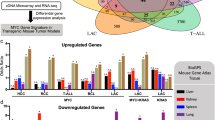
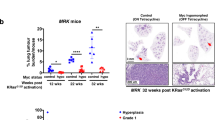
Key Points
Summary
-
The proto-oncogene c-MYC encodes a transcription factor that is implicated in various cellular processes ? cell growth, proliferation, loss of differentiation and apoptosis.
-
c-MYC activates a variety of known target genes as part of a heterodimeric complex with the protein MAX. For example, cyclin D2 and CDK2 are essential for cell-cycle progression, and translation initiation factors eIF4 and eIF2 are important in cell growth.
-
MYC?MAX heterodimers regulate gene activation through chromatin remodelling: association with co-activator TRRAP, which contains HAT activity, leads to acetylation of nucleosomal histones.
-
c-MYC inhibits the differentiation of many cell types. Conversely, MAD/MXI1 transcription factors promote differentiation by antagonizing c-MYC function by forming dimers with MAX. MAD?MAX dimers recruit corepressors (such as SIN3) and HDACs to target DNA, leading to histone deacetylation and subsequent repression of MYC target genes.
-
c-MYC sensitizes cells to a wide range of pro-apoptotic stimuli in vitro via cytochrome c release from mitochondria and subsequent formation of the apoptosome with APAF1 and procaspase-9.
-
Oncogenic c-MYC implies constitutive or deregulated expression of c-MYC that is no longer dependent on external signals and is associated with many human cancers.
-
Conditional transgenic mice, which allow regulated activation of c-MYC in distinct tissues (epidermis and pancreatic islets), have highlighted which cellular response attributed to c-MYC alone (proliferation or apoptosis) predominates in intact tissues in vivo.
-
Regulatable c-MYC transgenic mouse models of cancer have highlighted oncogenic properties of c-MYC in vivo when its apoptotic pathway is blocked, which include induction of angiogenesis, loss of cell?cell contacts and local tissue invasion.
-
Conditional mouse transgenic systems have ascertained when cancer-initiating oncogenic mutations (such as c-MYC and RAS) remain essential for maintenance of the established tumour in vivo. These findings are important for the development of candidate drug molecules that are directed against the oncoprotein.
Abstract
Deregulated expression of c-MYC occurs in a broad range of human cancers and is often associated with poor prognosis, indicating a key role for this oncogene in tumour progression. However, as established human tumours often bear multiple genetic lesions, it is difficult to determine whether c-MYC is instrumental in the initiation/progression of the tumour, or indeed whether inactivating c-MYC would lead to tumour regression. Regulatable transgenic mouse models of oncogenesis have shed light on these issues and provide hope for effective cancer therapies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Change history
08 October 2002
Text accompanying certain citations was incorrectly placed (refs. 53, 55, 60, 84, 87, 92, 104, 119, 127, 135 in pdf and print) The text was moved to Refs. 54,56, 61, 87, 90, 97, 107, 126, 108, 111 respectively.
References
Vennstrom, B., Sheiness, D., Zabielski, J. & Bishop, J. M. Isolation and characterization of c-myc, a cellular homolog of the oncogene (v?myc) of avian myelocytomatosis virus strain 29. J. Virol. 42, 773?779 (1982).
Spencer, C. A. & Groudine M. Control of c?myc regulation in normal and neoplastic cells. Adv Cancer Res. 56, 1?48 (1991).
Dang, C. V. c?Myc target genes involved in cell growth, apoptosis, and metabolism. Mol Cell. Biol. 19, 1?11 (1999).
Nesbit, C. E., Tersak, J. M. & Prochownik, E. V. MYC oncogenes and human neoplastic disease. Oncogene 18, 3004?3016 (1999).
Schlagbauer?Wadl, H. et al. Influence of increased c?Myc expression on the growth characteristics of human melanoma. J. Invest. Dermatol. 112, 332?336 (1999).
Henriksson, M. & Luscher, B. Proteins of the Myc network: essential regulators of cell growth and differentiation. Adv. Cancer Res. 68, 109?182 (1996).
Blackwood, E. M. & Eisenman, R. N. Max: a helix?loop?helix zipper protein that forms a sequence-specific DNA-binding complex with Myc. Science 251, 1211?12127 (1991).
Amati, B. et al. Oncogenic activity of the c-Myc protein requires dimerization with Max. Cell 72, 233?245 (1993).
