Abstract
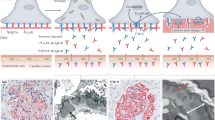
Since its first description in 1968, IgA nephropathy has remained the most common form of idiopathic glomerulonephritis leading to chronic kidney disease in developed countries. The exact pathogenesis of IgA nephropathy is still not well defined. Current data implicate an important genetic factor, especially in promoting the overproduction of an aberrant form of IgA1. The immunochemical aberrancy of IgA nephropathy is characterized by the undergalactosylation of O-glycans in the hinge region of IgA1. However, such aberrant glycosylation alone does not cause renal injury. The next stage of disease development requires the formation of glycan-specific IgG and IgA antibodies that recognize the undergalactosylated IgA1 molecule. These antibodies often have reactivity against antigens from extrinsic microorganisms and might arise from recurrent mucosal infection. B cells that respond to mucosal infections, particularly tonsillitis, might produce the nephritogenic IgA1 molecule. With increased immune-complex formation and decreased clearance owing to reduced uptake by the liver, IgA1 binds to the glomerular mesangium via an as yet unidentified receptor. Glomerular IgA1 deposits trigger the local production of cytokines and growth factors, leading to the activation of mesangial cells and the complement system. Emerging data suggest that mesangial-derived mediators following glomerular deposition of IgA1 lead to podocyte and tubulointerstitial injury via mesangio–podocytic–tubular crosstalk. This Review summarizes the latest findings in the pathogenesis of IgA nephropathy.
Key Points
-
IgA nephropathy is mediated by the deposition of IgA1 immune complexes
-
Increased synthesis of aberrantly galactosylated IgA1 is the first step in disease pathogenesis and remains fundamental to the formation of immune complexes
-
The formation of glycan-specific IgG and IgA antibodies that recognize the undergalactosylated IgA1 is required for the development of disease
-
Genetic factors heavily influence the production of undergalactosylated IgA1 and familial clustering of IgA nephropathy is well recognized
-
The initial event of mesangial deposition of IgA1 immune complexes induces injury to podocytes and renal tubular epithelial cells via a mesangio–podocytic–tubular crosstalk involving various mediators
-
The tubulointerstitial injury that follows chronic inflammation subsequently leads to end-stage renal disease
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Sanders, J. T. & Wyatt, R. J. IgA nephropathy and Henoch-Schönlein purpura nephritis. Curr. Opin. Pediatr. 20, 163–170 (2008).
Roufosse, C. A. & Cook, H. T. Pathological predictors of prognosis in immunoglobulin A nephropathy: a review. Curr. Opin. Nephrol. Hypertens. 18, 212–219 (2009).
Schena, F. P. A retrospective analysis of the natural history of primary IgA nephropathy worldwide. Am. J. Med. 89, 209–215 (1990).
Nair, R. & Walker, P. D. Is IgA nephropathy the commonest primary glomerulopathy among young adults in the USA? Kidney Int. 69, 1455–1458 (2006).
Suzuki, H. et al. Aberrantly glycosylated IgA1 in IgA nephropathy patients is recognized by IgG antibodies with restricted heterogeneity. J. Clin. Invest. 119, 1668–1677 (2009).
Suzuki, Y. et al. Pathological role of tonsillar B cells in IgA nephropathy. Clin. Dev. Immunol. 2011, 639074 (2011).
Mestecky, J. & McGhee, J. R. Immunoglobulin A (IgA): molecular and cellular interactions involved in IgA biosynthesis and immune response. Adv. Immunol. 40, 153–245 (1987).
Hiki, Y. et al. Mass spectrometry proves under-O-glycosylation of glomerular IgA1 in IgA nephropathy. Kidney Int. 59, 1077–1085 (2001).
Tomana, M. et al. Galactose-deficient IgA1 in sera of IgA nephropathy patients is present in complexes with IgG. Kidney Int. 52, 509–516 (1997).
