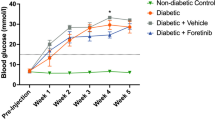
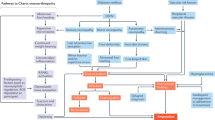
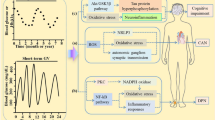
Abstract
In patients with diabetes, nerve injury is a common complication that leads to chronic pain, numbness and substantial loss of quality of life. Good glycemic control can decrease the incidence of diabetic neuropathy, but more than half of all patients with diabetes still develop this complication. There is no approved treatment to prevent or halt diabetic neuropathy, and only symptomatic pain therapies, with variable efficacy, are available. New insights into the mechanisms leading to the development of diabetic neuropathy continue to point to systemic and cellular imbalances in metabolites of glucose and lipids. In the PNS, sensory neurons, Schwann cells and the microvascular endothelium are vulnerable to oxidative and inflammatory stress in the presence of these altered metabolic substrates. This Review discusses the emerging cellular mechanisms that are activated in the diabetic milieu of hyperglycemia, dyslipidemia and impaired insulin signaling. We highlight the pathways to cellular injury, thereby identifying promising therapeutic targets, including mitochondrial function and inflammation.
Key Points
-
Multiple metabolic imbalances underlie the development of diabetic neuropathy
-
Hyperglycemia, dyslipidemia and cardiovascular dysfunction are each independent risk factors for neuropathy
-
Targeting risk factors as well as cellular oxidative stress and inflammation will be important in future treatment approaches
-
Injury to neurons, Schwann cells and microvascular endothelial cells in the diabetic milieu contributes to the pathogenesis of neuropathy
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Dyck, P. J. et al. The prevalence by staged severity of various types of diabetic neuropathy, retinopathy, and nephropathy in a population-based cohort: the Rochester Diabetic Neuropathy Study. Neurology 43, 817–824 (1993).
Dyck, P. J. et al. Variables influencing neuropathic endpoints: the Rochester Diabetic Neuropathy Study of Healthy Subjects. Neurology 45, 1115–1121 (1995).
Edwards, J. L., Vincent, A. M., Cheng, H. T. & Feldman, E. L. Diabetic neuropathy: mechanisms to management. Pharmacol. Ther. 120, 1–34 (2008).
Guastella, V. & Mick, G. Strategies for the diagnosis and treatment of neuropathic pain secondary to diabetic peripheral sensory polyneuropathy. Diabetes Metab. 35, 12–19 (2009).
Veves, A., Backonja, M. & Malik, R. A. Painful diabetic neuropathy: epidemiology, natural history, early diagnosis, and treatment options. Pain Med. 9, 660–674 (2008).
Gandhi, R. A., Marques, J. L., Selvarajah, D., Emery, C. J. & Tesfaye, S. Painful diabetic neuropathy is associated with greater autonomic dysfunction than painless diabetic neuropathy. Diabetes Care 33, 1585–1590 (2010).
Obrosova, I. G. Diabetic painful and insensate neuropathy: pathogenesis and potential treatments. Neurotherapeutics 6, 638–647 (2009).
Athans, W. & Stephens, H. Open calcaneal fractures in diabetic patients with neuropathy: a report of three cases and literature review. Foot Ankle Int. 29, 1049–1053 (2008).
Pop-Busui, R. et al. DCCT and EDIC studies in type 1 diabetes: lessons for diabetic neuropathy regarding metabolic memory and natural history. Curr. Diab. Rep. 10, 276–282 (2010).
Lo, Y. L. et al. The laterality index in the evaluation of distal sensorimotor neuropathy. J. Clin. Neuromuscul. Dis. 10, 18–21 (2008).
Koura, N. H. A comparison of sciatic nerve neuropathy in diabetic and aged rats. Folia Biol. (Krakow) 51, 213–218 (2003).
Said, G., Baudoin, D. & Toyooka, K. Sensory loss, pains, motor deficit and axonal regeneration in length-dependent diabetic polyneuropathy. J. Neurol. 255, 1693–1702 (2008).
Cameron, N. E. & Cotter, M. A. Diabetes causes an early reduction in autonomic ganglion blood flow in rats. J. Diabetes Complications 15, 198–202 (2001).
Zent, R. & Pozzi, A. Angiogenesis in diabetic nephropathy. Semin. Nephrol. 27, 161–171 (2007).
Morales, A. A better future for children with type 1 diabetes: review of the conclusions from the Diabetes Control and Complications Trial and the Epidemiology of Diabetes Interventions and Complications study. J. Ark. Med. Soc. 106, 90–93 (2009).
Dyck, P. J. et al. Risk factors for severity of diabetic polyneuropathy: intensive longitudinal assessment of the Rochester Diabetic Neuropathy Study cohort. Diabetes Care 22, 1479–1486 (1999).
