human choriocarcinoma cell line, CC1. Hum Mol Genet 4: 313–314.
Iversen PL . (2001). Phosphorodiamidate morpholino oligomers: favorable properties for sequence-specific gene inactivation. Curr Opin Mol Ther 3: 235–238.
Kim E, Giese A, Deppert W . (2009). Wild-type p53 in cancer cells: when a guardian turns into a blackguard. Biochem Pharmacol 77: 11–20.
Koonin EV, Rogozin IB, Glazko GV . (2005). p53 gain-of-function: tumor biology and bioinformatics come together. Cell Cycle 4: 686–688.
Lang GA, Iwakuma T, Suh YA, Liu G, Rao VA, Parant JM et al. (2004). Gain of function of a p53 hot spot mutation in a mouse model of Li-Fraumeni syndrome. Cell 119: 861–872.
Lim LY, Vidnovic N, Ellisen LW, Leong CO . (2009). Mutant p53 mediates survival of breast cancer cells. Br J Cancer 101: 1606–1612.
Lohrum MA, Woods DB, Ludwig RL, Bálint E, Vousden KH . (2001). C-terminal ubiquitination of p53 contributes to nuclear export. Mol Cell Biol 21: 8521–8532.
Lowe SW, Ruley HE, Jacks T, Housman DE . (1993a). p53-dependent apoptosis modulates the cytotoxicity of anticancer agents. Cell 74: 957–967.
Lowe SW, Schmitt EM, Smith SW, Osborne BA, Jacks T . (1993b). p53 is required for radiation-induced apoptosis in mouse thymocytes. Nature 362: 847–849.
Maier B, Gluba W, Bernier B, Turner T, Mohammad K, Guise T et al. (2004). Modulation of mammalian life span by the short isoform of p53. Genes Dev 18: 306–319.
McGill G, Fisher DE . (1999). P53 and cancer therapy: a double-edged sword. J Clin Invest 104: 223–225.
Muller PA, Caswell PT, Doyle B, Iwanicki MP, Tan EH, Karim S et al. (2009). Mutant p53 drives invasion by promoting integrin recycling. Cell 139: 1327–1341.
Nakamura S, Roth JA, Mukhopadhyay T . (2000). Multiple lysine mutations in the C-terminal domain of p53 interfere with MDM2-dependent protein degradation and ubiquitination. Mol Cell Biol 20: 9391–9398.
Nelson MH, Stein DA, Kroeker AD, Hatlevig SA, Iversen PL, Moulton HM . (2005). Arginine-rich peptide conjugation to morpholino oligomers: Effects on antisense activity and specificity. Bioconjug Chem 16: 959–966.
O'Connor PM, Jackman J, Bae I, Myers TG, Fan S, Mutoh M et al. (1997). Characterization of the p53 tumor suppressor pathway in cell lines of the National Cancer Institute anticancer drug screen and correlations with the growth-inhibitory potency of 123 anticancer agents. Cancer Res 57: 4285–4300.
Olive KP, Tuveson DA, Ruhe ZC, Yin B, Willis NA, Bronson RT et al. (2004). Mutant p53 gain of function in two mouse models of Li-Fraumeni syndrome. Cell 119: 847–860.
Ray PS, Grover R, Das S . (2006). Two internal ribosome entry sites mediate the translation of p53 isoforms. EMBO Rep 7: 404–410.
Rodriguez MS, Desterro JM, Lain S, Lane DP, Hay RT . (2000). Multiple C-terminal lysine residues target p53 for ubiquitin-proteasome-mediated degradation. Mol Cell Biol 20: 8458–8467.
Rovinski B, Munroe D, Peacock J, Mowat M, Bernstein A, Benchimol S . (1987). Deletion of 5′-coding sequences of the cellular p53 gene in mouse erythroleukemia: a novel mechanism of oncogene regulation. Mol Cell Biol 7: 847–853.
Stambolsky P, Tabach Y, Fontemaggi G, Weisz L, Maor-Aloni R, Sigfried Z et al. (2010). Modulation of the vitamin D3 response by cancer-associated mutant p53. Cancer Cell 17: 273–285.
Summerton J, Weller D . (1997). Morpholino antisense oligomers: design, preparation, and properties. Antisense Nucleic Acid Drug Dev 7: 187–195.
Vousden KH, Prives C . (2009). Blinded by the light: the growing complexity of p53. Cell 137: 413–431.
Wang C, Curtis JE, Minden MD, McCulloch EA . (1989). Expression of a retinoic acid receptor gene in myeloid leukemia cells. Leukemia 3: 264–269.
Wu RP, Youngblood DS, Hassinger JN, Lovejoy CE, Nelson MH, Iversen PL et al. (2007). Cell-penetrating peptides as transporters for morpholino oligomers: effects of amino acid composition on intracellular delivery and cytotoxicity. Nucleic Acids Res 35: 5182–5191.
Yin Y, Stephen CW, Luciani MG, Fahraeus R . (2002). p53 stability and activity is regulated by Mdm2-mediated induction of alternative p53 translation products. Nat Cell Biol 4: 462–467.
Zhu J, Zhou W, Jiang J, Chen X . (1998). Identification of a novel p53 functional domain that is necessary for mediating apoptosis. J Biol Chem 273: 13030–13036.
Acknowledgements
This work was supported by a grant from Eleos Inc to SB. SB is supported by a Canada Research Chair.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
SB is a member of the scientific advisory board of Eleos Inc. LJS is the founder and CSO of Eleos Inc. PLI is a full-time employee of AVI Biopharma Inc.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Ma, W., Lin, Y., Xuan, W. et al. Inhibition of p53 expression by peptide-conjugated phosphorodiamidate morpholino oligomers sensitizes human cancer cells to chemotherapeutic drugs. Oncogene 31, 1024–1033 (2012). https://doi.org/10.1038/onc.2011.300
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.300
Keywords
This article is cited by
-
MicroRNA-375 is a therapeutic target for castration-resistant prostate cancer through the PTPN4/STAT3 axis
Experimental & Molecular Medicine (2022)
-
Suppression of Jab1/CSN5 induces radio- and chemo-sensitivity in nasopharyngeal carcinoma through changes to the DNA damage and repair pathways
Oncogene (2013)