Tomana, M. et al. Circulating immune complexes in IgA nephropathy consist of IgA1 with galactose-deficient hinge region and antiglycan antibodies. J. Clin. Invest. 104, 73–81 (1999).
Ju, T. & Cummings, R. D. A unique molecular chaperone Cosmc required for activity of the mammalian core 1 β3-galactosyltransferase. Proc. Natl Acad. Sci. USA 99, 16613–16618 (2002).
Raska, M. et al. Identification and characterization of CMP-NeuAc:GalNAc-IgA1 α2,6-sialyltransferase in IgA1-producing cells. J. Mol. Biol. 369, 69–78 (2007).
Suzuki, H. et al. IgA1-secreting cell lines from patients with IgA nephropathy produce aberrantly glycosylated IgA1. J. Clin. Invest. 118, 629–639 (2008).
Li, G. S., Zhang, H., Lv, J. C., Shen, Y. & Wang, H. Y. Variants of C1GALT1 gene are associated with the genetic susceptibility to IgA nephropathy. Kidney Int. 71, 448–453 (2007).
Pirulli, D. et al. Genetic variant of C1GalT1 contributes to the susceptibility to IgA nephropathy. J. Nephrol. 22, 152–159 (2009).
Zhu, L. et al. Interaction between variants of two glycosyltransferase genes in IgA nephropathy. Kidney Int. 76, 190–198 (2009).
Buck, K. S. et al. B-cell O-galactosyltransferase activity, and expression of O-glycosylation genes in bone marrow in IgA nephropathy. Kidney Int. 73, 1128–1136 (2008).
Smith, A. C., de Wolff, J. F., Molyneux, K., Feehally, J. & Barratt, J. O-glycosylation of serum IgD in IgA nephropathy. J. Am. Soc. Nephrol. 17, 1192–1199 (2006).
Leung, J. C., Tang, S. C., Lam, M. F., Chan, T. M. & Lai, K. N. Charge-dependent binding of polymeric IgA1 to human mesangial cells in IgA nephropathy. Kidney Int. 59, 277–285 (2001).
Leung, J. C., Poon, P. Y. & Lai, K. N. Increased sialylation of polymeric immunoglobulin A1: mechanism of selective glomerular deposition in immunoglobulin A nephropathy? J. Lab. Clin. Med. 133, 152–160 (1999).
Lai, K. N., Ho, R. T., Lai, C. K., Chan, C. H. & Li, P. K. Increase of both circulating Th1 and Th2 T lymphocyte subsets in IgA nephropathy. Clin. Exp. Immunol. 96, 116–121 (1994).
Ebihara, I. et al. Th2 predominance at the single-cell level in patients with IgA nephropathy. Nephrol. Dial. Transplant. 16, 1783–1789 (2001).
Lai, K. N. et al. CD4-positive cells from patients with IgA nephropathy demonstrate increased mRNA of cytokines that induce the IgA switch and differentiation. J. Pathol. 174, 13–22 (1994).
Lai, K. N., Ho, R. T., Leung, J. C., Lai, F. M. & Li, P. K. Increased mRNA encoding for transforming factor-β in CD4+ cells from patients with IgA nephropathy. Kidney Int. 46, 862–868 (1994).
Toyabe, S., Harada, W. & Uchiyama, M. Oligoclonally expanding γδ T lymphocytes induce IgA switching in IgA nephropathy. Clin. Exp. Immunol. 124, 110–117 (2001).
Lai, K. N. et al. Characteristics of polymeric λ-IgA binding to leukocytes in IgA nephropathy. J. Am. Soc. Nephrol. 13, 2309–2319 (2002).
Yu, H. H. et al. Genetics and immunopathogenesis of IgA nephropathy. Clin. Rev. Allergy Immunol. 41, 198–213 (2011).
van den Wall Bake, A. W. et al. The bone marrow as production site of the IgA deposited in the kidneys of patients with IgA nephropathy. Clin. Exp. Immunol. 72, 321–325 (1988).
van den Wall Bake, A. W. et al. Elevated production of polymeric and monomeric IgA1 by the bone marrow in IgA nephropathy. Kidney Int. 35, 1400–1404 (1989).
