Key Points
-
Traditional vascular risk factors are more common in young patients with stroke—especially those over 35 years of age—than was previously thought; thus, 'young' stroke increasingly resembles 'old' stroke
-
Long-term secondary prevention after stroke seems as important in young adults as in older individuals, although trials are needed to establish which patients will benefit from different treatment strategies
-
Poor functional outcome and psychosocial problems are common among young patients (under 50 years of age) with stroke
-
The long-term prevalence of psychosocial consequences, and their influence on daily life, must be further investigated, so as to optimize specific rehabilitation programmes for young patients with stroke
Abstract
Contrary to trends in most other diseases, the average age of ischaemic stroke onset is decreasing, owing to a rise in the incidence of stroke among 'young' individuals (under 50 years of age). This Review provides a critical overview of the risk factors and aetiology of young ischaemic stroke and addresses its long-term prognosis, including cardiovascular risk, functional outcome and psychosocial consequences. We highlight the diminishing role of 'rare' risk factors in the pathophysiology of young stroke in light of the rising prevalence of 'traditional' vascular risk factors in younger age groups. Long-term prognosis is of particular interest to young patients, because of their long life expectancy and major responsibilities during a demanding phase of life. The prognosis of young stroke is not as favourable as previously thought, with respect either to mortality or cardiovascular disease or to psychosocial consequences. Therefore, secondary stroke prevention is probably a life-long endeavour in most young stroke survivors. Due to under-representation of young patients in past trials, new randomized trials focusing on this age group are needed to confirm the benefits of long-term secondary preventive medication. The high prevalence of poor functional outcome and psychosocial problems warrants further study to optimize treatment and rehabilitation for these young patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
07 October 2014
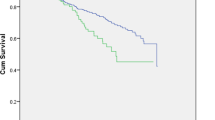
In Figure 3 of the originally published article, the x-axis label was incorrect: it should read 'Follow-up duration (years)'. This error has been corrected in the HTML and PDF versions of the article.
References
Johnston, S. C., Mendis, S. & Mathers, C. D. Global variation in stroke burden and mortality: estimates from monitoring, surveillance, and modelling. Lancet Neurol. 8, 345–354 (2009).
Adamson, J., Beswick, A. & Ebrahim, S. Is stroke the most common cause of disability? J. Stroke Cerebrovasc. Dis. 13, 171–177 (2004).
Nedeltchev, K. et al. Ischaemic stroke in young adults: predictors of outcome and recurrence. J. Neurol. Neurosurg. Psychiatry 76, 191–195 (2005).
Rutten-Jacobs, L. C. et al. Risk factors and prognosis of young stroke. The FUTURE study: a prospective cohort study. Study rationale and protocol. BMC Neurol. 11, 109–116 (2011).
Putaala, J. et al. Analysis of 1008 consecutive patients aged 15 to 49 with first-ever ischemic stroke: the Helsinki young stroke registry. Stroke 40, 1195–1203 (2009).
Kappelle, L. J. et al. Prognosis of young adults with ischemic stroke. A long-term follow-up study assessing recurrent vascular events and functional outcome in the Iowa Registry of Stroke in Young Adults. Stroke 25, 1360–1365 (1994).
Waje-Andreassen, U. et al. Ischaemic stroke at a young age is a serious event—final results of a population-based long-term follow-up in Western Norway. Eur. J. Neurol. 20, 818–823 (2013).
Yesilot Barlas, N. et al. Etiology of first-ever ischaemic stroke in European young adults: the 15 cities young stroke study. Eur. J. Neurol. 20, 1431–1439 (2013).
von Sarnowski, B. et al. Lifestyle risk factors for ischemic stroke and transient ischemic attack in young adults in the Stroke in Young Fabry Patients study. Stroke 44, 119–125 (2013).
Kissela, B. M. et al. Age at stroke: temporal trends in stroke incidence in a large, biracial population. Neurology 79, 1781–1787 (2012).
Kittner, S. J. & Singhal, A. B. Premature atherosclerosis: a major contributor to early-onset ischemic stroke. Neurology 80, 1272–1273 (2013).