Amati, B., Littlewood, T. D., Evan, G. I. & Land, H. The c?Myc protein induces cell cycle progression and apoptosis through dimerization with Max. EMBO J. 12, 5083?5087 (1993).References 7?9 identify Max as an essential partner for c-Myc transcriptional activity and physiological functions of c-Myc.
Sakamuro, D. & Prendergast, G. C. New Myc-interacting proteins: a second Myc network emerges. Oncogene 18, 2942?2954 (1999).
Amati, B., Alevizopoulos, K. & Vlach, J. Myc and the cell cycle. Front Biosci. 3, D250?D268 (1998).
Eilers, M. Control of cell proliferation by Myc family genes. Mol. Cells 9, 1?6 (1999).
Amati, B. Integrating Myc and TGF-beta signalling in cell?cycle control. Nature Cell Biol. 3, E112?E113 (2001).
Davis, A. C., Wims, M., Spotts, G. D., Hann, S. R. & Bradley, A. A null c?myc mutation causes lethality before 10.5 days of gestation in homozygotes and reduced fertility in heterozygous female mice. Genes Dev. 7, 671?682 (1993).
Schmid, P., Schulz, W. A. & Hameister, H. Dynamic expression pattern of the myc protooncogene in midgestation mouse embryos. Science 243, 226?229 (1989).
Hirvonen, H. et al. Expression of the myc proto?oncogenes in developing human fetal brain. Oncogene 5, 1787?1797 (1990).
Mateyak, M. K., Obaya, A. J., Adachi, S. & Sedivy, J. M. Phenotypes of c-Myc-deficient rat fibroblasts isolated by targeted homologous recombination. Cell Growth Differ. 8, 1039?1048 (1997).
Rosenwald, I. B., Rhoads, D. B., Callanan, L. D., Isselbacher, K. J. & Schmidt, E. V. Increased expression of eukaryotic translation initiation factors eIF-4E and eIF-2 alpha in response to growth induction by c-myc. Proc. Natl Acad. Sci. USA 90, 6175?6178 (1993).
Jones, R. M. et al. An essential E box in the promoter of the gene encoding the mRNA cap-binding protein (eukaryotic initiation factor 4E) is a target for activation by c-myc. Mol. Cell. Biol. 16, 4754?4764 (1996).
Coller, H. A. et al. Expression analysis with oligonucleotide microarrays reveals that MYC regulates genes involved in growth, cell cycle, signaling, and adhesion. Proc. Natl Acad. Sci. USA 97, 3260?3265 (2000).
Johnston, L. A., Prober, D. A., Edgar, B. A., Eisenman, R. N. & Gallant, P. Drosophila myc regulates cellular growth during development. Cell. 98, 779?790 (1999).
Iritani, B. M. & Eisenman, R. N. c-Myc enhances protein synthesis and cell size during B lymphocyte development. Proc. Natl Acad. Sci. USA 96, 13180?13185 (1999).
Schuhmacher, M. et al. Control of cell growth by c-Myc in the absence of cell division. Curr. Biol. 9, 1255?1258 (1999).
Beier, R. et al. Induction of cyclin E-cdk2 kinase activity, E2F?dependent transcription and cell growth by Myc are genetically separable events. EMBO J. 19, 5813?5823 (2000).References 21?24 . Important indication for role of Myc in regulating the size of cells. Thus, Myc-induced tissue growth might not just reflect cell-cycling.
Steiner, P. et al. Identification of a Myc-dependent step during the formation of active G1 cyclin?cdk complexes. EMBO J. 14, 4814?4826 (1995).
Berns, K., Hijmans, E. M. & Bernards, R. Repression of c-Myc responsive genes in cycling cells causes G1 arrest through reduction of cyclin E/CDK2 kinase activity. Oncogene 15, 1347?1356 (1997).
Bouchard, C. et al. Direct induction of cyclin D2 by Myc contributes to cell cycle progression and sequestration of p27. EMBO J. 18, 5321?5333 (1999).
Hermeking, H. et al. Identification of CDK4 as a target of c?MYC. Proc. Natl Acad. Sci. USA 97, 2229?2234 (2000).
Perez?Roger, I., Kim, S. H., Griffiths, B., Sewing, A. & Land, H. Cyclins D1 and D2 mediate myc-induced proliferation via sequestration of p27(Kip1)and p21(Cip1). EMBO J. 18, 5310?5320 (1999).