Harper, S. J. et al. Increased immunoglobulin A and immunoglobulin A1 cells in bone marrow trephine biopsy specimens in immunoglobulin A nephropathy. Am. J. Kidney Dis. 24, 888–892 (1994).
Smith, A. C., Molyneux, K., Feehally, J. & Barratt, J. O-glycosylation of serum IgA1 antibodies against mucosal and systemic antigens in IgA nephropathy. J. Am. Soc. Nephrol. 17, 3520–3528 (2006).
Oortwijn, B. D. et al. A pathogenic role for secretory IgA in IgA nephropathy. Kidney Int. 69, 1131–1138 (2006).
Layward, L., Allen, A. C., Harper, S. J., Hattersley, J. M. & Feehally, J. Increased and prolonged production of specific polymeric IgA after systemic immunization with tetanus toxoid in IgA nephropathy. Clin. Exp. Immunol. 88, 394–398 (1992).
Suzuki, H. et al. Th1 polarization in murine IgA nephropathy directed by bone marrow-derived cells. Kidney Int. 72, 319–327 (2007).
Suzuki, Y. & Tomino, Y. The mucosa-bone-marrow axis in IgA nephropathy. Contrib. Nephrol. 157, 70–79 (2007).
Béné, M. C., Hurault De Ligny, B., Kessler, M. & Faure, G. C. Confirmation of tonsillar anomalies in IgA nephropathy: a multicenter study. Nephron 58, 425–428 (1991).
Kusakari, C. et al. Immunopathological features of palatine tonsil characteristic of IgA nephropathy: IgA1 localization in follicular dendritic cells. Clin. Exp. Immunol. 95, 42–48 (1994).
Harper, S. J. et al. Expression of J chain mRNA in duodenal IgA plasma cells in IgA nephropathy. Kidney Int. 45, 836–844 (1994).
Qin, W. et al. External suppression causes the low expression of the Cosmc gene in IgA nephropathy. Nephrol. Dial. Transplant. 23, 1608–1614 (2008).
Coppo, R. et al. Toll-like receptor 4 expression is increased in circulating mononuclear cells of patients with immunoglobulin A nephropathy. Clin. Exp. Immunol. 159, 73–81 (2010).
Coppo, R., Amore, A., Peruzzi, L., Vergano, L. & Camilla, R. Innate immunity and IgA nephropathy. J. Nephrol. 23, 626–632 (2010).
Suzuki, H. et al. Toll-like receptor 9 affects severity of IgA nephropathy. J. Am. Soc. Nephrol. 19, 2384–2395 (2008).
Monteiro, R. C., Cooper, M. D. & Kubagawa, H. Molecular heterogeneity of Fc alpha receptors detected by receptor-specific monoclonal antibodies. J. Immunol. 148, 1764–1770 (1992).
Morton, H. C., van Egmond, M. & van de Winkel, J. G. Structure and function of human IgA Fc receptors (FcαR). Crit. Rev. Immunol. 16, 423–440 (1996).
Reterink, T. J. et al. Size-dependent effect of IgA on the IgA Fc receptor (CD89). Eur. J. Immunol. 27, 2219–2224 (1997).
Monteiro, R. C., Kubagawa, H. & Cooper, M. D. Cellular distribution, regulation, and biochemical nature of an Fc alpha receptor in humans. J. Exp. Med. 171, 597–613 (1990).
Grossetête, B. et al. Down-regulation of Fcα receptors on blood cells of IgA nephropathy patients: evidence for a negative regulatory role of serum IgA. Kidney Int. 53, 1321–1335 (1998).
Gómez-Guerrero, C., González, E. & Egido, J. Evidence for a specific IgA receptor in rat and human mesangial cells. J. Immunol. 151, 7172–7181 (1993).
Mostov, K. E., Friedlander, M. & Blobel, G. The receptor for transepithelial transport of IgA and IgM contains multiple immunoglobulin-like domains. Nature 308, 37–43 (1984).