O'Donnell, M. J. et al. Risk factors for ischaemic and intracerebral haemorrhagic stroke in 22 countries (the INTERSTROKE study): a case–control study. Lancet 376, 112–123 (2010).
Pollock, A., St George, B., Fenton, M. & Firkins, L. Top 10 research priorities relating to life after stroke—consensus from stroke survivors, caregivers, and health professionals. Int. J. Stroke 9, 313–320 (2014).
Ferro, J. M., Massaro, A. R. & Mas, J. L. Aetiological diagnosis of ischaemic stroke in young adults. Lancet Neurol. 9, 1085–1096 (2010).
Warlow, C. P. et al. Stroke: Practical Management 3rd edn 353–410 (Wiley-Blackwell, 2008).
Ay, H. et al. An evidence-based causative classification system for acute ischemic stroke. Ann. Neurol. 58, 688–697 (2005).
Scott, R. M. & Smith, E. R. Moyamoya disease and moyamoya syndrome. N. Engl. J. Med. 360, 1226–1237 (2009).
Schievink, W. I. Spontaneous dissection of the carotid and vertebral arteries. N. Engl. J. Med. 344, 898–906 (2001).
Debette, S. & Leys, D. Cervical-artery dissections: predisposing factors, diagnosis, and outcome. Lancet Neurol. 8, 668–678 (2009).
Ferro, J. M. Vasculitis of the central nervous system. J. Neurol. 245, 766–776 (1998).
Oyoo, O. & Espinoza, L. R. Infection-related vasculitis. Curr. Rheumatol. Rep. 7, 281–287 (2005).
Urbanus, R. T. et al. Antiphospholipid antibodies and risk of myocardial infarction and ischaemic stroke in young women in the RATIO study: a case–control study. Lancet Neurol. 8, 998–1005 (2009).
Brey, R. L. et al. Antiphospholipid antibodies and stroke in young women. Stroke 33, 2396–2400 (2002).
Hill, A. B. The environment and disease: association or causation? Proc. R. Soc. Med. 58, 295–300 (1965).
Grau, A. J., Urbanek, C. & Palm, F. Common infections and the risk of stroke. Nat. Rev. Neurol. 6, 681–694 (2010).
Pezzini, A. et al. Common genetic markers and prediction of recurrent events after ischemic stroke in young adults. Neurology 73, 717–723 (2009).
Putaala, J. et al. Demographic and geographic vascular risk factor differences in European young adults with ischemic stroke: the 15 Cities young stroke study. Stroke 43, 2624–2630 (2012).
Rutten-Jacobs, L. C. et al. Long-term risk of recurrent vascular events after young stroke: the FUTURE study. Ann. Neurol. 74, 592–601 (2013).
Varona, J. F., Bermejo, F., Guerra, J. M. & Molina, J. A. Long-term prognosis of ischemic stroke in young adults. Study of 272 cases. J. Neurol. 251, 1507–1514 (2004).
George, M. G., Tong, X., Kuklina, E. V. & Labarthe, D. R. Trends in stroke hospitalizations and associated risk factors among children and young adults, 1995–2008. Ann. Neurol. 70, 713–721 (2011).
Reis, J. P. et al. Cardiovascular health through young adulthood and cognitive functioning in midlife. Ann. Neurol. 73, 170–179 (2013).
Ji, R., Schwamm, L. H., Pervez, M. A. & Singhal, A. B. Ischemic stroke and transient ischemic attack in young adults: risk factors, diagnostic yield, neuroimaging, and thrombolysis. JAMA Neurol. 70, 51–57 (2013).
Makowski, M. R. & Botnar, R. M. MR imaging of the arterial vessel wall: molecular imaging from bench to bedside. Radiology 269, 34–51 (2013).
Fonarow, G. C. et al. Age-related differences in characteristics, performance measures, treatment trends, and outcomes in patients with ischemic stroke. Circulation 121, 879–891 (2010).
Marini, C. et al. Stroke in young adults in the community-based L'Aquila registry: incidence and prognosis. Stroke 32, 52–56 (2001).
Putaala, J. et al. Causes of death and predictors of 5-year mortality in young adults after first-ever ischemic stroke: the Helsinki Young Stroke Registry. Stroke 40, 2698–2703 (2009).