O'Hagan, R. C. et al. Myc-enhanced expression of Cul1 promotes ubiquitin-dependent proteolysis and cell cycle progression. Genes Dev. 14, 2185?2191 (2000).
Muller, D. et al. Cdk2-dependent phosphorylation of p27 facilitates its Myc-induced release from cyclin E/cdk2 complexes. Oncogene 15, 2561?2576 (1997).
Staller, P. et al. Repression of p15INK4b expression by Myc through association with Miz?1. Nature Cell Biol. 3, 392?399 (2001).
Herold, S. et al. Negative regulation of the mammalian UV response by Myc through association with Miz-1. Mol. Cell (in the press).
McMahon, S. B., Van Buskirk, H. A., Dugan, K. A., Copeland, T. D. & Cole, M. D. The novel ATM-related protein TRRAP is an essential cofactor for the c-Myc and E2F oncoproteins. Cell 94, 363?374 (1998).Important paper on the mechanics of Myc transcriptional activity.
McMahon, S. B., Wood, M. A. & Cole, M. D. The essential cofactor TRRAP recruits the histone acetyltransferase hGCN5 to c-Myc. Mol. Cell. Biol. 20, 556?562 (2000).
Amati, B., Frank, S. R., Donjerkovic, D. & Taubert, S. Function of the c-Myc oncoprotein in chromatin remodeling and transcription. Biochim. Biophys. Acta 1471, M135?M145 (2001).
Bouchard, C. et al. Regulation of cyclin D2 gene expression by the Myc/Max/Mad network: Myc-dependent TRRAP recruitment and histone acetylation at the cyclin D2 promoter. Genes Dev. 15, 2042?2047 (2001).
Frank, S. R., Schroeder, M., Fernandez, P., Taubert, S. & Amati, B. Binding of c-Myc to chromatin mediates mitogen-induced acetylation of histone H4 and gene activation. Genes Dev. 15, 2069?2082 (2001).
Eberhardy, S. R., D'Cunha, C. A. & Farnham, P. J. Direct examination of histone acetylation on Myc target genes using chromatin immunoprecipitation. J. Biol. Chem. 275, 33798?33805 (2000).
La Rocca, S. A., Crouch, D. H. & Gillespie, D. A. c-Myc inhibits myogenic differentiation and myoD expression by a mechanism which can be dissociated from cell transformation. Oncogene 9, 3499?3508 (1994).
Ryan, K. M. & Birnie, G. D. Cell-cycle progression is not essential for c-Myc to block differentiation. Oncogene 14, 2835?2843 (1997).
Grandori, C., Cowley, S. M., James, L. P. & Eisenman, R. N. The Myc/Max/Mad network and the transcriptional control of cell behavior. Annu. Rev. Cell Dev. Biol. 16, 653?699 (2000).
Foley, K. P. & Eisenman, R. N. Two MAD tails: what the recent knockouts of Mad1 and Mxi1 tell us about the MYC/MAX/MAD network. Biochim. Biophys. Acta 1423, M37?M47 (1999).
Dean, M., Levine, R. A. & Campisi, J. c-myc regulation during retinoic acid-induced differentiation of F9 cells is posttranscriptional and associated with growth arrest. Mol. Cell. Biol. 6, 518?524 (1986).
Dotto, G. P., Gilman, M. Z., Maruyama, M. & Weinberg, R. A. c-Myc and c-fos expression in differentiating mouse primary keratinocytes. EMBO J. 5, 2853?2857 (1986).
Lemaitre, J. M., Buckle, R. S. & Mechali, M. c?Myc in the control of cell proliferation and embryonic development. Adv Cancer Res. 70, 95?144 (1996).
Chin, L. et al. Contrasting roles for Myc and Mad proteins in cellular growth and differentiation. Proc. Natl Acad. Sci. USA 92, 8488?8492 (1995).
Hurlin, P. J. et al. Regulation of Myc and Mad during epidermal differentiation and HPV-associated tumorigenesis. Oncogene 11, 2487?2501 (1995).
Hurlin, P. J. et al. Mad3 and Mad4: novel Max-interacting transcriptional repressors that suppress c-myc dependent transformation and are expressed during neural and epidermal differentiation. EMBO J. 14, 5646?5659 (1995).
Gandarillas, A. & Watt, F. M. c-Myc promotes differentiation of human epidermal stem cells. Genes Dev. 11, 2869?2882 (1997).