Moura, I. C. et al. The glomerular response to IgA deposition in IgA nephropathy. Semin. Nephrol. 28, 88–95 (2008).
Diven, S. C. et al. IgA induced activation of human mesangial cells: independent of FcalphaR1 (CD 89). Kidney Int. 54, 837–847 (1998).
Leung, J. C., Tsang, A. W., Chan, D. T. & Lai, K. N. Absence of CD89, polymeric immunoglobulin receptor, and asialoglycoprotein receptor on human mesangial cells. J. Am. Soc. Nephrol. 11, 241–249 (2000).
Vuong, M. T. et al. Association of soluble CD89 levels with disease progression but not susceptibility in IgA nephropathy. Kidney Int. 78, 1281–1287 (2010).
Stockert, R. J. The asialoglycoprotein receptor: relationships between structure, function, and expression. Physiol. Rev. 75, 591–609 (1995).
Pacifico, F. et al. Differential expression of the asialoglycoprotein receptor in discrete brain areas, in kidney and thyroid. Biochem. Biophys. Res. Commun. 210, 138–144 (1995).
Park, J. H., Cho, E. W., Shin, S. Y., Lee, Y. J. & Kim, K. L. Detection of the asialoglycoprotein receptor on cell lines of extrahepatic origin. Biochem. Biophys. Res. Commun. 244, 304–311 (1998).
Gómez-Guerrero, C., Duque, N. & Egido, J. Mesangial cells possess an asialoglycoprotein receptor with affinity for human immunoglobulin A. J. Am. Soc. Nephrol. 9, 568–576 (1998).
Piskurich, J. F. et al. Transcriptional regulation of the human polymeric immunoglobulin receptor gene by interferon-γ. Mol. Immunol. 34, 75–91 (1997).
Krajci, P. et al. Molecular cloning of the human transmembrane secretory component (poly-Ig receptor) and its mRNA expression in human tissues. Biochem. Biophys. Res. Commun. 158, 783–789 (1989).
Moura, I. C. et al. Identification of the transferrin receptor as a novel immunoglobulin (Ig)A1 receptor and its enhanced expression on mesangial cells in IgA nephropathy. J. Exp. Med. 194, 417–425 (2001).
Moura, I. C. et al. Glycosylation and size of IgA1 are essential for interaction with mesangial transferrin receptor in IgA nephropathy. J. Am. Soc. Nephrol. 15, 622–634 (2004).
McDonald, K. J., Cameron, A. J., Allen, J. M. & Jardine, A. G. Expression of Fc α/μ receptor by human mesangial cells: a candidate receptor for immune complex deposition in IgA nephropathy. Biochem. Biophys. Res. Commun. 290, 438–442 (2002).
Chan, L. Y., Leung, J. C., Tsang, A. W., Tang, S. C. & Lai, K. N. Activation of tubular epithelial cells by mesangial-derived TNF-α: glomerulotubular communication in IgA nephropathy. Kidney Int. 67, 602–612 (2005).
Lai, K. N. et al. Activation of podocytes by mesangial-derived TNF-α: glomerulo-podocytic communication in IgA nephropathy. Am. J. Physiol. Renal Physiol. 294, F945–F955 (2008).
Lavigne, K. A. et al. Familial IgA nephropathy in southeastern Kentucky. Clin. Nephrol. 73, 115–121 (2010).
Sissons, J. G. et al. Isolated glomerulonephritis with mesangial IgA deposits. Br. Med. J. 3, 611–614 (1975).
Johnston, P. A., Brown, J. S., Braumholtz, D. A. & Davison, A. M. Clinico-pathological correlations and long-term follow-up of 253 United Kingdom patients with IgA nephropathy. A report from the MRC Glomerulonephritis Registry. Q. J. Med. 84, 619–627 (1992).
Rambausek, M., Hartz, G., Waldherr, R., Andrassy, K. & Ritz, E. Familial glomerulonephritis. Pediatr. Nephrol. 1, 416–418 (1987).