Obrosova, I. G. et al. High-fat diet induced neuropathy of pre-diabetes and obesity: effects of “healthy” diet and aldose reductase inhibition. Diabetes 56, 2598–2608 (2007).
Brownlee, M. Biochemistry and molecular cell biology of diabetic complications. Nature 414, 813–820 (2001).
Cheng, C. & Zochodne, D. W. Sensory neurons with activated caspase-3 survive long-term experimental diabetes. Diabetes 52, 2363–2371 (2003).
Schmeichel, A. M., Schmelzer, J. D. & Low, P. A. Oxidative injury and apoptosis of dorsal root ganglion neurons in chronic experimental diabetic neuropathy. Diabetes 52, 165–171 (2003).
Vincent, A. M. et al. Dyslipidemia-induced neuropathy in mice: the role of oxLDL/LOX-1. Diabetes 58, 2376–2385 (2009).
Vincent, A. M., McLean, L. L., Backus, C. & Feldman, E. L. Short-term hyperglycemia produces oxidative damage and apoptosis in neurons. FASEB J. 19, 638–640 (2005).
Lupachyk, S., Shevalye, H., Maksimchyk, Y., Drel, V. R. & Obrosova, I. G. PARP inhibition alleviates diabetes-induced systemic oxidative stress and neural tissue 4-hydroxynonenal adduct accumulation: correlation with peripheral nerve function. Free Radic. Biol. Med. 50, 1400–1409 (2011).
Chowdhury, S. K. et al. Mitochondrial respiratory chain dysfunction in dorsal root ganglia of streptozotocin-induced diabetic rats and its correction by insulin treatment. Diabetes 59, 1082–1091 (2010).
Vincent, A. M., Russell, J. W., Low, P. & Feldman, E. L. Oxidative stress in the pathogenesis of diabetic neuropathy. Endocr. Rev. 25, 612–628 (2004).
Vincent, A. M. et al. SOD2 protects neurons from injury in cell culture and animal models of diabetic neuropathy. Exp. Neurol. 208, 216–227 (2007).
Vincent, A. M. & Feldman, E. L. New insights into the mechanisms of diabetic neuropathy. Rev. Endocr. Metab. Disord. 5, 227–236 (2004).
Bruckner, B. A., Ammini, C. V., Otal, M. P., Raizada, M. K. & Stacpoole, P. W. Regulation of brain glucose transporters by glucose and oxygen deprivation. Metabolism 48, 422–431 (1999).
Yan, L. J., Levine, R. L. & Sohal, R. S. Oxidative damage during aging targets mitochondrial aconitase. Proc. Natl Acad. Sci. USA 94, 11168–11172 (1997).
Morrison, A. D., Clements, R. S. Jr & Winegrad, A. I. Effects of elevated glucose concentrations on the metabolism of the aortic wall. J. Clin. Invest. 51, 3114–3123 (1972).
Obrosova, I., Faller, A., Burgan, J., Ostrow, E. & Williamson, J. R. Glycolytic pathway, redox state of NAD(P)-couples and energy metabolism in lens in galactose-fed rats: effect of an aldose reductase inhibitor. Curr. Eye Res. 16, 34–43 (1997).
Obrosova, I. G. Diabetes and the peripheral nerve. Biochim. Biophys. Acta 1792, 931–940 (2009).
Schemmel, K. E., Padiyara, R. S. & D'Souza, J. J. Aldose reductase inhibitors in the treatment of diabetic peripheral neuropathy: a review. J. Diabetes Complications 24, 354–360 (2010).
Obrosova, I. G. Increased sorbitol pathway activity generates oxidative stress in tissue sites for diabetic complications. Antioxid. Redox Signal. 7, 1543–1552 (2005).
Askwith, T., Zeng, W., Eggo, M. C. & Stevens, M. J. Oxidative stress and dysregulation of the taurine transporter in high-glucose-exposed human Schwann cells: implications for pathogenesis of diabetic neuropathy. Am. J. Physiol. Endocrinol. Metab. 297, E620–E628 (2009).
Tattersall, R. Alpha-glucosidase inhibition as an adjunct to the treatment of type 1 diabetes. Diabet. Med. 10, 688–693 (1993).
Issad, T. & Kuo, M. O-GlcNAc modification of transcription factors, glucose sensing and glucotoxicity. Trends Endocrinol. Metab. 19, 380–389 (2008).
Wiggin, T. D. et al. Rosiglitazone treatment reduces diabetic neuropathy in streptozotocin-treated DBA/2J mice. Endocrinology 149, 4928–4937 (2008).
Duran-Jimenez, B. et al. Advanced glycation end products in extracellular matrix proteins contribute to the failure of sensory nerve regeneration in diabetes. Diabetes 58, 2893–2903 (2009).
Balakumar, P., Rohilla, A., Krishan, P., Solairaj, P. & Thangathirupathi, A. The multifaceted therapeutic potential of benfotiamine. Pharmacol. Res. 61, 482–488 (2010).