Arnold, I. & Watt, F. M. c-Myc activation in transgenic mouse epidermis results in mobilization of stem cells and differentiation of their progeny. Curr. Biol. 11, 558?568 (2001).
Iritani, B. M. & Eisenman, R. N. c-Myc enhances protein synthesis and cell size during B lymphocyte development. Proc. Natl Acad. Sci. USA 96, 13180?13185 (1999).
Ayer, D. E., Lawrence, Q. A. & Eisenman, R. N. Mad?Max transcriptional repression is mediated by ternary complex formation with mammalian homologs of yeast repressor Sin3. Cell 80, 767?776 (1995).
Schreiber?Agus, N. et al. An amino-terminal domain of Mxi1 mediates anti-Myc oncogenic activity and interacts with a homolog of the yeast transcriptional repressor SIN3. Cell 80, 777?786 (1995).Important papers that demonstrate transcriptional repression by Mad-family proteins in association with Sin3 protein.
Evan, G. I. et al. Induction of apoptosis in fibroblasts by c-myc protein. Cell 69, 119?128 (1992).
Askew, D. S., Ashmun, R. A., Simmons, B. C. & Cleveland, J. L. Constitutive c-myc expression in an IL-3-dependent myeloid cell line suppresses cell cycle arrest and accelerates apoptosis. Oncogene 6, 1915?1922 (1991).References 55?56 are the first in vitro demonstrations of Myc-induced apoptosis using conditional and constitutive expression systems, respectively.
Debbas, M. & White, E. Wild-type p53 mediates apoptosis by E1A, which is inhibited by E1B. Genes Dev. 7, 546?554 (1993).
Qin, X. Q., Livingston, D. M., Kaelin, W. G. Jr & Adams, P. D. Deregulated transcription factor E2F-1 expression leads to S-phase entry and p53?mediated apoptosis. Proc. Natl Acad. Sci. USA 91, 10918?10922 (1994).
Shan, B. & Lee, W. H. Deregulated expression of E2F-1 induces S?phase entry and leads to apoptosis. Mol. Cell. Biol. 14, 8166?8173 (1994).
Wu, X. & Levine, A. J. p53 and E2F-1 cooperate to mediate apoptosis. Proc. Natl Acad. Sci. USA 91, 3602?3606 (1994).
Strasser, A., Harris, A. W., Bath, M. L. & Cory, S. Novel primitive lymphoid tumours induced in transgenic mice by cooperation between myc and bcl-2. Nature 348, 331?333 (1990).First in vivo evidence that Bcl2 is tumorigenic and can collaborate with Myc in tumour development.
Adams, J. M. et al. The c-myc oncogene driven by immunoglobulin enhancers induces lymphoid malignancy in transgenic mice. Nature 318, 533?538 (1985).
Blyth, K. et al. Synergy between a human c?myc transgene and p53 null genotype in murine thymic lymphomas: contrasting effects of homozygous and heterozygous p53 loss. Oncogene 10, 1717?1723 (1995).
Elson, A., Deng, C., Campos?Torres, J., Donehower, L. A. & Leder, P. The MMTV/c-myc transgene and p53 null alleles collaborate to induce T-cell lymphomas, but not mammary carcinomas in transgenic mice. Oncogene 11, 181?190 (1995).
Jacobs, J. J. et al. Bmi-1 collaborates with c-Myc in tumorigenesis by inhibiting c-Myc-induced apoptosis via INK4a/ARF. Genes Dev. 13, 2678?2690 (1999).
Eischen, C. M., Weber, J. D., Roussel, M. F., Sherr, C. J. & Cleveland, J. L. Disruption of the ARF?Mdm2?p53 tumor suppressor pathway in Myc-induced lymphomagenesis. Genes Dev. 13, 2658?2669 (1999).
Tanaka, H. et al. E2F1 and c-Myc potentiate apoptosis through inhibition of NF-κB activity that facilitates MnSOD-mediated ROS elimination. Mol. Cell 9, 1017?1029 (2002).
Vafa, O. et al. c-Myc can induce DNA damage, increase reactive oxygen species, and mitigate p53 function: a mechanism for oncogene-induced genetic instability. Mol. Cell 9, 1031?1044 (2002).
Hueber, A. O. et al. Requirement for the CD95 receptor-ligand pathway in c-Myc-induced apoptosis. Science 278, 1305?1309 (1997).
Klefstrom, J. et al. c-Myc induces cellular susceptibility to the cytotoxic action of TNF-alpha. EMBO J. 13, 5442?5450 (1994).
