Key Points
-
The selective oestrogen receptor modulator tamoxifen is the most widely used antioestrogen for the treatment of hormone-dependent breast cancer.
-
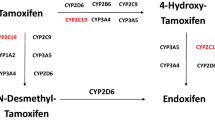
Hepatic, drug-metabolizing cytochrome P450s (CYPs) catalyse the oxidation of tamoxifen to several metabolites. The metabolites, endoxifen and 4-hydroxytamoxifen, have greater binding affinities for oestrogen receptors and suppress cell proliferation more effectively than tamoxifen does. Plasma concentrations of endoxifen are considerably higher than those of 4-hydroxytamoxifen, suggesting that endoxifen is the main pharmacologically active species of tamoxifen in vivo. The conversion of tamoxifen to endoxifen is predominantly catalysed by CYP2D6.
-
Many polymorphisms in CYP2D6 have been identified. In Caucasian populations, 6–10% of people inherit two alleles containing polymorphisms and/or a gene deletion, leading to no protein expression or the expression of a protein with no CYP2D6 enzymatic activity. These individuals have impaired metabolism of CYP2D6 substrates and are called poor metabolizers of CYP2D6. Some drugs, such as the antidepressants fluoxetine or paroxetine, are potent inhibitors of CYP2D6 and can confer a poor metabolizer phenotype on individuals with normal CYP2D6 activity.
-
The findings of pharmacokinetic studies indicate that the conversion of endoxifen is reduced in poor metabolizers of CYP2D6, either by genotype or by co-prescribed fluoxetine or paroxetine, which are commonly prescribed to manage hot flashes.
-
Recent data suggest that poor metabolizers of CYP2D6 do not derive as much benefit from tamoxifen therapy as other patients do; however, some studies have yielded conflicting results.
-
The analysis of CYP2D6 genotype might represent an early example of a pharmacogenetic tool for optimizing breast cancer therapy; however, the findings of larger, well-designed studies that support the current data are necessary before a change in clinical practice is advocated.
Abstract
Tamoxifen is the most widely used anti-oestrogen for the treatment of hormone-dependent breast cancer. The pharmacological activity of tamoxifen is dependent on its conversion by the hepatic drug-metabolizing enzyme cytochrome P450 2D6 (CYP2D6) to its abundant metabolite, endoxifen. Patients with reduced CYP2D6 activity, as a result of either their genotype or induction by the co-administration of drugs that inhibit CYP2D6 function, produce little endoxifen and seem to derive inferior therapeutic benefit from tamoxifen. Here we review the existing data that relate CYP2D6 genotypes to response to tamoxifen and discuss whether the analysis of the CYP2D6 genotype might be an early example of a pharmacogenetic tool for optimizing breast cancer therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Evans, W. E. & McLeod, H. L. Pharmacogenomics—drug disposition, drug targets, and side effects. N. Engl. J. Med. 348, 538–549 (2003).
Evans, W. E. & Relling, M. V. Moving towards individualized medicine with pharmacogenomics. Nature 429, 464–468 (2004).
Parkin, D. M., Bray, F., Ferlay, J. & Pisani, P. Global cancer statistics, 2002. CA Cancer J. Clin. 55, 74–108 (2005).
Wu, X. et al. The tamoxifen metabolite, endoxifen, is a potent antiestrogen that targets estrogen receptor α for degradation in breast cancer cells. Cancer Res. 69, 1722–1727 (2009).
Ingelman-Sundberg, M. Genetic polymorphisms of cytochrome P450 2D6 (CYP2D6): clinical consequences, evolutionary aspects and functional diversity. Pharmacogenomics J. 5, 6–13 (2005).
Jin, Y. et al. CYP2D6 genotype, antidepressant use, and tamoxifen metabolism during adjuvant breast cancer treatment. J. Natl Cancer Inst. 97, 30–39 (2005). This study found a gene dose effect of CYP2D6 genotype on steady-state endoxifen plasma concentrations: PM/PM patients had the lowest concentrations, EM/EM patients had the highest and EM/PM patients had intermediate concentrations. It also showed that co-administered antidepressants that are potent inhibitors of CYP2D6 reduced the formation of endoxifen.