Miyazawa, T., Nakagawa, K., Shimasaki, S. & Nagai, R. Lipid glycation and protein glycation in diabetes and atherosclerosis. Amino Acids doi:10.1007/s00726-010-0772-3.
Vincent, A. M. et al. Receptor for advanced glycation end products activation injures primary sensory neurons via oxidative stress. Endocrinology 148, 548–558 (2007).
Drel, V. R., Pacher, P., Stevens, M. J. & Obrosova, I. G. Aldose reductase inhibition counteracts nitrosative stress and poly(ADP-ribose) polymerase activation in diabetic rat kidney and high-glucose-exposed human mesangial cells. Free Radic. Biol. Med. 40, 1454–1465 (2006).
Toth, C. et al. Receptor for advanced glycation end products (RAGEs) and experimental diabetic neuropathy. Diabetes 57, 1002–1017 (2008).
Cameron, N. E., Gibson, T. M., Nangle, M. R. & Cotter, M. A. Inhibitors of advanced glycation end product formation and neurovascular dysfunction in experimental diabetes. Ann. NY Acad. Sci. 1043, 784–792 (2005).
Bierhaus, A. & Nawroth, P. P. Multiple levels of regulation determine the role of the receptor for AGE (RAGE) as common soil in inflammation, immune responses and diabetes mellitus and its complications. Diabetologia 52, 2251–2263 (2009).
Vincent, A. M., Hinder, L. M., Pop-Busui, R. & Feldman, E. L. Hyperlipidemia: a new therapeutic target for diabetic neuropathy. J. Peripher. Nerv. Syst. 14, 257–267 (2009).
Fioretto, P., Dodson, P. M., Ziegler, D. & Rosenson, R. S. Residual microvascular risk in diabetes: unmet needs and future directions. Nat. Rev. Endocrinol. 6, 19–25 (2010).
Wiggin, T. D. et al. Elevated triglycerides correlate with progression of diabetic neuropathy. Diabetes 58, 1634–1640 (2009).
Tesfaye, S. et al. Diabetic neuropathies: update on definitions, diagnostic criteria, estimation of severity, and treatments. Diabetes Care 33, 2285–2293 (2010).
Tesfaye, S. & Selvarajah, D. The Eurodiab study: what has this taught us about diabetic peripheral neuropathy? Curr. Diab. Rep. 9, 432–434 (2009).
Lacigova, S. et al. Influence of cardiovascular autonomic neuropathy on atherogenesis and heart function in patients with type 1 diabetes. Diabetes Res. Clin. Pract. 83, 26–31 (2009).
Elliott, J. et al. Large-fiber dysfunction in diabetic peripheral neuropathy is predicted by cardiovascular risk factors. Diabetes Care 32, 1896–1900 (2009).
Karvestedt, L. et al. Peripheral sensory neuropathy associates with micro- or macroangiopathy: results from a population-based study of type 2 diabetic patients in Sweden. Diabetes Care 32, 317–322 (2009).
Rockenfeller, P. et al. Fatty acids trigger mitochondrion-dependent necrosis. Cell Cycle 9, 2836–2842 (2010).
Vincent, A. M. et al. Mitochondrial biogenesis and fission in axons in cell culture and animal models of diabetic neuropathy. Acta Neuropathol. 120, 477–489 (2010).
McAlpine, C. S., Bowes, A. J. & Werstuck, G. H. Diabetes, hyperglycemia and accelerated atherosclerosis: evidence supporting a role for endoplasmic reticulum (ER) stress signaling. Cardiovasc. Hematol. Disord. Drug Targets. 10, 151–157 (2010).
Jiao, P. et al. FFA-induced adipocyte inflammation and insulin resistance: involvement of ER stress and IKKβ pathways. Obesity doi:10.1038/oby.2010.200.
McCall, K. D. et al. Phenylmethimazole blocks palmitate-mediated induction of inflammatory cytokine pathways in 3T3L1 adipocytes and RAW 264.7 macrophages. J. Endocrinol. 207, 343–353 (2010).
Almaguel, F. G. et al. Lipotoxicity-mediated cell dysfunction and death involve lysosomal membrane permeabilization and cathepsin L activity. Brain Res. 1318, 133–143 (2010).
Padilla, A., Descorbeth, M., Almeyda, A. L., Payne, K. & De Leon, M. Hyperglycemia magnifies Schwann cell dysfunction and cell death triggered by PA-induced lipotoxicity. Brain Res. 1370, 64–79 (2011).
Tsintzas, K. et al. Elevated free fatty acids attenuate the insulin-induced suppression of PDK4 gene expression in human skeletal muscle: potential role of intramuscular long-chain acyl-coenzyme A. J. Clin. Endocrinol. Metab. 92, 3967–3972 (2007).