Lutz, W., Fulda, S., Jeremias, I., Debatin, K. M. & Schwab, M. MycN and IFNg cooperate in apoptosis of human neuroblastoma cells. Oncogene 17, 339?346 (1998).
Juin, P., Hueber, A. O., Littlewood, T. & Evan, G. c-Myc-induced sensitization to apoptosis is mediated through cytochrome c release. Genes Dev. 13, 1367?1381 (1999).
Acehan, D. et al. Three-dimensional structure of the apoptosome: implications for assembly, procaspase-9 binding, and activation. Mol. Cell 9, 423?432 (2002).
Martinou, J. C. & Green, D. R. Breaking the mitochondrial barrier. Nature Rev. Mol. Cell Biol. 2, 63?67 (2001).
Soucie, E. L. et al. Myc potentiates apoptosis by stimulating Bax activity at the mitochondria. Mol. Cell. Biol. 21, 4725?4736 (2001).
Juin, P. et al. c-Myc functionally cooperates with Bax to induce apoptosis. Mol. Cell. Biol. 22, 6158?6169 (2002).
Gross, A., Jockel, J., Wei, M. C. & Korsmeyer, S. J. Enforced dimerization of BAX results in its translocation, mitochondrial dysfunction and apoptosis. EMBO J. 17, 3878?3885 (1998).
Khaled, A. R., Kim, K., Hofmeister, R., Muegge, K. & Durum, S. K. Withdrawal of IL-7 induces Bax translocation from cytosol to mitochondria through a rise in intracellular pH. Proc. Natl Acad. Sci. USA 96, 14476?14481 (1999).
Eskes, R., Desagher, S., Antonsson, B. & Martinou, J. C. Bid induces the oligomerization and insertion of Bax into the outer mitochondrial membrane. Mol. Cell. Biol. 20, 929?935 (2000).
Brenner, C. et al. Bcl-2 and Bax regulate the channel activity of the mitochondrial adenine nucleotide translocator. Oncogene 19, 329?336 (2000).
Eskes, R. et al. Bax-induced cytochrome c release from mitochondria is independent of the permeability transition pore but highly dependent on Mg2+ ions. J. Cell Biol. 143, 217?224 (1998).
Green, D. R. & Evan, G. I. A matter of life and death. Cancer Cell 1, 19?30 (2002).
Ionov, Y., Yamamoto, H., Krajewski, S., Reed, J. C. & Perucho, M. Mutational inactivation of the proapoptotic gene BAX confers selective advantage during tumor clonal evolution. Proc. Natl Acad. Sci. USA 97, 10872?10877 (2000).
Kauffmann-Zeh, A. et al. Suppression of c-Myc-induced apoptosis by Ras signalling through PI(3)K and PKB. Nature 385, 544?548 (1997).
Zha, J., Harada, H., Yang, E., Jockel, J. & Korsmeyer, S. J. Serine phosphorylation of death agonist BAD in response to survival factor results in binding to 14-3-3 not BCL?X(L). Cell 87, 619?628 (1996).
Zindy, F. et al. Myc signaling via the ARF tumor suppressor regulates p53-dependent apoptosis and immortalization. Genes Dev. 12, 2424?2433 (1998).
Pelengaris, S., Khan, M. & Evan, G. I. Suppression of Myc-induced apoptosis in beta cells exposes multiple oncogenic properties of Myc and triggers carcinogenic progression. Cell 109, 321?334 (2002).This paper demonstrates the multiple innate oncogenic properties of c-Myc activation in vivo , when c-Myc-induced apoptosis is inhibited.
Kohl, N. E. & Ruley, H. E. Role of c-myc in the transformation of REF52 cells by viral and cellular oncogenes. Oncogene 2, 41?48 (1987).
Mougneau, E., Lemieux, L., Rassoulzadegan, M. & Cuzin, F. Biological activities of v-myc and rearranged c-myc oncogenes in rat fibroblast cells in culture. Proc. Natl Acad. Sci. USA 81, 5758?5762 (1984).
Land, H., Parada, L. F. & Weinberg, R. A. Tumorigenic conversion of primary embryo fibroblasts requires at least two cooperating oncogenes. Nature 304, 596?602 (1983).Seminal early paper on oncogene co-operation.
