Abstract
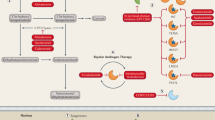
The survival benefit conferred by two hormonal agents in phase III trials has clinically validated the long suspected and now widely recognized phenomenon of castration-resistant prostate cancer (CRPC) hormone dependence. Abiraterone inhibits steroid 17α-hydroxylase/17,20-lyase (CYP17A1) and blocks androgen synthesis, whereas enzalutamide directly binds and antagonizes the androgen receptor. Both agents are highly effective against CRPC and significantly prolong survival following docetaxel treatment. However, this clinical validation of the androgen pathway has led to questions regarding the fundamental mechanisms of CRPC, as well as resistance to abiraterone and enzalutamide. Our understanding of the predominant steroid transformation pathways that lead to dihydrotestosterone synthesis in CRPC is evolving. The role of steroidogenesis in the development of resistance to abiraterone and enzalutamide remains uncertain. The specific roles of candidate enzyme targets in the development of resistance to these agents must be defined if we are to identify novel targets for improved pharmacologic therapies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Huggins, C. & Hodges, C. V. Studies on prostate cancer I: the effect of estrogen and of androgen injection on serum phosphatases in metastatic carcinoma of the prostate. Cancer Res. 1, 293–297 (1941).
Bruchovsky, N. & Wilson, J. D. The conversion of testosterone to 5-alpha-androstan-17-beta-ol-3-one by rat prostate in vivo and in vitro. J. Biol. Chem. 243, 2012–2021 (1968).
Anderson, K. M. & Liao, S. Selective retention of dihydrotestosterone by prostatic nuclei. Nature 219, 277–279 (1968).
Sharifi, N., Gulley, J. L. & Dahut, W. L. Androgen deprivation therapy for prostate cancer. JAMA 294, 238–244 (2005).
Scher, H. I. & Sawyers, C. L. Biology of progressive, castration-resistant prostate cancer: directed therapies targeting the androgen-receptor signaling axis. J. Clin. Oncol. 23, 8253–8261 (2005).
Geller, J. et al. DHT concentrations in human prostate cancer tissue. J. Clin. Endocrinol. Metab. 46, 440–444 (1978).
Titus, M. A., Schell, M. J., Lih, F. B., Tomer, K. B. & Mohler, J. L. Testosterone and dihydrotestosterone tissue levels in recurrent prostate cancer. Clin. Cancer Res. 11, 4653–4657 (2005).
Montgomery, R. B. et al. Maintenance of intratumoral androgens in metastatic prostate cancer: a mechanism for castration-resistant tumor growth. Cancer Res. 68, 4447–4454 (2008).
Chen, C. D. et al. Molecular determinants of resistance to antiandrogen therapy. Nat. Med. 10, 33–39 (2004).
Edwards, J., Krishna, N. S., Grigor, K. M. & Bartlett, J. M. Androgen receptor gene amplification and protein expression in hormone refractory prostate cancer. Br. J. Cancer 89, 552–556 (2003).
Bubendorf, L. et al. Survey of gene amplifications during prostate cancer progression by high-throughout fluorescence in situ hybridization on tissue microarrays. Cancer Res. 59, 803–806 (1999).
Leversha, M. A. et al. Fluorescence in situ hybridization analysis of circulating tumor cells in metastatic prostate cancer. Clin. Cancer Res. 15, 2091–2097 (2009).
Ryan, C. J. & Tindall, D. J. Androgen receptor rediscovered: the new biology and targeting the androgen receptor therapeutically. J. Clin. Oncol. 29, 3651–3658 (2011).
de Bono, J. S. et al. Abiraterone and increased survival in metastatic prostate cancer. N. Engl. J. Med. 364, 1995–2005 (2011).
Tran, C. et al. Development of a second-generation antiandrogen for treatment of advanced prostate cancer. Science 324, 787–790 (2009).
Scher, H. I. et al. Increased survival with enzalutamide in prostate cancer after chemotherapy. N. Engl. J. Med. http://dx.doi.org/10.1056/NEJMoa1207506.
Potter, G. A., Barrie, S. E., Jarman, M. & Rowlands, M. G. Novel steroidal inhibitors of human cytochrome P45017 alpha (17 alpha-hydroxylase-C17, 20-lyase): potential agents for the treatment of prostatic cancer. J. Med. Chem. 38, 2463–2471 (1995).
Labrie, F. Blockade of testicular and adrenal androgens in prostate cancer treatment. Nat. Rev. Urol. 8, 73–85 (2011).
Auchus, R. J. Overview of dehydroepiandrosterone biosynthesis. Semin. Reprod. Med. 22, 281–288 (2004).
Simard, J. et al. Molecular biology of the 3beta-hydroxysteroid dehydrogenase/delta5-delta4 isomerase gene family. Endocr. Rev. 26, 525–582 (2005).
Lorence, M. C., Murry, B. A., Trant, J. M. & Mason, J. I. Human 3 beta-hydroxysteroid dehydrogenase/delta 5----4isomerase from placenta: expression in nonsteroidogenic cells of a protein that catalyzes the dehydrogenation/isomerization of C21 and C19 steroids. Endocrinology 126, 2493–2498 (1990).
Knudsen, K. E. & Penning, T. M. Partners in crime: deregulation of AR activity and androgen synthesis in prostate cancer. Trends Endocrinol. Metab. 21, 315–324 (2010).
Tomkins, G. M. The enzymatic reduction of delta 4-3-ketosteroids. J. Biol. Chem. 225, 13–24 (1957).
Russell, D. W. & Wilson, J. D. Steroid 5 alpha-reductase: two genes/two enzymes. Annu. Rev. Biochem. 63, 25–61 (1994).