Geng, H. et al. The effects of ox-LDL in human atherosclerosis may be mediated in part via the toll-like receptor 4 pathway. Mol. Cell. Biochem. 342, 201–206 (2010).
Ishiyama, J., Taguchi, R., Yamamoto, A. & Murakami, K. Palmitic acid enhances lectin-like oxidized LDL receptor (LOX-1) expression and promotes uptake of oxidized LDL in macrophage cells. Atherosclerosis 209, 118–124 (2010).
Nowicki, M. et al. Oxidized low-density lipoprotein (oxLDL)-induced cell death in dorsal root ganglion cell cultures depends not on the lectin-like oxLDL receptor-1 but on the toll-like receptor-4. J. Neurosci. Res. 88, 403–412 (2010).
Honjo, T. et al. Essential role of NOXA1 in generation of reactive oxygen species induced by oxidized low-density lipoprotein in human vascular endothelial cells. Endothelium 15, 137–141 (2008).
Stielow, C. et al. Novel Nox inhibitor of oxLDL-induced reactive oxygen species formation in human endothelial cells. Biochem. Biophys. Res. Commun. 344, 200–205 (2006).
Shafaati, M. et al. Marked accumulation of 27-hydroxycholesterol in the brains of Alzheimer's patients with the Swedish APP 670/671 mutation. J. Lipid Res. 52, 1004–1010 (2011).
Jang, E. R. & Lee, C. S. 7-ketocholesterol induces apoptosis in differentiated PC12 cells via reactive oxygen species-dependent activation of NF-κB and Akt pathways. Neurochem. Int. 58, 52–59 (2011).
Sims-Robinson, C., Kim, B., Rosko, A. & Feldman, E. L. How does diabetes accelerate Alzheimer disease pathology? Nat. Rev. Neurol. 6, 551–559 (2010).
Sahin, M. et al. High prevalence of neuropathy in patients with impaired 60-minute oral glucose tolerance test but normal fasting and 120-minute glucose levels. Minerva Endocrinol. 33, 289–296 (2008).
Smith, A. G. & Singleton, J. R. Impaired glucose tolerance and neuropathy. Neurologist 14, 23–29 (2008).
Kim, B., Sullivan, K. A., Backus, C. & Feldman, E. L. Cortical neurons develop insulin resistance and blunted Akt signaling: a potential mechanism contributing to enhanced ischemic injury in diabetes. Antioxid. Redox Signal. 14, 1829–1839 (2011).
Chillaron, J. J. et al. Estimated glucose disposal rate in assessment of the metabolic syndrome and microvascular complications in patients with type 1 diabetes. J. Clin. Endocrinol. Metab. 94, 3530–3534 (2009).
Frangioudakis, G. & Cooney, G. J. Acute elevation of circulating fatty acids impairs downstream insulin signalling in rat skeletal muscle in vivo independent of effects on stress signalling. J. Endocrinol. 197, 277–285 (2008).
Pratchayasakul, W. et al. Effects of high-fat diet on insulin receptor function in rat hippocampus and the level of neuronal corticosterone. Life Sci. 88, 619–627 (2011).
Ziegler, D. et al. Oral treatment with alpha-lipoic acid improves symptomatic diabetic polyneuropathy: the SYDNEY 2 trial. Diabetes Care 29, 2365–2370 (2006).
Genuth, S. Insights from the diabetes control and complications trial/epidemiology of diabetes interventions and complications study on the use of intensive glycemic treatment to reduce the risk of complications of type 1 diabetes. Endocr. Pract. 12 (Suppl. 1), 34–41 (2006).
Martin, C. L. et al. Neuropathy among the diabetes control and complications trial cohort 8 years after trial completion. Diabetes Care 29, 340–344 (2006).
Ce, G. V. et al. Endothelial dysfunction is related to poor glycemic control in adolescents with type 1 diabetes under 5 years of disease: evidence of metabolic memory. J. Clin. Endocrinol. Metab. 96, 1493–1499 (2011).
Jax, T. W. Metabolic memory: a vascular perspective. Cardiovasc. Diabetol. 9, 51 (2010).
Tonna, S., El-Osta, A., Cooper, M. E. & Tikellis, C. Metabolic memory and diabetic nephropathy: potential role for epigenetic mechanisms. Nat. Rev. Nephrol. 6, 332–341 (2010).
[No authors listed] Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). UK Prospective Diabetes Study (UKPDS) Group. Lancet 352, 837–853 (1998).
Van Acker, K. et al. Prevalence and impact on quality of life of peripheral neuropathy with or without neuropathic pain in type 1 and type 2 diabetic patients attending hospital outpatients clinics. Diabetes Metab. 35, 206–213 (2009).
Jensen, T. S. et al. New perspectives on the management of diabetic peripheral neuropathic pain. Diab. Vasc. Dis. Res. 3, 108–119 (2006).