Schena, F. P., Scivittaro, V. & Ranieri, E. IgA nephropathy: pros and cons for a familial disease. Contrib. Nephrol. 104, 36–45 (1993).
Scolari, F. et al. Familial clustering of IgA nephropathy: further evidence in an Italian population. Am. J. Kidney Dis. 33, 857–865 (1999).
Levy, M. Familial cases of Berger's disease and anaphylactoid purpura: more frequent than previously thought. Am. J. Med. 87, 246–248 (1989).
Tam, K. Y. et al. Macromolecular IgA1 taken from patients with familial IgA nephropathy or their asymptomatic relatives have higher reactivity to mesangial cells in vitro. Kidney Int. 75, 1330–1339 (2009).
Izzi, C. et al. Familial aggregation of primary glomerulonephritis in an Italian population isolate: Valtrompia study. Kidney Int. 69, 1033–1040 (2006).
Julian, B. A. et al. Familial IgA nephropathy. Evidence of an inherited mechanism of disease. N. Engl. J. Med. 312, 202–208 (1985).
Schena, F. P. et al. Increased risk of end-stage renal disease in familial IgA nephropathy. J. Am. Soc. Nephrol. 13, 453–460 (2002).
Tam, K. Y. et al. In vitro enhanced chemotaxis of CD25+ mononuclear cells in patients with familial IgAN through glomerulotubular interactions. Am. J. Physiol. Renal Physiol. 299, F359–F368 (2010).
Julian, B. A., Woodford, S. Y., Baehler, R. W., McMorrow, R. G. & Wyatt, R. J. Familial clustering and immunogenetic aspects of IgA nephropathy. Am. J. Kidney Dis. 12, 366–370 (1988).
Izzi, C. et al. IgA nephropathy: the presence of familial disease does not confer an increased risk for progression. Am. J. Kidney Dis. 47, 761–769 (2006).
Frascá, G. M. et al. Thin basement membrane disease in patients with familial IgA nephropathy. J. Nephrol. 17, 778–785 (2004).
Miyazaki, M. Immunological abnormalities in family members of patients with IgA nephropathy. Jpn J. Med. 29, 469–477 (1990).
Kiryluk, K. et al. Aberrant glycosylation of IgA1 is inherited in both pediatric IgA nephropathy and Henoch-Schönlein purpura nephritis. Kidney Int. 80, 79–87 (2011).
Gharavi, A. G. et al. Aberrant IgA1 glycosylation is inherited in familial and sporadic IgA nephropathy. J. Am. Soc. Nephrol. 19, 1008–1014 (2008).
Paterson, A. D. et al. Genome-wide linkage scan of a large family with IgA nephropathy localizes a novel susceptibility locus to chromosome 2q36. J. Am. Soc. Nephrol. 18, 2408–2415 (2007).
Gharavi, A. G. et al. IgA nephropathy, the most common cause of glomerulonephritis, is linked to 6q22-23. Nat. Genet. 26, 354–357 (2000).
Bisceglia, L. et al. Genetic heterogeneity in Italian families with IgA nephropathy: suggestive linkage for two novel IgA nephropathy loci. Am. J. Hum. Genet. 79, 1130–1134 (2006).
Karnib, H. H. et al. Characterization of a large Lebanese family segregating IgA nephropathy. Nephrol. Dial. Transplant. 22, 772–777 (2007).
Hsu, S. I. et al. Genome-wide linkage scan for familial IgA nephropathy among southeast Asian Chinese: evidence for a suggestive novel susceptibility locus on chromosome 8p23 [abstract]. American Society of Nephrology Renal Week F-PO1405 (2009).
Feehally, J. et al. HLA has strongest association with IgA nephropathy in genome-wide analysis. J. Am. Soc. Nephrol. 21, 1791–1797 (2010).
Gharavi, A. G. et al. Genome-wide association study identifies susceptibility loci for IgA nephropathy. Nat. Genet. 43, 321–327 (2011).