Rutten-Jacobs, L. C. et al. Long-term mortality after stroke among adults aged 18 to 50 years. JAMA 309, 1136–1144 (2013).
Waje-Andreassen, U., Naess, H., Thomassen, L., Eide, G. E. & Vedeler, C. A. Long-term mortality among young ischemic stroke patients in western Norway. Acta Neurol. Scand. 116, 150–156 (2007).
Camerlingo, M. et al. Recurrence after first cerebral infarction in young adults. Acta Neurol. Scand. 102, 87–93 (2000).
Naess, H., Nyland, H. I., Thomassen, L., Aarseth, J. & Myhr, K. M. Long-term outcome of cerebral infarction in young adults. Acta Neurol. Scand. 110, 107–112 (2004).
Putaala, J. et al. Recurrent ischemic events in young adults after first-ever ischemic stroke. Ann. Neurol. 68, 661–671 (2010).
Redfors, P. et al. Stroke subtype predicts outcome in young and middle-aged stroke sufferers. Acta Neurol. Scand. 126, 329–335 (2012).
Singhal, A. B. et al. Recognition and management of stroke in young adults and adolescents. Neurology 81, 1089–1097 (2013).
Marini, C., Totaro, R. & Carolei, A. Long-term prognosis of cerebral ischemia in young adults. National Research Council Study Group on Stroke in the Young. Stroke 30, 2320–2325 (1999).
Tsivgoulis, G. et al. Racial disparities in early mortality in 1,134 young patients with acute stroke. Neurol. Sci. http://dx.doi.org/10.1007/s10072-014-1640–9.
Leys, D. et al. Clinical outcome in 287 consecutive young adults (15 to 45 years) with ischemic stroke. Neurology 59, 26–33 (2002).
Adams, R. J. et al. Update to the AHA/ASA recommendations for the prevention of stroke in patients with stroke and transient ischemic attack. Stroke 39, 1647–1652 (2008).
Rodgers, A. et al. Perindopril-based blood pressure lowering in individuals with cerebrovascular disease: consistency of benefits by age, sex and region. J. Hypertens. 22, 653–659 (2004).
Calvet, D. & Mas, J. L. Closure of patent foramen ovale in cryptogenic stroke: a never ending story. Curr. Opin. Neurol. 27, 13–19 (2014).
Carroll, J. D. et al. Closure of patent foramen ovale versus medical therapy after cryptogenic stroke. N. Engl. J. Med. 368, 1092–1100 (2013).
Meier, B. et al. Percutaneous closure of patent foramen ovale in cryptogenic embolism. N. Engl. J. Med. 368, 1083–1091 (2013).
Furie, K. L. et al. Guidelines for the prevention of stroke in patients with stroke or transient ischemic attack: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 42, 227–276 (2011).
Panichpisal, K., Rozner, E. & Levine, S. R. The management of stroke in antiphospholipid syndrome. Curr. Rheumatol. Rep. 14, 99–106 (2012).
Knoflach, M. et al. Functional recovery after ischemic stroke—a matter of age: data from the Austrian Stroke Unit Registry. Neurology 78, 279–285 (2012).
Spengos, K. & Vemmos, K. Risk factors, etiology, and outcome of first-ever ischemic stroke in young adults aged 15 to 45—the Athens young stroke registry. Eur. J. Neurol. 17, 1358–1364 (2010).
Arntz, R. M. et al. Epilepsy after TIA or stroke in young patients impairs long-term functional outcome: the FUTURE Study. Neurology 81, 1907–1913 (2013).
Varona, J. F. Long-term prognosis of ischemic stroke in young adults. Stroke Res. Treat. 2011, 879817 (2010).
Lamy, C. et al. Early and late seizures after cryptogenic ischemic stroke in young adults. Neurology 60, 400–404 (2003).
Neau, J. P. et al. Functional recovery and social outcome after cerebral infarction in young adults. Cerebrovasc. Dis. 8, 296–302 (1998).
Arntz, R. et al. Post-stroke epilepsy in young adults: a long-term follow-up study. PLoS ONE 8, e55498 (2013).