Bril, V. et al. Evidence-based guideline: Treatment of painful diabetic neuropathy: report of the American Academy of Neurology, the American Association of Neuromuscular and Electrodiagnostic Medicine, and the American Academy of Physical Medicine and Rehabilitation. PM R. 3, 345–352, 352.e1–21 (2011).
Lunn, M. P., Hughes, R. A. & Wiffen, P. J. Duloxetine for treating painful neuropathy or chronic pain. Cochrane Database of Systematic Reviews, Issue 4, Art. No.: CD007115. doi:10.1002/14651858.CD007115.pub2 (2009).
Moore, R. A., Straube, S., Wiffen, P. J., Derry, S. & McQuay, H. J. Pregabalin for acute and chronic pain in adults. Cochrane Database of Systematic Reviews, Issue 3, Art. No.: CD007076. doi:10.1002/14651858.CD007076.pub2 (2009).
Saarto, T. & Wiffen, P. J. Antidepressants for neuropathic pain. Cochrane Database of Systematic Reviews, Issue 4, Art. No.: CD005454. doi:10.1002/14651858.CD005454.pub2 (2007).
Wiffen, P. et al. Anticonvulsant drugs for acute and chronic pain. Cochrane Database of Systematic Reviews, Issue 3, Art. No.: CD001133. doi:10.1002/14651858.CD001133.pub2 (2005).
Schwartz, S. et al. Safety and efficacy of tapentadol ER in patients with painful diabetic peripheral neuropathy: results of a randomized-withdrawal, placebo-controlled trial. Curr. Med. Res. Opin. 27, 151–162 (2010).
Gaede, P. et al. Multifactorial intervention and cardiovascular disease in patients with type 2 diabetes. N. Engl. J. Med. 348, 383–393 (2003).
Tesfaye, S. et al. Factors that impact symptomatic diabetic peripheral neuropathy in placebo-administered patients from two 1-year clinical trials. Diabetes Care 30, 2626–2632 (2007).
Asnaghi, V., Gerhardinger, C., Hoehn, T., Adeboje, A. & Lorenzi, M. A role for the polyol pathway in the early neuroretinal apoptosis and glial changes induced by diabetes in the rat. Diabetes 52, 506–511 (2003).
Drel, V. R. et al. Aldose reductase inhibitor fidarestat counteracts diabetes-associated cataract formation, retinal oxidative-nitrosative stress, glial activation, and apoptosis. Int. J. Mol. Med. 21, 667–676 (2008).
Ekshyyan, O. & Aw, T. Y. Apoptosis in acute and chronic neurological disorders. Front. Biosci. 9, 1567–1576 (2004).
Russell, J. W., Sullivan, K. A., Windebank, A. J., Herrmann, D. N. & Feldman, E. L. Neurons undergo apoptosis in animal and cell culture models of diabetes. Neurobiol. Dis. 6, 347–363 (1999).
Srinivasan, S., Stevens, M. J., Sheng, H., Hall, K. E. & Wiley, J. W. Serum from patients with type 2 diabetes with neuropathy induces complement-independent, calcium-dependent apoptosis in cultured neuronal cells. J. Clin. Invest. 102, 1454–1462 (1998).
Schmidt, R. E. Neuronal preservation in the sympathetic ganglia of rats with chronic streptozotocin-induced diabetes. Brain Res. 921, 256–259 (2001).
Kuhad, A. & Chopra, K. Tocotrienol attenuates oxidative-nitrosative stress and inflammatory cascade in experimental model of diabetic neuropathy. Neuropharmacology 57, 456–462 (2009).
Curtin, N. J. PARP inhibitors for cancer therapy. Expert Rev. Mol. Med. 7, 1–20 (2005).
Negi, G., Kumar, A. & Sharma, S. S. Concurrent targeting of nitrosative stress-PARP pathway corrects functional, behavioral and biochemical deficits in experimental diabetic neuropathy. Biochem. Biophys. Res. Commun. 391, 102–106 (2009).
Obrosova, I. G. et al. Aldose reductase inhibition counteracts oxidative-nitrosative stress and poly(ADP-ribose) polymerase activation in tissue sites for diabetes complications. Diabetes 54, 234–242 (2005).
Liang, H. & Tan, A. R. Iniparib, a PARP1 inhibitor for the potential treatment of cancer, including triple-negative breast cancer. IDrugs 13, 646–656 (2010).
Tutt, A. et al. Oral poly(ADP-ribose) polymerase inhibitor olaparib in patients with BRCA1 or BRCA2 mutations and advanced breast cancer: a proof-of-concept trial. Lancet 376, 235–244 (2010).
Kruse, V. et al. PARP inhibitors in oncology: a new synthetic lethal approach to cancer therapy. Acta Clin. Belg. 66, 2–9 (2011).
Chattopadhyay, M. et al. Prolonged preservation of nerve function in diabetic neuropathy in mice by herpes simplex virus-mediated gene transfer. Diabetologia 50, 1550–1558 (2007).