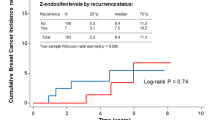
Goetz, M. P. et al. The impact of cytochrome P450 2D6 metabolism in women receiving adjuvant tamoxifen. Breast Cancer Res. Treat. 101, 113–121 (2007). This study, which used the same cohort as in reference 38, found that postmenopausal women who received tamoxifen for early breast cancer with decreased metabolism of CYP2D6, by genotype or enzyme inhibition, had a shorter time to recurrence and worse relapse-free survival than patients with EM/EM genotypes who were not taking a CYP2D6 inhibitor.
Dahlman-Wright, K. et al. International Union of Pharmacology. LXIV. Estrogen receptors. Pharmacol. Rev. 58, 773–781 (2006).
Osborne, C. K., Shou, J., Massarweh, S. & Schiff, R. Crosstalk between estrogen receptor and growth factor receptor pathways as a cause for endocrine therapy resistance in breast cancer. Clin. Cancer Res. 11, S865–S870 (2005).
Massarweh, S. & Schiff, R. Unraveling the mechanisms of endocrine resistance in breast cancer: new therapeutic opportunities. Clin. Cancer Res. 13, 1950–1954 (2007).
Osborne, C. K. Tamoxifen in the treatment of breast cancer. N. Engl. J. Med. 339, 1609–1618 (1998).
Dehal, S. S. & Kupfer, D. CYP2D6 catalyzes tamoxifen 4-hydroxylation in human liver. Cancer Res. 57, 3402–3406 (1997).
Desta, Z., Ward, B. A., Soukhova, N. V. & Flockhart, D. A. Comprehensive evaluation of tamoxifen sequential biotransformation by the human cytochrome P450 system in vitro: prominent roles for CYP3A and CYP2D6. J. Pharmacol. Exp. Ther. 310, 1062–1075 (2004). This comprehensive in vitro study used human liver microsomes and expressed human CYPs to characterize the oxidation of tamoxifen, showing that the conversion of tamoxifen to its secondary metabolite endoxifen occurs through two primary metabolites and is predominantly mediated by CYP2D6, CYP3A4 and CYP3A5.
Stearns, V. et al. Active tamoxifen metabolite plasma concentrations after coadministration of tamoxifen and the selective serotonin reuptake inhibitor paroxetine. J. Natl Cancer Inst. 95, 1758–1764 (2003).
Johnson, M. D. et al. Pharmacological characterization of 4-hydroxy-N-desmethyl tamoxifen, a novel active metabolite of tamoxifen. Breast Cancer Res. Treat. 85, 151–159 (2004). This study showed that endoxifen has essentially equivalent activity to the potent metabolite 4-hydroxytamoxifen with respect to binding to ERs, ability to inhibit oestrogen-stimulated breast cancer cell proliferation and the regulation of oestrogen-responsive genes.
Lim, Y. C., Desta, Z., Flockhart, D. A. & Skaar, T. C. Endoxifen (4-hydroxy-N-desmethyl-tamoxifen) has anti-estrogenic effects in breast cancer cells with potency similar to 4-hydroxy-tamoxifen. Cancer Chemother. Pharmacol. 55, 471–478 (2005).
Lim, Y. C. et al. Endoxifen, a secondary metabolite of tamoxifen, and 4-OH-tamoxifen induce similar changes in global gene expression patterns in MCF-7 breast cancer cells. J. Pharmacol. Exp. Ther. 318, 503–512 (2006). This study investigated the effects of endoxifen and 4-hydroxytamoxifen on gene expression in MCF-7 cells, showing that endoxifen and 4-hydroxytamoxifen have similar effects on global gene expression patterns and that most of the affected genes are oestrogen-regulated genes.
Borges, S. et al. Quantitative effect of CYP2D6 genotype and inhibitors on tamoxifen metabolism: implication for optimization of breast cancer treatment. Clin. Pharmacol. Ther. 80, 61–74 (2006).
Nelson, D. R. et al. Comparison of cytochrome P450 (CYP) genes from the mouse and human genomes, including nomenclature recommendations for genes, pseudogenes and alternative-splice variants. Pharmacogenetics 14, 1–18 (2004).
Ingelman-Sundberg, M., Sim, S. C., Gomez, A. & Rodriguez-Antona, C. Influence of cytochrome P450 polymorphisms on drug therapies: pharmacogenetic, pharmacoepigenetic and clinical aspects. Pharmacol. Ther. 116, 496–526 (2007).
Bradford, L. D. CYP2D6 allele frequency in European Caucasians, Asians, Africans and their descendants. Pharmacogenomics 3, 229–243 (2002).
