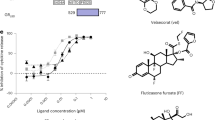
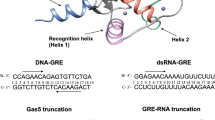
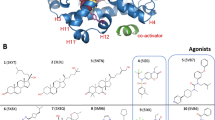
Abstract
The 2.7 Å X-ray crystal structure of the DNA-binding domain (DBD) of the orphan nuclear receptor, nerve growth factor-induced-B (NGFI-B), complexed to its high-affinity DNA target, represents the first structure analysis of a nuclear receptor DBD bound as a monomer to DNA. The structure of the core DBD and its interactions with the major groove of the DNA are similar to previously crystallographically solved DBD–DNA complexes in this superfamily; however, residues C-terminal to this core form a separate and unique substructure that interacts extensively and in a sequence-specific way with the minor groove of its DNA target, in particular with the characteristic 3 A-T base-pair identity element that extends 5' to the usual nuclear receptor half-site (AGGTCA).
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
References
Milbrandt, J. Nerve growth factor induces a gene homologous to the glucocorticoid receptor gene. Neuron 1, 183–188 (1988).
Calnan, B.J., Szychowski, S., Chan, F.K., Cado, D. & Winoto, A. A role for the orphan steroid receptor Nur77 in apoptosis accompanying antigen-induced negative selection. Immunity 3, 273–282 ( 1995).
Cheng, L.E., Chan, F.K., Cado, D. & Winoto, A. Functional redundancy of the Nur77 and Nor-1 orphan steroid receptors in T-cell apoptosis. Embo J. 16, 1865–1875 ( 1997).
Liu, Z.G., Smith, S.W., McLaughlin, K.A., Schwartz, L.M. & Osborne, B.A. Apoptotic signals delivered through the T-cell receptor of a T-cell hybrid require the immediate-early gene nur77. Nature 367, 281–284 (1994).
Bandoh, S., Tsukada, T., Maruyama, K., Ohkura, N. & Yamaguchi, K. Differential expression of NGFI-B and RNR-1 genes in various tissues and developing brain of the rat: comparative study by quantitative reverse transcription–polymerase chain reaction. J. Neuroendocrinol. 9, 3– 8 (1997).
Crawford, P.A., Sadovsky, Y., Woodson, K., Lee, S.L. & Milbrandt, J. Adrenocortical function and regulation of the steroid 21-hydroxylase gene in NGFI-B-deficient mice. Mol. Cell. Biol. 15, 4331–4316 (1995).
Li, Y. & Lau, L.F. Adrenocorticotropic hormone regulates the activities of the orphan nuclear receptor Nur77 through modulation of phosphorylation. Endocrinology 138, 4138 –4146 (1997).
Kastner, P., Mark, M. & Chambon, P. Nonsteroid nuclear receptors: what are genetic studies telling us about their role in real life? Cell 83, 859–869 (1995).
Mangelsdorf, D.J. et al. The nuclear receptor superfamily: the second decade. Cell 83, 835–839 ( 1995).
Wilson, T.E., Fahrner, T.J., Johnston, M. & Milbrandt, J. Identification of the DNA binding site for NGFI-B by genetic selection in yeast. Science 252, 1296– 1300 (1991).
Danielsen, M., Hinck, L. & Ringold, G.M. Two amino acids within the knuckle of the first zinc finger specify DNA response element activation by the glucocorticoid receptor. Cell 57, 1131–1138 (1989).
Mader, S., Kumar, V., de Verneuil, H. & Chambon, P. Three amino acids of the oestrogen receptor are essential to its ability to distinguish an oestrogen from a glucocorticoid-responsive element. Nature 338, 271–274 ( 1989).
Umesono, K. & Evans, R.M. Determinants of target gene specificity for steroid/thyroid hormone receptors. Cell 57, 1139–1146 (1989).
Wilson, T.E., Paulsen, R.E., Padgett, K.A. & Milbrandt, J. Participation of non-zinc finger residues in DNA binding by two nuclear orphan receptors. Science 256, 107– 110 (1992).
Wilson, T.E., Fahrner, T.J. & Milbrandt, J. The orphan receptors NGFI-B and steroidogenic factor 1 establish monomer binding as a third paradigm of nuclear receptor–DNA interaction. Mol. Cell Biol. 13, 5794– 5804 (1993).
Connolly, M.L. The molecular surface package. J. Mol. Graphics 11, 139–141 (1993).
Gewirth, D.T. & Sigler, P.B. The basis for half-site specificity explored through a non-cognate steroid receptor–DNA complex. Nature Struct. Biol. 2, 386–394 (1995).
Segard-Maurel, I. et al. Glucocorticosteroid receptor dimerization investigated by analysis of receptor binding to glucocorticosteroid responsive elements using a monomer–dimer equilibrium model. Biochemistry 35, 1634–1642 (1996).
Luisi, B.F. et al. Crystallographic analysis of the interaction of the glucocorticoid receptor with DNA. Nature 352, 497– 505 (1991).
Rastinejad, F., Perlmann, T., Evans, R.M. & Sigler, P.B. Structural determinants of nuclear receptor assembly on DNA direct repeats. Nature 375, 203–211 (1995).
Mangelsdorf, D.J. & Evans, R.M. The RXR heterodimers and orphan receptors. Cell 83, 841– 850 (1995).
Huth, J.R. et al. The solution structure of an HMG-I(Y)–DNA complex defines a new architectural minor groove binding motif. Nature Struct. Biol. 4, 657–665 ( 1997).
Davis, I.J., Hazel, T.G., Chen, R.H., Blenis, J. & Lau, L.F. Functional domains and phosphorylation of the orphan receptor Nur77. Mol. Endocrinol. 7, 953– 964 (1993).