Jeong, J. O. et al. Dual angiogenic and neurotrophic effects of bone marrow-derived endothelial progenitor cells on diabetic neuropathy. Circulation 119, 699–708 (2009).
Naruse, K. et al. Therapeutic neovascularization using cord blood-derived endothelial progenitor cells for diabetic neuropathy. Diabetes 54, 1823–1828 (2005).
Benter, I. F. et al. Angiotensin-(1–7) prevents activation of NADPH oxidase and renal vascular dysfunction in diabetic hypertensive rats. Am. J. Nephrol. 28, 25–33 (2008).
Wind, S. et al. Comparative pharmacology of chemically distinct NADPH oxidase inhibitors. Br. J. Pharmacol. 161, 885–898 (2010).
Stefanska, J. et al. Apocynin decreases hydrogen peroxide and nitrate concentrations in exhaled breath in healthy subjects. Pulm. Pharmacol. Ther. 23, 48–54 (2010).
Matsushima, S. et al. Increased myocardial NAD(P)H oxidase-derived superoxide causes the exacerbation of postinfarct heart failure in type 2 diabetes. Am. J. Physiol. Heart Circ. Physiol. 297, H409–H416 (2009).
Sonta, T. et al. Evidence for contribution of vascular NAD(P)H oxidase to increased oxidative stress in animal models of diabetes and obesity. Free Radic. Biol. Med. 37, 115–123 (2004).
Cotter, M. A. & Cameron, N. E. Effect of the NAD(P)H oxidase inhibitor, apocynin, on peripheral nerve perfusion and function in diabetic rats. Life Sci. 73, 1813–1824 (2003).
Loesch, A., Tang, H., Cotter, M. A. & Cameron, N. E. Sciatic nerve of diabetic rat treated with epoetin delta: effects on C-fibers and blood vessels including pericytes. Angiology 61, 651–668 (2010).
Anand, P. et al. Clinical trial of the p38 MAP kinase inhibitor dilmapimod in neuropathic pain following nerve injury. Eur. J. Pain doi:10.1016/j.ejpain.2011.04.005.
Genovese, M. C. et al. A 24-week, randomized, double-blind, placebo-controlled, parallel group study of the efficacy of oral SCIO-469, a p38 mitogen-activated protein kinase inhibitor, in patients with active rheumatoid arthritis. J. Rheumatol. 38, 846–854 (2011).
Kellogg, A. P., Converso, K., Wiggin, T., Stevens, M. & Pop-Busui, R. Effects of cyclooxygenase-2 gene inactivation on cardiac autonomic and left ventricular function in experimental diabetes. Am. J. Physiol. Heart Circ. Physiol. 296, H453–H461 (2009).
Lecka-Czernik, B. Aleglitazar, a dual PPARα and PPARγ agonist for the potential oral treatment of type 2 diabetes mellitus. IDrugs 13, 793–801 (2010).
Cavender, M. A. & Lincoff, A. M. Therapeutic potential of aleglitazar, a new dual PPAR-α/γ agonist: implications for cardiovascular disease in patients with diabetes mellitus. Am. J. Cardiovasc. Drugs 10, 209–216 (2010).
Canty, J. M. Jr & Smith, T. P. Jr. Modulation of coronary autoregulatory responses by endothelium-derived nitric oxide. Int. J. Cardiol. 50, 207–215 (1995).
Jude, E. B., Dang, C. & Boulton, A. J. Effect of L-arginine on the microcirculation in the neuropathic diabetic foot in type 2 diabetes mellitus: a double-blind, placebo-controlled study. Diabet. Med. 27, 113–116 (2010).
Shimoshige, Y., Enomoto, R., Aoki, T., Matsuoka, N. & Kaneko, S. The involvement of aldose reductase in alterations to neurotrophin receptors and neuronal cytoskeletal protein mRNA levels in the dorsal root ganglion of streptozotocin-induced diabetic rats. Biol. Pharm. Bull. 33, 67–71 (2010).
Kuzumoto, Y., Kusunoki, S., Kato, N., Kihara, M. & Low, P. A. Effect of the aldose reductase inhibitor fidarestat on experimental diabetic neuropathy in the rat. Diabetologia 49, 3085–3093 (2006).
Bril, V., Hirose, T., Tomioka, S. & Buchanan, R. Ranirestat for the management of diabetic sensorimotor polyneuropathy. Diabetes Care 32, 1256–1260 (2009).
Ramirez, M. A. & Borja, N. L. Epalrestat: an aldose reductase inhibitor for the treatment of diabetic neuropathy. Pharmacotherapy 28, 646–655 (2008).
Hotta, N. et al. Long-term clinical effects of epalrestat, an aldose reductase inhibitor, on diabetic peripheral neuropathy: the 3-year, multicenter, comparative Aldose Reductase Inhibitor-Diabetes Complications Trial. Diabetes Care 29, 1538–1544 (2006).