Gaedigk, A. et al. Cytochrome P4502D6 (CYP2D6) gene locus heterogeneity: characterization of gene duplication events. Clin. Pharmacol. Ther. 81, 242–251 (2007).
Kusama, M., Hisaka, A., Hibino, Y. & Suzuki, H. The influence of Asian specific variant, CYP2D6*10 on in vitro formation of endoxifen, an active metabolite of tamoxifen. Abstr. 21st Japanese Society for the Study of Xenobiotics Annual Meeting 21, 229 (2006).
Lim, H. S. et al. Clinical implications of CYP2D6 genotypes predictive of tamoxifen pharmacokinetics in metastatic breast cancer. J. Clin. Oncol. 25, 3837–3845 (2007).
Kimmick, G. G., Lovato, J., McQuellon, R., Robinson, E. & Muss, H. B. Randomized, double-blind, placebo-controlled, crossover study of sertraline (Zoloft) for the treatment of hot flashes in women with early stage breast cancer taking tamoxifen. Breast J. 12, 114–122 (2006).
Loprinzi, C. L. et al. Venlafaxine in management of hot flashes in survivors of breast cancer: a randomised controlled trial. Lancet 356, 2059–2063 (2000).
Hemeryck, A. & Belpaire, F. M. Selective serotonin reuptake inhibitors and cytochrome P-450 mediated drug–drug interactions: an update. Curr. Drug Metab. 3, 13–37 (2002).
Schroth, W. et al. Breast cancer treatment outcome with adjuvant tamoxifen relative to patient CYP2D6 and CYP2C19 genotypes. J. Clin. Oncol. 25, 5187–5193 (2007).
Sim, S. C. et al. A common novel CYP2C19 gene variant causes ultrarapid drug metabolism relevant for the drug response to proton pump inhibitors and antidepressants. Clin. Pharmacol. Ther. 79, 103–113 (2006).
Desta, Z., Zhao, X., Shin, J. G. & Flockhart, D. A. Clinical significance of the cytochrome P450 2C19 genetic polymorphism. Clin. Pharmacokinet. 41, 913–958 (2002).
Okishiro, M. et al. Genetic polymorphisms of CYP2D6 10 and CYP2C19 2, 3 are not associated with prognosis, endometrial thickness, or bone mineral density in Japanese breast cancer patients treated with adjuvant tamoxifen. Cancer 115, 952–961 (2009).
Ogura, K. et al. Quaternary ammonium-linked glucuronidation of trans-4-hydroxytamoxifen, an active metabolite of tamoxifen, by human liver microsomes and UDP-glucuronosyltransferase 1A4. Biochem. Pharmacol. 71, 1358–1369 (2006).
Nishiyama, T. et al. Reverse geometrical selectivity in glucuronidation and sulfation of cis- and trans-4-hydroxytamoxifens by human liver UDP-glucuronosyltransferases and sulfotransferases. Biochem. Pharmacol. 63, 1817–1830 (2002).
Gjerde, J. et al. Effects of CYP2D6 and SULT1A1 genotypes including SULT1A1 gene copy number on tamoxifen metabolism. Ann. Oncol. 19, 56–61 (2008).
Blevins-Primeau, A. S. et al. Functional significance of UDP-glucuronosyltransferase variants in the metabolism of active tamoxifen metabolites. Cancer Res. 69, 1892–1900 (2009).
Spina, E., Santoro, V. & D'Arrigo, C. Clinically relevant pharmacokinetic drug interactions with second-generation antidepressants: an update. Clin. Ther. 30, 1206–1227 (2008).
Newton, D. J., Wang, R. W. & Lu, A. Y. Cytochrome P450 inhibitors. Evaluation of specificities in the in vitro metabolism of therapeutic agents by human liver microsomes. Drug Metab. Dispos. 23, 154–158 (1995).
Goetz, M. P. et al. Pharmacogenetics of tamoxifen biotransformation is associated with clinical outcomes of efficacy and hot flashes. J. Clin. Oncol. 23, 9312–9318 (2005). This study showed that postmenopausal women who received tamoxifen for early breast cancer with PM/PM genotypes tended to have a higher risk of disease relapse and a lower incidence of hot flashes than patients with EM/EM or EM/PM genotypes.
Bijl, M. J. et al. The CYP2D6*4 polymorphism affects breast cancer survival in tamoxifen users. Breast Cancer Res. Treat. 3 Feb 2009 (doi: 10.1007/s10549-008-0272-2).