Klit, H., Finnerup, N. B. & Jensen, T. S. Central post-stroke pain: clinical characteristics, pathophysiology, and management. Lancet Neurol. 8, 857–868 (2009).
Naess, H., Lunde, L., Brogger, J. & Waje-Andreassen, U. Post-stroke pain on long-term follow-up: the Bergen stroke study. J. Neurol. 257, 1446–1452 (2010).
Raffaeli, W., Minella, C. E., Magnani, F. & Sarti, D. Population-based study of central post-stroke pain in Rimini district, Italy. J. Pain Res. 6, 705–711 (2013).
Hansen, A. P. et al. Pain following stroke: a prospective study. Eur. J. Pain 16, 1128–1136 (2012).
Naess, H. & Nyland, H. Poor health-related quality of life is associated with long-term mortality in young adults with cerebral infarction. J. Stroke Cerebrovasc. Dis. 22, e79–e83 (2013).
Hommel, M., Miguel, S. T., Naegele, B., Gonnet, N. & Jaillard, A. Cognitive determinants of social functioning after a first ever mild to moderate stroke at vocational age. J. Neurol. Neurosurg. Psychiatry 80, 876–880 (2009).
Cao, M., Ferrari, M., Patella, R., Marra, C. & Rasura, M. Neuropsychological findings in young-adult stroke patients. Arch. Clin. Neuropsychol. 22, 133–142 (2007).
Malm, J. et al. Cognitive impairment in young adults with infratentorial infarcts. Neurology 51, 433–440 (1998).
Schaapsmeerders, P. et al. Long-term cognitive impairment after first-ever ischemic stroke in young adults. Stroke 44, 1621–1628 (2013).
Sachdev, P. S. et al. The neuropsychological profile of vascular cognitive impairment in stroke and TIA patients. Neurology 62, 912–919 (2004).
Gratton, C., Nomura, E. M., Perez, F. & D'Esposito, M. Focal brain lesions to critical locations cause widespread disruption of the modular organization of the brain. J. Cogn. Neurosci. 24, 1275–1285 (2012).
Ferro, J. M. & Madureira, S. Aphasia type, age and cerebral infarct localisation. J. Neurol. 244, 505–509 (1997).
Kauranen, T. et al. The severity of cognitive deficits predicts return to work after a first-ever ischaemic stroke. J. Neurol. Neurosurg. Psychiatry 84, 316–321 (2013).
Rowe, F. & VIS Group UK. Visual perceptual consequences of stroke. Strabismus 17, 24–28 (2009).
Naess, H., Nyland, H. I., Thomassen, L., Aarseth, J. & Myhr, K. M. Mild depression in young adults with cerebral infarction at long-term follow-up: a population-based study. Eur. J. Neurol. 12, 194–198 (2005).
Santos, C. O., Caeiro, L., Ferro, J. M. & Figueira, M. L. A study of suicidal thoughts in acute stroke patients. J. Stroke Cerebrovasc. Dis. 21, 749–754 (2012).
Pompili, M. et al. Do stroke patients have an increased risk of developing suicidal ideation or dying by suicide? An overview of the current literature. CNS Neurosci. Ther. 18, 711–721 (2012).
Krishnan, K. R., Hays, J. C. & Blazer, D. G. MRI-defined vascular depression. Am. J. Psychiatry 154, 497–501 (1997).
Calvert, T., Knapp, P. & House, A. Psychological associations with emotionalism after stroke. J. Neurol. Neurosurg. Psychiatry 65, 928–929 (1998).
Naess, H., Nyland, H. I., Thomassen, L., Aarseth, J. & Myhr, K. M. Fatigue at long-term follow-up in young adults with cerebral infarction. Cerebrovasc. Dis. 20, 245–250 (2005).
van der Zee, C. H., Visser-Meily, J. M., Lindeman, E., Jaap Kappelle, L. & Post, M. W. Participation in the chronic phase of stroke. Top. Stroke Rehabil. 20, 52–61 (2013).
Andersen, G., Christensen, D., Kirkevold, M. & Johnsen, S. P. Post-stroke fatigue and return to work: a 2-year follow-up. Acta Neurol. Scand. 125, 248–253 (2012).