Skalska, S. et al. Neuropathy in a rat model of mild diabetes induced by multiple low doses of streptozotocin: effects of the antioxidant stobadine in comparison with a high-dose α-lipoic acid treatment. Gen. Physiol. Biophys. 29, 50–58 (2010).
Poh, Z. X. & Goh, K. P. A current update on the use of alpha lipoic acid in the management of type 2 diabetes mellitus. Endocr. Metab. Immune Disord. Drug Targets. 9, 392–398 (2009).
Vallianou, N., Evangelopoulos, A. & Koutalas, P. Alpha-lipoic acid and diabetic neuropathy. Rev. Diabet. Stud. 6, 230–236 (2009).
Foster, T. S. Efficacy and safety of α-lipoic acid supplementation in the treatment of symptomatic diabetic neuropathy. Diabetes Educ. 33, 111–117 (2007).
Hoyer, S. & Betz, K. Elimination of the delayed postischemic energy deficit in cerebral cortex and hippocampus of aged rats with a dried, deproteinized blood extract (Actovegin). Arch. Gerontol. Geriatr. 9, 181–192 (1989).
Ziegler, D. et al. Treatment of symptomatic polyneuropathy with actovegin in type 2 diabetic patients. Diabetes Care 32, 1479–1484 (2009).
Lee, H. J. et al. Fenofibrate lowers abdominal and skeletal adiposity and improves insulin sensitivity in OLETF rats. Biochem. Biophys. Res. Commun. 296, 293–299 (2002).
Li, P. et al. Fenofibrate promotes ischemia-induced revascularization through the adiponectin-dependent pathway. Am. J. Physiol. Endocrinol. Metab. 299, E560–E566 (2010).
Zhao, Z. et al. Rosiglitazone and fenofibrate improve insulin sensitivity of pre-diabetic OLETF rats by reducing malonyl-CoA levels in the liver and skeletal muscle. Life Sci. 84, 688–695 (2009).
Ansquer, J. C., Foucher, C., Aubonnet, P. & Le Malicot, K. Fibrates and microvascular complications in diabetes—insight from the FIELD study. Curr. Pharm. Des. 15, 537–552 (2009).
Rajamani, K. et al. Effect of fenofibrate on amputation events in people with type 2 diabetes mellitus (FIELD study): a prespecified analysis of a randomised controlled trial. Lancet 373, 1780–1788 (2009).
Eroglu, C. et al. Gabapentin receptor α2δ-1 is a neuronal thrombospondin receptor responsible for excitatory CNS synaptogenesis. Cell 139, 380–392 (2009).
Backonja, M. M. Use of anticonvulsants for treatment of neuropathic pain. Neurology 59, S14–S17 (2002).
Ermis, N. et al. Gabapentin therapy improves heart rate variability in diabetic patients with peripheral neuropathy. J. Diabetes Complications 24, 229–233 (2010).
Nakamura, J. et al. Polyol pathway hyperactivity is closely related to carnitine deficiency in the pathogenesis of diabetic neuropathy of streptozotocin-diabetic rats. J. Pharmacol. Exp. Ther. 287, 897–902 (1998).
Evans, J. D., Jacobs, T. F. & Evans, E. W. Role of acetyl-L-carnitine in the treatment of diabetic peripheral neuropathy. Ann. Pharmacother. 42, 1686–1691 (2008).
Cameron, N. E. & Cotter, M. A. The effects of 5-hydroxytryptamine 5-HT2 receptor antagonists on nerve conduction velocity and endoneurial perfusion in diabetic rats. Naunyn Schmiedebergs Arch. Pharmacol. 367, 607–614 (2003).
O'Donnell, M. E. et al. The effects of cilostazol on peripheral neuropathy in diabetic patients with peripheral arterial disease. Angiology 59, 695–704 (2008).
Laczy, B. et al. Effects of pentoxifylline and pentosan polysulphate combination therapy on diabetic neuropathy in type 2 diabetes mellitus. Acta Diabetol. 46, 105–111 (2009).
Karachalias, N., Babaei-Jadidi, R., Rabbani, N. & Thornalley, P. J. Increased protein damage in renal glomeruli, retina, nerve, plasma and urine and its prevention by thiamine and benfotiamine therapy in a rat model of diabetes. Diabetologia 53, 1506–1516 (2010).
Katare, R. G. et al. Vitamin B1 analog benfotiamine prevents diabetes-induced diastolic dysfunction and heart failure through Akt/Pim-1-mediated survival pathway. Circ. Heart Fail. 3, 294–305 (2010).
Hills, C. E. & Brunskill, N. J. Cellular and physiological effects of C-peptide. Clin. Sci. (Lond.) 116, 565–574 (2009).