Land, H., Chen, A. C., Morgenstern, J. P., Parada, L. F. & Weinberg, R. A. Behavior of myc and ras oncogenes in transformation of rat embryo fibroblasts. Mol. Cell. Biol. 6, 1917?1925 (1986).
Lugo, T. G. & Witte, O. N. The BCR?ABL oncogene transforms Rat-1 cells and cooperates with v-myc. Mol. Cell. Biol. 9, 1263?1270 (1989).
Fanidi, A., Harrington, E. A. & Evan, G. I. Cooperative interaction between c-myc and bcl-2 proto?oncogenes. Nature 359, 554?556 (1992).
Reed, J. C. et al. BCL2-mediated tumorigenicity of a human T-lymphoid cell line: synergy with MYC and inhibition by BCL2 antisense. Proc. Natl Acad. Sci. USA 87, 3660?3664 (1990).
Morgenbesser, S. D. & DePinho, R. A. Use of transgenic mice to study myc family gene function in normal mammalian development and in cancer. Semin. Cancer Biol. 5, 21?36 (1994).
Evan, G. I. & Vousden, K. H. Proliferation, cell cycle and apoptosis in cancer. Nature 411, 342?348 (2001).
Pelengaris, S., Littlewood, T., Khan, M., Elia, G. & Evan, G. Reversible activation of c-Myc in skin: induction of a complex neoplastic phenotype by a single oncogenic lesion. Mol. Cell 3, 565?577 (1999).First application of conditional c-Myc expression in keratinocytes in vivo reveals angiogenic and other neoplastic actions of c-Myc in the absence of other co-operating oncogenic lesions in the skin of transgenic mice.
Littlewood, T. D., Hancock, D. C., Danielian, P. S., Parker, M. G. & Evan, G. I. A modified oestrogen receptor ligand-binding domain as an improved switch for the regulation of heterologous proteins. Nucleic Acids Res. 23, 1686?1690 (1995).
Hanahan, D. & Folkman, J. Patterns and emerging mechanisms of the angiogenic switch during tumorigenesis. Cell 86, 353?364 (1996).
Janz, A., Sevignani, C., Kenyon, K., Ngo, C. V. & Thomas?Tikhonenko, A. Activation of the myc oncoprotein leads to increased turnover of thrombospondin-1 mRNA. Nucleic Acids Res. 28, 2268?2275 (2000).
Ngo, C. V. et al. An in vivo function for the transforming Myc protein: elicitation of the angiogenic phenotype. Cell Growth Differ. 11, 201?210 (2000).
Brandvold, K. A., Neiman, P. & Ruddell, A. Angiogenesis is an early event in the generation of myc-induced lymphomas. Oncogene 19, 2780?2785 (2000).
Breit, S. et al. The N-myc oncogene in human neuroblastoma cells: down-regulation of an angiogenesis inhibitor identified as activin A. Cancer Res. 60, 4596?4601 (2000).
Fotsis, T. et al. Down-regulation of endothelial cell growth inhibitors by enhanced MYCN oncogene expression in human neuroblastoma cells. Eur. J. Biochem. 263, 757?764 (1999).
Okajima, E. & Thorgeirsson, U. P. Different regulation of vascular endothelial growth factor expression by the ERK and p38 kinase pathways in v-ras, v-raf, and v-myc transformed cells. Biochem. Biophys. Res. Commun. 270, 108?111 (2000).Interesting observation that even a transient inactivation of c-Myc sufficed to reverse tumorigenic phenotype that is induced under the influence of c-Myc. Furthermore, re-induction of c-Myc induced apoptosis.
Christofori, G., Naik, P. & Hanahan D. A second signal supplied by insulin-like growth factor II in oncogene-induced tumorigenesis. Nature 369, 414?418 (1994).
Felsher, D. W. & Bishop, J. M. Reversible tumorigenesis by MYC in hematopoietic lineages. Mol. Cell. 4, 199?207 (1999).Tumour reversal after suppression of ectopic Myc expression in mouse haematopoietic lineages induced by Myc.
D'Cruz, C. M. et al. c-MYC induces mammary tumorigenesis by means of a preferred pathway involving spontaneous Kras2 mutations. Nature Med. 7, 235?239 (2001).Important paper showing that although the majority of mouse mammary tumours that are induced by c-Myc expression regressed following de-activation of c-Myc, those which had acquired additional Kras2 mutations did not.
Wu, Y. et al. Recurrent allelic deletions at mouse chromosomes 4 and 14 in Myc-induced liver tumors. Oncogene 21, 1518?1526 (2002).