Snaphaan, L., van der Werf, S. & de Leeuw, F. E. Time course and risk factors of post-stroke fatigue: a prospective cohort study. Eur. J. Neurol. 18, 611–617 (2011).
Tang, W. K. et al. Acute basal ganglia infarcts in poststroke fatigue: an MRI study. J. Neurol. 257, 178–182 (2010).
Korpelainen, J. T., Kauhanen, M. L., Kemola, H., Malinen, U. & Myllyla, V. V. Sexual dysfunction in stroke patients. Acta Neurol. Scand. 98, 400–405 (1998).
Bugnicourt, J. M., Hamy, O., Canaple, S., Lamy, C. & Legrand, C. Impaired sexual activity in young ischaemic stroke patients: an observational study. Eur. J. Neurol. 21, 140–146 (2014).
Calabro, R. S., Gervasi, G. & Bramanti, P. Male sexual disorders following stroke: an overview. Int. J. Neurosci. 121, 598–604 (2011).
Roding, J., Glader, E. L., Malm, J. & Lindstrom, B. Life satisfaction in younger individuals after stroke: different predisposing factors among men and women. J. Rehabil. Med. 42, 155–161 (2010).
Vestling, M., Tufvesson, B. & Iwarsson, S. Indicators for return to work after stroke and the importance of work for subjective well-being and life satisfaction. J. Rehabil. Med. 35, 127–131 (2003).
Persson, J., Ferraz-Nunes, J. & Karlberg, I. Economic burden of stroke in a large county in Sweden. BMC Health Serv. Res. 12, 341–348 (2012).
Glozier, N., Hackett, M. L., Parag, V., Anderson, C. S. & Auckland Regional Community Stroke Study Group (ARCOS). The influence of psychiatric morbidity on return to paid work after stroke in younger adults: the Auckland Regional Community Stroke (ARCOS) Study, 2002 to 2003. Stroke 39, 1526–1532 (2008).
Hannerz, H., Holbaek Pedersen, B., Poulsen, O. M., Humle, F. & Andersen, L. L. A nationwide prospective cohort study on return to gainful occupation after stroke in Denmark 1996–2006. BMJ Open 1, e000180 (2011).
Trygged, S., Ahacic, K. & Kåreholt, I. Income and education as predictors of return to working life among younger stroke patients. BMC Public Health 11, 742 (2011).
Hofgren, C., Bjorkdahl, A., Esbjornsson, E. & Sunnerhagen, K. S. Recovery after stroke: cognition, ADL function and return to work. Acta Neurol. Scand. 115, 73–80 (2007).
Roding, J., Lindstrom, B., Malm, J. & Ohman, A. Frustrated and invisible—younger stroke patients' experiences of the rehabilitation process. Disabil. Rehabil. 25, 867–874 (2003).
Hoffmann, T., Bennett, S., Koh, C. L. & McKenna, K. T. Occupational therapy for cognitive impairment in stroke patients. Cochrane Database of Systematic Reviews, Issue 9. Art. No.: CD006430. http://dx.doi.org/10.1002/14651858.CD006430.pub2.
McGeough, E. et al. Interventions for post-stroke fatigue. Cochrane Database of Systematic Reviews, Issue 3. Art. No.: CD007030. http://dx.doi.org/10.1002/14651858.CD007030.pub2.
Flaster, M., Sharma, A. & Rao, M. Poststroke depression: a review emphasizing the role of prophylactic treatment and synergy with treatment for motor recovery. Top. Stroke Rehabil. 20, 139–150 (2013).
Sacco, S., Ricci, S. & Carolei, A. Migraine and vascular diseases: a review of the evidence and potential implications for management. Cephalalgia 32, 785–795 (2012).
Kurth, T., Chabriat, H. & Bousser, M. G. Migraine and stroke: a complex association with clinical implications. Lancet Neurol. 11, 92–100 (2012).
Pezzini, A. et al. Predictors of migraine subtypes in young adults with ischemic stroke: the Italian Project on Stroke in Young Adults. Stroke 42, 17–21 (2011).
Janssen, A. W., de Leeuw, F. E. & Janssen, M. C. Risk factors for ischemic stroke and transient ischemic attack in patients under age 50. J. Thromb. Thrombolysis 31, 85–91 (2011).