Stevens, M. J., Zhang, W., Li, F. & Sima, A. A. C-peptide corrects endoneurial blood flow but not oxidative stress in type 1 BB/Wor rats. Am. J. Physiol. Endocrinol. Metab. 287, E497–E505 (2004).
Zhang, W. et al. Human C-peptide dose dependently prevents early neuropathy in the BB/Wor-rat. Int. J. Exp. Diabetes Res. 2, 187–193 (2001).
Ekberg, K. & Johansson, B. L. Effect of C-peptide on diabetic neuropathy in patients with type 1 diabetes. Exp. Diabetes Res. 2008, 457912 (2008).
Apfel, S. C., Arezzo, J. C., Brownlee, M., Federoff, H. & Kessler, J. A. Nerve growth factor administration protects against experimental diabetic sensory neuropathy. Brain Res. 634, 7–12 (1994).
Walwyn, W. M. et al. HSV-1-mediated NGF delivery delays nociceptive deficits in a genetic model of diabetic neuropathy. Exp. Neurol. 198, 260–270 (2006).
Cheng, H. T., Dauch, J. R., Hayes, J. M., Hong, Y. & Feldman, E. L. Nerve growth factor mediates mechanical allodynia in a mouse model of type 2 diabetes. J. Neuropathol. Exp. Neurol. 68, 1229–1243 (2009).
Apfel, S. C. et al. Recombinant human nerve growth factor in the treatment of diabetic polyneuropathy. NGF Study Group. Neurology 51, 695–702 (1998).
Apfel, S. C. Nerve growth factor for the treatment of diabetic neuropathy: what went wrong, what went right, and what does the future hold? Int. Rev. Neurobiol. 50, 393–413 (2002).
Joy, S. V. et al. Ruboxistaurin, a protein kinase C β inhibitor, as an emerging treatment for diabetes microvascular complications. Ann. Pharmacother. 39, 1693–1699 (2005).
Danis, R. P. & Sheetz, M. J. Ruboxistaurin: PKC-β inhibition for complications of diabetes. Expert Opin. Pharmacother. 10, 2913–2925 (2009).
Nakae, M. et al. Effects of basic fibroblast growth factor on experimental diabetic neuropathy in rats. Diabetes 55, 1470–1477 (2006).
Feldman, E. L. & Stevens, M. J. Clinical testing in diabetic peripheral neuropathy. Can. J. Neurol. Sci. 21, S3–S7 (1994).
Hsieh, S. T. Pathology and functional diagnosis of small-fiber painful neuropathy. Acta Neurol. Taiwan 19, 82–89 (2010).
Gibbons, C. H., Freeman, R. & Veves, A. Diabetic neuropathy: a cross-sectional study of the relationships among tests of neurophysiology. Diabetes Care 33, 2629–2634 (2010).
Asad, A. et al. Comparison of nerve conduction studies with diabetic neuropathy symptom score and diabetic neuropathy examination score in type-2 diabetics for detection of sensorimotor polyneuropathy. J. Pak. Med. Assoc. 59, 594–598 (2009).
Singleton, J. R. et al. The Utah Early Neuropathy Scale: a sensitive clinical scale for early sensory predominant neuropathy. J. Peripher. Nerv. Syst. 13, 218–227 (2008).
Chao, C. C. et al. Pathophysiology of neuropathic pain in type 2 diabetes: skin denervation and contact heat-evoked potentials. Diabetes Care 33, 2654–2659 (2010).
England, J. D. et al. Practice parameter: the evaluation of distal symmetric polyneuropathy: the role of autonomic testing, nerve biopsy, and skin biopsy (an evidence-based review). Report of the American Academy of Neurology, the American Association of Neuromuscular and Electrodiagnostic Medicine, and the American Academy of Physical Medicine and Rehabilitation. PM R. 1, 14–22 (2009).
Author information
Authors and Affiliations
Contributions
A. M. Vincent researched data for the article. All authors contributed to discussions of the content, writing of the article, and review and/or editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Vincent, A., Callaghan, B., Smith, A. et al. Diabetic neuropathy: cellular mechanisms as therapeutic targets. Nat Rev Neurol 7, 573–583 (2011). https://doi.org/10.1038/nrneurol.2011.137
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneurol.2011.137
This article is cited by
-
Lipid variability and risk of microvascular complications in patients with diabetes: a systematic review and meta-analysis
BMC Endocrine Disorders (2024)
-
Cytokine expression profiles in white blood cells of patients with small fiber neuropathy
BMC Neuroscience (2023)
-
Pyruvate dehydrogenase kinase 1 protects against neuronal injury and memory loss in mouse models of diabetes
Cell Death & Disease (2023)
-
The TRIM21-FOXD1-BCL-2 axis underlies hyperglycaemic cell death and diabetic tissue damage
Cell Death & Disease (2023)
-
The role and mechanisms of microvascular damage in the ischemic myocardium
Cellular and Molecular Life Sciences (2023)