Felsher, D. W. & Bishop, J. M. Transient excess of MYC activity can elicit genomic instability and tumorigenesis. Proc. Natl Acad. Sci. USA 96, 3940?3944 (1999).
Fest, T. et al. c-MYC overexpression in Ba/F3 cells simultaneously elicits genomic instability and apoptosis. Oncogene 21, 2981?2990 (2002).References 68, 110 and 111 show that c-Myc can promote genomic instability/DNA damage, which could contribute to the oncogenic action of Myc.
Jain, M. et al. Sustained loss of a neoplastic phenotype by brief inactivation of MYC. Science 297, 102?104 (2002).
Oster, S. K., Ho, C. S., Soucie, E. L. & Penn, L. Z. The myc oncogene: MarvelouslY Complex. Adv. Cancer Res. 84, 81?154 (2002).
Bernasconi, N. L., Wormhoudt, T. A. & Laird?Offringa, I. A. Post-transcriptional deregulation of myc genes in lung cancer cell lines. Am. J. Respir. Cell Mol. Biol. 23, 560?565 (2000).
Chappell, S. A. et al. A mutation in the c-myc?IRES leads to enhanced internal ribosome entry in multiple myeloma: a novel mechanism of oncogene de-regulation. Oncogene 19, 4437?4440 (2000).
Barone, M. V. & Courtneidge, S. A. Myc but not Fos rescue of PDGF signalling block caused by kinas-inactive Src. Nature 378, 509?512 (1995).
Bowman, T. et al. Stat3-mediated Myc expression is required for Src transformation and PDGF-induced mitogenesis. Proc. Natl Acad. Sci. USA 98, 7319?7324 (2001).
Chiariello, M., Marinissen, M. J. & Gutkind, J. S. Regulation of c-myc expression by PDGF through Rho GTPases. Nature Cell Biol. 3, 580?586 (2001).
Kolligs, F. T. et al. Gamma-catenin is regulated by the APC tumor suppressor and its oncogenic activity is distinct from that of beta-catenin. Genes Dev 14, 1319?1331 (2000).
Huang, H. et al. Gene expression profiling of low-grade diffuse astrocytomas by cDNA arrays. Cancer Res. 60, 6868?6874 (2000).
Watatani, M. et al. Identification of high-risk breast cancer patients from genetic changes of their tumors. Surg. Today 30, 516?522 (2000).
Gregory, M. A. & Hann, S. R. c-Myc proteolysis by the ubiquitin-proteasome pathway: stabilization of c-Myc in Burkitt's lymphoma cells. Mol. Cell. Biol. 20, 2423?2435 (2000).
Salghetti, S. E., Kim, S. Y. & Tansey, W. P. Destruction of Myc by ubiquitin-mediated proteolysis: cancer-associated and transforming mutations stabilize Myc. EMBO J. 18, 717?726 (1999).
Sears, R. et al. Multiple Ras-dependent phosphorylation pathways regulate Myc protein stability. Genes Dev. 14, 2501?2514 (2000).
Hahn, W. C. & Weinberg, R. A. Modelling the molecular circuitry of cancer. Nature Rev. Cancer 2, 331?341 (2002).
Hahn, W. C. et al. Creation of human tumour cells with defined genetic elements. Nature 400, 464?468 (1999).First demonstration that human cells can be directly transformed by introducing defined genes ( SV40, HRAS and TERT).
Dickson, M. A. et al. Human keratinocytes that express hTERT and also bypass a p16(INK4a)-enforced mechanism that limits life span become immortal yet retain normal growth and differentiation characteristics. Mol. Cell. Biol. 20, 1436?1447 (2000).
Morales, C. P. et al. Absence of cancer-associated changes in human fibroblasts immortalized with telomerase. Nature Genet. 21, 115?118 (1999).
Greenberg, R. A. et al. Telomerase reverse transcriptase gene is a direct target of c-Myc but is not functionally equivalent in cellular transformation. Oncogene 18, 1219?1226 (1999).
Wu, K. J. et al. Direct activation of TERT transcription by c-MYC. Nature Genet. 21, 220?224 (1999).
Ewald, D. et al. Time-sensitive reversal of hyperplasia in transgenic mice expressing SV40 T antigen. Science 273, 1384?1386 (1996).
Chin, L. et al. Essential role for oncogenic Ras in tumour maintenance. Nature 400, 468?472 (1999).