Camerlingo, M., Romorini, A., Ferrante, C., Valente, L. & Moschini, L. Migraine and cerebral infarction in young people. Neurol. Sci. 31, 293–297 (2010).
Spector, J. T. et al. Migraine headache and ischemic stroke risk: an updated meta-analysis. Am. J. Med. 123, 612–624 (2010).
Westover, A. N., McBride, S. & Haley, R. W. Stroke in young adults who abuse amphetamines or cocaine: a population-based study of hospitalized patients. Arch. Gen. Psychiatry 64, 495–502 (2007).
Phillips, M. C. et al. Ischaemic stroke among young people aged 15 to 50 years in Adelaide, South Australia. Med. J. Aust. 195, 610–614 (2011).
Sloan, M. A. et al. Illicit drug-associated ischemic stroke in the Baltimore–Washington Young Stroke Study. Neurology 50, 1688–1693 (1998).
Barber, P. A. et al. Cannabis, ischemic stroke, and transient ischemic attack: a case–control study. Stroke 44, 2327–2329 (2013).
de los Rios, F. et al. Trends in substance abuse preceding stroke among young adults: a population-based study. Stroke 43, 3179–3183 (2012).
Davis, D. et al. Patent foramen ovale, ischemic stroke and migraine: systematic review and stratified meta-analysis of association studies. Neuroepidemiology 40, 56–67 (2013).
Meissner, I. et al. Patent foramen ovale: innocent or guilty? Evidence from a prospective population-based study. J. Am. Coll. Cardiol. 47, 440–445 (2006).
Di Tullio, M. R., Sacco, R. L., Sciacca, R. R., Jin, Z. & Homma, S. Patent foramen ovale and the risk of ischemic stroke in a multiethnic population. J. Am. Coll. Cardiol. 49, 797–802 (2007).
Alsheikh-Ali, A. A., Thaler, D. E. & Kent, D. M. Patent foramen ovale in cryptogenic stroke: incidental or pathogenic? Stroke 40, 2349–2355 (2009).
Plu-Bureau, G., Hugon-Rodin, J., Maitrot-Mantelet, L. & Canonico, M. Hormonal contraceptives and arterial disease: an epidemiological update. Best Pract. Res. Clin. Endocrinol. Metab. 27, 35–45 (2013).
Baillargeon, J. P., McClish, D. K., Essah, P. A. & Nestler, J. E. Association between the current use of low-dose oral contraceptives and cardiovascular arterial disease: a meta-analysis. J. Clin. Endocrinol. Metab. 90, 3863–3870 (2005).
Balci, K., Utku, U., Asil, T. & Celik, Y. Ischemic stroke in young adults: risk factors, subtypes, and prognosis. Neurologist 17, 16–20 (2011).
Nightingale, A. L. & Farmer, R. D. Ischemic stroke in young women: a nested case–control study using the UK General Practice Research Database. Stroke 35, 1574–1578 (2004).
Slooter, A. J. et al. Prothrombotic conditions, oral contraceptives, and the risk of ischemic stroke. J. Thromb. Haemost. 3, 1213–1217 (2005).
Chakhtoura, Z. et al. Progestogen-only contraceptives and the risk of stroke: a meta-analysis. Stroke 40, 1059–1062 (2009).
Lamy, C., Hamon, J. B., Coste, J. & Mas, J. L. Ischemic stroke in young women: risk of recurrence during subsequent pregnancies. French Study Group on Stroke in Pregnancy. Neurology 55, 269–274 (2000).
Kittner, S. J. et al. Pregnancy and the risk of stroke. N. Engl. J. Med. 335, 768–774 (1996).
Tate, J. & Bushnell, C. Pregnancy and stroke risk in women. Womens Health (Lond. Engl.) 7, 363–374 (2011).
Treadwell, S. D., Thanvi, B. & Robinson, T. G. Stroke in pregnancy and the puerperium. Postgrad. Med. J. 84, 238–245 (2008).
Grosset, D. G., Ebrahim, S., Bone, I. & Warlow, C. Stroke in pregnancy and the puerperium: what magnitude of risk? J. Neurol. Neurosurg. Psychiatry 58, 129–131 (1995).