Kiyotani, K. et al. Impact of CYP2D6*10 on recurrence-free survival in breast cancer patients receiving adjuvant tamoxifen therapy. Cancer Sci. 99, 995–999 (2008).
Xu, Y. & Villalona-Calero, M. A. Irinotecan: mechanisms of tumor resistance and novel strategies for modulating its activity. Ann. Oncol. 13, 1841–1851 (2002).
Bonanni, B. et al. Polymorphism in the CYP2D6 tamoxifen-metabolizing gene influences clinical effect but not hot flashes: data from the Italian Tamoxifen Trial. J. Clin. Oncol. 24, 3708–3709; author reply 3709 (2006).
Nowell, S. A. et al. Association of genetic variation in tamoxifen-metabolizing enzymes with overall survival and recurrence of disease in breast cancer patients. Breast Cancer Res. Treat. 91, 249–258 (2005).
Wegman, P. et al. Genotype of metabolic enzymes and the benefit of tamoxifen in postmenopausal breast cancer patients. Breast Cancer Res. 7, R284–R290 (2005).
Wegman, P. et al. Genetic variants of CYP3A5, CYP2D6, SULT1A1, UGT2B15 and tamoxifen response in postmenopausal patients with breast cancer. Breast Cancer Res. 9, R7 (2007).
Bratherton, D. G. et al. A comparison of two doses of tamoxifen (Nolvadex) in postmenopausal women with advanced breast cancer: 10 mg bd versus 20 mg bd. Br. J. Cancer 50, 199–205 (1984).
Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. Lancet 365, 1687–1717 (2005).
Newman, W. G. et al. Impaired tamoxifen metabolism reduces survival in familial breast cancer patients. Clin. Cancer Res. 14, 5913–5918 (2008).
Lash, T. L. et al. Tamoxifen's protection against breast cancer recurrence is not reduced by concurrent use of the SSRI citalopram. Br. J. Cancer 99, 616–621 (2008).
Shin, H. R., Jung, K. W., Won, Y. J., Park, J. G. & 139 KCCR-affiliated Hospitals. 2002 annual report of the Korean Central Cancer Registry: based on registered data from 139 hospitals. Cancer Res. Treat 36, 103–114 (2004).
Yu, K. D. et al. Development and trends of surgical modalities for breast cancer in China: a review of 16-year data. Ann. Surg. Oncol. 14, 2502–2509 (2007).
Parton, M. & Smith, I. E. Controversies in the management of patients with breast cancer: adjuvant endocrine therapy in premenopausal women. J. Clin. Oncol. 26, 745–752 (2008).
Furlanut, M. et al. Tamoxifen and its main metabolites serum and tissue concentrations in breast cancer women. Ther. Drug Monit. 29, 349–352 (2007).
Gaedigk, A. et al. The CYP2D6 activity score: translating genotype information into a qualitative measure of phenotype. Clin. Pharmacol. Ther. 83, 234–242 (2008).
Parte, P. & Kupfer, D. Oxidation of tamoxifen by human flavin-containing monooxygenase (FMO) 1 and FMO3 to tamoxifen-N-oxide and its novel reduction back to tamoxifen by human cytochromes P450 and hemoglobin. Drug Metab. Dispos. 33, 1446–1452 (2005).
Early Breast Cancer Trialists' Collaborative Group. Tamoxifen for early breast cancer: an overview of the randomised trials. Lancet 351, 1451–1467 (1998).
Ingle, J. N. Pharmacogenomics of tamoxifen and aromatase inhibitors. Cancer 112, 695–699 (2008).
Clarke, R., Leonessa, F., Welch, J. N. & Skaar, T. C. Cellular and molecular pharmacology of antiestrogen action and resistance. Pharmacol. Rev. 53, 25–71 (2001).
Perez, E. A. Safety profiles of tamoxifen and the aromatase inhibitors in adjuvant therapy of hormone-responsive early breast cancer. Ann. Oncol. 18 (Suppl. 8), 26–35 (2007).
Ntukidem, N. I. et al. Estrogen receptor genotypes, menopausal status, and the lipid effects of tamoxifen. Clin. Pharmacol. Ther. 83, 702–710 (2008).
Morello, K. C., Wurz, G. T. & DeGregorio, M. W. Pharmacokinetics of selective estrogen receptor modulators. Clin. Pharmacokinet. 42, 361–372 (2003).