Huettner, C. S., Zhang, P., Van Etten, R. A. & Tenen, D. G. Reversibility of acute B-cell leukaemia induced by BCR?ABL1. Nature Genet. 24, 57?60 (2000).
Jackson, E. L. et al. Analysis of lung tumor initiation and progression using conditional expression of oncogenic K-ras. Genes Dev. 15, 3243?3248 (2001).
Fisher, G. H. et al. Induction and apoptotic regression of lung adenocarcinomas by regulation of a K-Ras transgene in the presence and absence of tumor suppressor genes. Genes Dev. 15, 3249?3262 (2001).
Author information
Authors and Affiliations
Corresponding author
Related links
Related links
DATABASES
Cancer.gov
GenBank
FlyBase
InterPro
LocusLink
Glossary
- SURVIVAL FACTORS
-
Extracellular or intracellular molecules that block apoptosis. Extracellular molecules such as IGF1 mediate cell survival via its receptor, by activating the RAS?PI3K?AKT pathway.
- PARACRINE
-
Secretion of factors from cells within a given tissue that influence neighbouring cells.
- BCL2 FAMILY
-
A family of proteins that comprises both pro-apoptotic and anti-apoptotic members, the balance of which determines whether or not a cell commits apoptosis by regulating the release of cytochrome c from mitochondria.
- BH-123 AND 'BH3-ONLY' PROTEINS
-
Pro-apoptotic members of the BCL2 family. BH-123 proteins share three of the BCL2 homology (BH) domains with the anti-apoptotic proteins, whereas 'BH3-only' proteins possess only the BH3 domain that is necessary for their pro-apoptotic activity.
- CDKN2A
-
The CDKN2A locus encodes two structurally distinct proteins (INK4A and ARF) by reading a shared second exon in different translational reading frames. INK4A can induce cell-cycle arrest by binding to and preventing the ability of CDK4 and CDK6 to phosphorylate and functionally impair the tumour suppressor retinoblastoma. ARF prevents the degradation and inactivation of the tumour suppressor p53 by binding to MDM2, which might lead to cell-cycle arrest.
- ANOIKIS
-
A form of apoptotic cell death that occurs after detachment from the extracellular matrix or from neighbouring cells. Loss of cell adhesion results in the loss of signalling pathways that mediate cell survival (such as via integrin or cadherin proteins).
- RIP1?TAg2 MODEL
-
A transgenic mouse strain that expresses the simian virus T antigen (TAg) under the rat insulin II promoter (RIP) in the pancreatic islet β-cells. Carcinomas develop in the pancreatic islets cells and progress through characteristic stages.
- LASER-CAPTURE MICRODISSECTION
-
An innovative technique that provides a simple and rapid method for the contamination-free selection of single cells or groups of cells from a wide variety of samples for DNA, RNA and protein analysis.
- RT-PCR
-
(Real-time reverse transcription polymerase chain reaction). PCR using a fluorescent probe that contains a 5′-fluorescent label and 3′-quencher dye. As reverse transcription occurs, the 5′-reporter dye is released and the level of fluorescence emission can be measured as the reaction is proceeding. This technique can be used throughout a patient's treatment programme to monitor the proportion of leukaemic cells that still carry a translocation-induced fusion mRNA.
- TOPONOMICS
-
Definition of protein expression/concentration in the context of cellular location ? for example, on a histological tissue section.
Rights and permissions
About this article
Cite this article
Pelengaris, S., Khan, M. & Evan, G. c-MYC: more than just a matter of life and death. Nat Rev Cancer 2, 764–776 (2002). https://doi.org/10.1038/nrc904
Issue Date:
DOI: https://doi.org/10.1038/nrc904
This article is cited by
-
WWOX promotes osteosarcoma development via upregulation of Myc
Cell Death & Disease (2024)
-
Molecular complexity of diffuse large B-cell lymphoma: a molecular perspective and therapeutic implications
Journal of Applied Genetics (2024)
-
LINC00478-derived novel cytoplasmic lncRNA LacRNA stabilizes PHB2 and suppresses breast cancer metastasis via repressing MYC targets
Journal of Translational Medicine (2023)
-
Visualizing single-molecule conformational transition and binding dynamics of intrinsically disordered proteins
Nature Communications (2023)
-
Metabolism-based targeting of MYC via MPC-SOD2 axis-mediated oxidation promotes cellular differentiation in group 3 medulloblastoma
Nature Communications (2023)