Fusco, M. R. & Harrigan, M. R. Cerebrovascular dissections—a review part I: spontaneous dissections. Neurosurgery 68, 242–257 (2011).
Schievink, W. I., Mokri, B. & Whisnant, J. P. Internal carotid artery dissection in a community. Rochester, Minnesota, 1987–1992 Stroke 24, 1678–1680 (1993).
Lee, V. H., Brown, R. D. Jr, Mandrekar, J. N. & Mokri, B. Incidence and outcome of cervical artery dissection: a population-based study. Neurology 67, 1809–1812 (2006).
Ducros, A. Reversible cerebral vasoconstriction syndrome. Lancet Neurol. 11, 906–917 (2012).
Ducros, A. et al. Hemorrhagic manifestations of reversible cerebral vasoconstriction syndrome: frequency, features, and risk factors. Stroke 41, 2505–2511 (2010).
Spengos, K. & Vemmos, K. N. Etiology and outcome of cardioembolic stroke in young adults in Greece. Hellenic J. Cardiol. 51, 127–132 (2010).
Hamedani, A. G. et al. Factor V Leiden and ischemic stroke risk: the Genetics of Early Onset Stroke (GEOS) study. J. Stroke Cerebrovasc. Dis. 22, 419–423 (2013).
Hamedani, A. G., Cole, J. W., Mitchell, B. D. & Kittner, S. J. Meta-analysis of factor V Leiden and ischemic stroke in young adults: the importance of case ascertainment. Stroke 41, 1599–1603 (2010).
Morris, J. G., Singh, S. & Fisher, M. Testing for inherited thrombophilias in arterial stroke: can it cause more harm than good? Stroke 41, 2985–2990 (2010).
Soare, A. M. & Popa, C. Deficiencies of proteins C, S and antithrombin and factor V Leiden and the risk of ischemic strokes. J. Med. Life 3, 235–238 (2010).
Boekholdt, S. M. & Kramer, M. H. Arterial thrombosis and the role of thrombophilia. Semin. Thromb. Hemost. 33, 588–596 (2007).
Fields, M. C. & Levine, S. R. Thrombophilias and stroke: diagnosis, treatment, and prognosis. J. Thromb. Thrombolysis 20, 113–126 (2005).
Moster, M. L. Coagulopathies and arterial stroke. J. Neuroophthalmol. 23, 63–71 (2003).
Brey, R. L. Management of the neurological manifestations of APS—what do the trials tell us? Thromb. Res. 114, 489–499 (2004).
Acknowledgements
F.-E.d.L. received funding from the Dutch Epilepsy Fund (grant 10-18). Also, this work was supported by a Vidi innovational grant from the Dutch Organization for Scientific Research (grant 016.126.351). The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
N.A.M.M.M. and L.C.A.R.-J. contributed equally to this article. N.A.M.M.M., L.C.A.R.-J. and F.-E.d.L. researched the data for the article and wrote the text. N.A.M.M.M., L.C.A.R.-J., E.J.v.D. and F.-E.d.L. provided substantial contributions to discussions of the content. All authors reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Maaijwee, N., Rutten-Jacobs, L., Schaapsmeerders, P. et al. Ischaemic stroke in young adults: risk factors and long-term consequences. Nat Rev Neurol 10, 315–325 (2014). https://doi.org/10.1038/nrneurol.2014.72
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneurol.2014.72
This article is cited by
-
Predictors of social risk for post-ischemic stroke reintegration
Scientific Reports (2024)
-
Factors affecting the quality of life after ischemic stroke in young adults: a scoping review
Health and Quality of Life Outcomes (2023)
-
Associations of effort-reward imbalance at work and quality of life among workers after stroke: a one-year longitudinal study in Thailand
BMC Public Health (2023)
-
Psychometric properties of the 12-item Stroke-Specific Quality of Life Scale among stroke survivors in Hong Kong
Scientific Reports (2023)
-
Emerging frontiers of artificial intelligence and machine learning in ischemic stroke: a comprehensive investigation of state-of-the-art methodologies, clinical applications, and unraveling challenges
EPMA Journal (2023)