Hirata, Y., Kiuchi, K., Chen, H.C., Milbrandt, J. & Guroff, G. The phosphorylation and DNA binding of the DNA-binding domain of the orphan nuclear receptor NGFI-B. J Biol. Chem. 268, 24808–24812 (1993).
Lavery, R. & Sklenar, H. Defining the structure of irregular nucleic acids: conventions and principles. J. Biomol. Struct. Dyn. 6, 655–667 ( 1989).
Kissinger, C.R., Liu, B.S., Martin-Blanco, E., Kornberg, T.B. & Pabo, C.O. Crystal structure of an engrailed homeodomain–DNA complex at 2.8 A resolution: a framework for understanding homeodomain–DNA interactions. Cell 63, 579–590 (1990).
Fraenkel, E. & Pabo, C.O. Comparison of X-ray and NMR structures for the Antennapedia homeodomain–DNA complex. Nature Struct. Biol. 5, 692–697 ( 1998).
Rupert, P.B., Daughdrill, G.W., Bowerman, B. & Matthews, B.W. A new DNA-binding motif in the Skn-1 binding domain–DNA complex. Nature Struct. Biol. 5, 484–491 (1998).
Schwabe, J.W., Chapman, L., Finch, J.T. & Rhodes, D. The crystal structure of the estrogen receptor DNA-binding domain bound to DNA: how receptors discriminate between their response elements. Cell 75, 567–578 (1993).
Sem, D.S. et al. NMR spectroscopic studies of the DNA-binding domain of the monomer-binding nuclear orphan receptor, human estrogen related receptor-2. The carboxyl-terminal extension to the zinc-finger region is unstructured in the free form of the protein. J. Biol. Chem. 272, 18038– 18043 (1997).
Zhao, Q., Khorasanizadeh, S., Miyoshi, Y., Lazar, M.A. & Rastinejad, F. Structural elements of an orphan nuclear receptor–DNA complex. Mol. Cell. 1, 849–861 (1998).
Otwinowski, Z. in Data collection and processing (eds Sawyer, L., Isaacs, N. & Bailey, S.W.) 56–62 (Science and Engineering Council/Daresbury Laboratory, Warrington, UK; 1993).
The CCP4 suite: programs for protein crystallography. Acta Crystallogr. D 50, 760–763 (1994).
Otwinowski, Z. in Proceedings of the CCP4 study weekend (eds Wolf, W., Evans,P.R., & Leslie, A.G.W.) 80–86 (SERC Daresbury Laboratory, Warrington, UK; 1991).
Jones, T.A., Zou, J.Y., Cowan, S.W. & Kjeldgaard, M. Improved methods for binding protein models in electron density maps and the location of errors in these models. Acta Crystallogr A 47, 110–119 (1991).
Read, R.J. Improved Fourier coefficients for maps using phases from partial structures with errors. Acta Crystallogr. A 42, 140 –149 (1986).
Brunger, A.T. X-PLOR Version 3.1: a system for X-ray crystallography and NMR, (Yale University Press, New Haven, Connecticut; 1992).
Jiang, J.S. & Brunger, A.T. Protein hydration observed by X-ray diffraction. Solvation properties of penicillopepsin and neuraminidase crystal structures. J. Mol. Biol. 243, 100 –115 (1994).
Carson, M. Ribbons 2.0. J. Appl. Crystallogr. 24, 958 –961 (1991).
Nicholls, A., Sharp, K.A. & Honig, B. Protein folding and association: insights from the interfacial and thermodynamic properties of hydrocarbons. Proteins 11, 281–296 (1991).
Evans, S.V. SETOR: Hardware-lighted three-dimensional–solid model representations of macromolecules. J. Mol. Graphics 11, 134–138 (1993).
McDonald, I.K. & Thornton, J.M. Satisfying hydrogen bonding potential in proteins. J. Mol. Biol. 238, 777–793 (1994).
Ikeda, Y. et al. Characterization of the mouse FTZ-F1 gene, which encodes a key regulator of steroid hydroxylase gene expression. Mol. Endocrinol. 7, 852–860 ( 1993).
Lavorgna, G., Ueda, H., Clos, J. & Wu, C. FTZ-F1, a steroid hormone receptor-like protein implicated in the activation of fushi tarazu. Science 252, 848–851 ( 1991).
Bonnelye, E. et al. The ERR-1 orphan receptor is a transcriptional activator expressed during bone development. Mol. Endocrinol. 11, 905–916 (1997).
Acknowledgements
We thank T. Wilson and J. Milbrandt for providing the initial clone of NGFI-B, M. Capel for access to and help in the use of X12B beamline at NSLS, Brookhaven National Laboratory. Work at Yale was supported in part by a grant from the NIH. G.M. is a recipient of a NIH National Research Service Award.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Meinke, G., Sigler, P. DNA-binding mechanism of the monomeric orphan nuclear receptor NGFI-B . Nat Struct Mol Biol 6, 471–477 (1999). https://doi.org/10.1038/8276
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/8276
This article is cited by
-
DNA-binding properties of the MADS-domain transcription factor SEPALLATA3 and mutant variants characterized by SELEX-seq
Plant Molecular Biology (2021)
-
The role of DNA shape in protein–DNA recognition
Nature (2009)
-
Equilibrium Analysis of the DNA Binding Domain of the Ultraspiracle Protein Interaction with the Response Element from the hsp27 Gene Promoter—the Application of Molecular Beacon Technology
Journal of Fluorescence (2008)
-
Energetics of the protein-DNA-water interaction
BMC Structural Biology (2007)
-
Crustacean endocrine toxicology: a review
Ecotoxicology (2007)