Luu-The, V., Belanger, A. & Labrie, F. Androgen biosynthetic pathways in the human prostate. Best Pract. Res. Clin. Endocrinol. Metab. 22, 207–221 (2008).
Chang, K. H. et al. Dihydrotestosterone synthesis bypasses testosterone to drive castration-resistant prostate cancer. Proc. Natl Acad. Sci. USA 108, 13728–13733 (2011).
Campbell, T., Tindall, D. J. & Figg, W. D. Dihydrotestosterone synthesis from adrenal precursors does not involve testosterone in castration-resistant prostate cancer. Cancer Biol. Ther. 13, 237–238 (2012).
Fenner, A. Prostate cancer: DHT bypasses testosterone to drive progression to castration resistance. Nat. Rev. Urol. 8, 470 (2011).
Locke, J. A. et al. Androgen levels increase by intratumoral de novo steroidogenesis during progression of castration-resistant prostate cancer. Cancer Res. 68, 6407–6415 (2008).
Efstathiou, E. et al. Effects of abiraterone acetate on androgen signaling in castrate-resistant prostate cancer in bone. J. Clin. Oncol. 30, 637–643 (2012).
Cai, C. et al. Intratumoral de novo steroid synthesis activates androgen receptor in castration-resistant prostate cancer and is upregulated by treatment with CYP17A1 inhibitors. Cancer Res. 71, 6503–6513 (2011).
Wilson, J. D. et al. 5alpha-androstane-3alpha, 17beta-diol is formed in tammar wallaby pouch young testes by a pathway involving 5alpha-pregnane-3alpha, 17alpha-diol-20-one as a key intermediate. Endocrinology 144, 575–580 (2003).
Miller, W. L. & Auchus, R. J. The molecular biology, biochemistry, and physiology of human steroidogenesis and its disorders. Endocr. Rev. 32, 81–151 (2011).
Auchus, R. J. The backdoor pathway to dihydrotestosterone. Trends Endocrinol. Metab. 15, 432–438 (2004).
Sharifi, N., McPhaul, M. J. & Auchus, R. J. “Getting from here to there”—mechanisms and limitations to the activation of the androgen receptor in castration-resistant prostate cancer. J. Investig. Med. 58, 938–944 (2010).
Mohler, J. L. et al. Activation of the androgen receptor by intratumoral bioconversion of androstanediol to dihydrotestosterone in prostate cancer. Cancer Res. 71, 1486–1496 (2011).
Biswas, M. G. & Russell, D. W. Expression cloning and characterization of oxidative 17beta- and 3alpha-hydroxysteroid dehydrogenases from rat and human prostate. J. Biol. Chem. 272, 15959–15966 (1997).
Bauman, D. R., Steckelbroeck, S., Williams, M. V., Peehl, D. M. & Penning, T. M. Identification of the major oxidative 3alpha-hydroxysteroid dehydrogenase in human prostate that converts 5alpha-androstane-3alpha, 17beta-diol to 5alpha-dihydrotestosterone: a potential therapeutic target for androgen-dependent disease. Mol. Endocrinol. 20, 444–458 (2006).
Belanger, A., Pelletier, G., Labrie, F., Barbier, O. & Chouinard, S. Inactivation of androgens by UDP-glucuronosyltransferase enzymes in humans. Trends Endocrinol. Metab. 14, 473–479 (2003).
Attard, G. et al. Clinical and biochemical consequences of CYP17A1 inhibition with abiraterone given with and without exogenous glucocorticoids in castrate men with advanced prostate cancer. J. Clin. Endocrinol. Metab. 97, 507–516 (2012).
Mostaghel, E. A. et al. Resistance to CYP17A1 inhibition with abiraterone in castration-resistant prostate cancer: induction of steroidogenesis and androgen receptor splice variants. Clin. Cancer Res. 17, 5913–5925 (2011).
Evaul, K., Li, R., Papari-Zareei, M., Auchus, R. J. & Sharifi, N. 3β-hydroxysteroid dehydrogenase is a possible pharmacological target in the treatment of castration-resistant prostate cancer. Endocrinology 151, 3514–3520 (2010).
Li, R. et al. Abiraterone inhibits 3β-hydroxysteroid dehydrogenase: a rationale for increasing drug exposure in castration-resistant prostate cancer. Clin. Cancer Res. 18, 3571–3579 (2012).
Richards, J. et al. Interactions of abiraterone, eplerenone, and prednisolone with wild-type and mutant androgen receptor: a rationale for increasing abiraterone exposure or combining with MDV3100. Cancer Res. 72, 2176–2182 (2012).
Mahon, K. L., Henshall, S. M., Sutherland, R. L. & Horvath, L. G. Pathways of chemotherapy resistance in castration-resistant prostate cancer. Endocr. Relat. Cancer 18, R103–123 (2011).
Author information
Authors and Affiliations
Contributions
Both authors contributed towards researching, discussing, writing, and editing the manuscript.
Corresponding author
Ethics declarations
Competing interests
N. Sharifi has been compensated as a consultant for Janssen.
Rights and permissions
About this article
Cite this article
Chang, KH., Sharifi, N. Prostate cancer—from steroid transformations to clinical translation. Nat Rev Urol 9, 721–724 (2012). https://doi.org/10.1038/nrurol.2012.175
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrurol.2012.175
This article is cited by
-
Prostate cancer
Nature Reviews Disease Primers (2021)
-
Ligand-independent requirements of steroid receptors EcR and USP for cell survival
Cell Death & Differentiation (2016)
-
Conversion of abiraterone to D4A drives anti-tumour activity in prostate cancer
Nature (2015)
-
Chrebp regulates the transcriptional activity of androgen receptor in prostate cancer
Tumor Biology (2014)