Kiryluk, K. et al. Genetic studies of IgA nephropathy: past, present, and future. Pediatr. Nephrol. 25, 2257–2268 (2010).
Lai, K. N. et al. Polymeric IgA1 from patients with IgA nephropathy upregulates transforming growth factor-β synthesis and signal transduction in human mesangial cells via the renin-angiotensin system. J. Am. Soc. Nephrol. 14, 3127–3137 (2003).
van Kooten, C., Daha, M. R. & Van Es, L. A. Tubular epithelial cells: A critical cell type in the regulation of renal inflammatory processes. Exp. Nephrol. 7, 429–437 (1999).
Lai, K. N., Leung, J. C., Chan, L. Y., Guo, H. & Tang, S. C. Interaction between proximal tubular epithelial cells and infiltrating monocytes/T cells in the proteinuric state. Kidney Int. 71, 526–538 (2007).
Tang, S. et al. Albumin stimulates interleukin-8 expression in proximal tubular epithelial cells in vitro and in vivo. J. Clin. Invest. 111, 515–527 (2003).
Lai, K. N. et al. Podocyte injury induced by mesangial-derived cytokines in IgA nephropathy. Nephrol. Dial. Transplant. 24, 62–72 (2009).
Coppo, R. et al. Aberrantly glycosylated IgA1 induces mesangial cells to produce platelet-activating factor that mediates nephrin loss in cultured podocyte. Kidney Int. 77, 417–427 (2010).
Lai, K. N. et al. Mesangial expression of angiotensin II receptor in IgA nephropathy and its regulation by polymeric IgA1. Kidney Int. 66, 1403–1416 (2004).
Wang, L., Flannery, P. J. & Spurney, R. F. Characterization of angiotensin II-receptor subtypes in podocytes. J. Lab. Clin. Med. 142, 313–321 (2003).
Chan, L. Y., Leung, J. C., Tang, S. C., Choy, C. B. & Lai, K. N. Tubular expression of angiotensin II receptors and their regulation in IgA nephropathy. J. Am. Soc. Nephrol. 16, 2306–2317 (2005).
Chansel, D. et al. Differential regulation of angiotensin II and losartan binding sites in glomeruli and mesangial cells. Am. J. Physiol. 266, F384–F393 (1994).
Makita, N., Iwai, N., Inagami, T. & Badr, K. F. Two distinct pathways in the down-regulation of type-1 angiotension II receptor gene in rat glomerular mesangial cells. Biochem. Biophys. Res. Commun. 185, 142–146 (1992).
Wagner, J., Gehlen, F., Ciechanowicz, A. & Ritz, E. Angiotensin II receptor type 1 gene expression in human glomerulonephritis and diabetes mellitus. J. Am. Soc. Nephrol. 10, 545–551 (1999).
Leung, J. C. et al. Oxidative damages in tubular epithelial cells in IgA nephropathy: role of crosstalk between angiotensin II and aldosterone. J. Transl. Med. 9, 169 (2011).
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The author declares no competing financial interests.
Rights and permissions
About this article
Cite this article
Lai, K. Pathogenesis of IgA nephropathy. Nat Rev Nephrol 8, 275–283 (2012). https://doi.org/10.1038/nrneph.2012.58
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneph.2012.58
This article is cited by
-
Identifying potential biomarkers for the diagnosis and treatment of IgA nephropathy based on bioinformatics analysis
BMC Medical Genomics (2023)
-
Cyclic neutropenia and concomitant IgA nephropathy: a case report
BMC Nephrology (2023)
-
Long-term outcomes of add-on direct renin inhibition in igA nephropathy: a propensity score-matched cohort study
Journal of Nephrology (2023)
-
Gross hematuria can be an impact of severe acute respiratory syndrome coronavirus 2 vaccination on immunoglobulin A nephropathy: a case report
Journal of Medical Case Reports (2022)
-
Increased retinal drusen in IgA glomerulonephritis are further evidence for complement activation in disease pathogenesis
Scientific Reports (2022)