Coombes, R. C. et al. A randomized trial of exemestane after two to three years of tamoxifen therapy in postmenopausal women with primary breast cancer. N. Engl. J. Med. 350, 1081–1092 (2004).
Howell, A. et al. Results of the ATAC (arimidex, tamoxifen, alone or in combination) trial after completion of 5 years' adjuvant treatment for breast cancer. Lancet 365, 60–62 (2005).
Kaufmann, M. et al. Improved overall survival in postmenopausal women with early breast cancer after anastrozole initiated after treatment with tamoxifen compared with continued tamoxifen: the ARNO 95 Study. J. Clin. Oncol. 25, 2664–2670 (2007).
Thurlimann, B. et al. A comparison of letrozole and tamoxifen in postmenopausal women with early breast cancer. N. Engl. J. Med. 353, 2747–2757 (2005).
Sachse, C., Brockmoller, J., Bauer, S. & Roots, I. Cytochrome P450 2D6 variants in a Caucasian population: allele frequencies and phenotypic consequences. Am. J. Hum. Genet. 60, 284–295 (1997).
Gough, A. C. et al. Identification of the primary gene defect at the cytochrome P450 CYP2D locus. Nature 347, 773–776 (1990).
Cai, W. M., Chen, B. & Zhang, W. X. Frequency of CYP2D6*10 and *14 alleles and their influence on the metabolic activity of CYP2D6 in a healthy Chinese population. Clin. Pharmacol. Ther. 81, 95–98 (2007).
Lee, S. Y. et al. Sequence-based CYP2D6 genotyping in the Korean population. Ther. Drug Monit. 28, 382–387 (2006).
Shen, H. et al. Comparative metabolic capabilities and inhibitory profiles of CYP2D6.1, CYP2D6.10, and CYP2D6.17. Drug Metab. Dispos. 35, 1292–1300 (2007).
Wennerholm, A. et al. Characterization of the CYP2D6*29 allele commonly present in a black Tanzanian population causing reduced catalytic activity. Pharmacogenetics 11, 417–427 (2001).
Punglia, R. S., Burstein, H. J., Winer, E. P. & Weeks, J. C. Pharmacogenomic variation of CYP2D6 and the choice of optimal adjuvant endocrine therapy for postmenopausal breast cancer: a modeling analysis. J. Natl Cancer Inst. 100, 642–648 (2008).
Gnant, M. et al. Endocrine therapy plus zoledronic acid in premenopausal breast cancer. N. Engl. J. Med. 360, 679–691 (2009).
Mahoney, M. C., Bevers, T., Linos, E. & Willett, W. C. Opportunities and strategies for breast cancer prevention through risk reduction. CA Cancer J. Clin. 58, 347–371 (2008).
Xu, Y. et al. Association between CYP2D6 *10 genotype and survival of breast cancer patients receiving tamoxifen treatment. Ann. Oncol. 19, 1423–1429 (2008).
Acknowledgements
This work was supported by the US National Institutes of Health Pharmacogenetics Research Network (Grant U01 GM63340).
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary information S1
Genes encoding many drug metabolizing enzymes and the nuclear factor NR1I2 have been evaluated in relation to steady-state tamoxifen and metabolite pharmacokinetics and clinical outcomes in women with breast cancer treated with adjuvant tamoxifen therapy. (PDF 184 kb)
Supplementary information S2
Summary of clinical studies that have evaluated the association between CYP2D6 genotype and response to tamoxifen therapy (PDF 264 kb)
Related links
Glossary
- Adjuvant treatment
-
Refers to additional treatment, which is usually given after surgery, in cases in which all detectable disease has been removed but there remains a statistical risk of relapse.
- Haem-thiolate enzymes
-
The collective name given to a class of haemoproteins in which a thiolate group (typically from a cysteine residue) is the axial ligand of haem iron.
- Hazard ratio
-
In a survival analysis, this is the effect of an explanatory variable on the hazard risk of an event.
- Allozyme
-
Variant forms of an enzyme that are encoded by different alleles at the same locus.
Rights and permissions
About this article
Cite this article
Hoskins, J., Carey, L. & McLeod, H. CYP2D6 and tamoxifen: DNA matters in breast cancer. Nat Rev Cancer 9, 576–586 (2009). https://doi.org/10.1038/nrc2683
Issue Date:
DOI: https://doi.org/10.1038/nrc2683