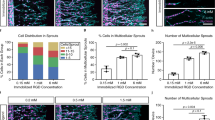
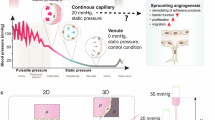
Abstract
Seeding a monolayer of primary human endothelial cells on the surface of a polymerized three-dimensional collagen matrix in the presence of pro-angiogenic stimuli allows manipulation and analysis of rapid sprouting responses. This protocol is useful for elucidating incompletely defined intracellular mechanisms downstream of pro-angiogenic factors that regulate sprout formation and initiation, and can also be used to test the efficacy of pro-and anti-angiogenic compounds. We present protocols to culture endothelial cells, prepare three-dimensional collagen matrices and quantify and image rapid endothelial sprouting responses (24 h). This protocol can be carried out using either type I or type II collagen matrices with primary endothelial cells isolated from macrovascular and microvascular sources of varying species.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Egeblad, M. et al. Visualizing stromal cell dynamics in different tumor microenvironments by spinning disk confocal microscopy. Dis. Model. Mech. 1, 155–167 (2008); discussion 165.
Kedrin, D. et al. Intravital imaging of metastatic behavior through a mammary imaging window. Nat. Methods 5, 1019–1021 (2008).
Kedrin, D., Wyckoff, J., Sahai, E., Condeelis, J. & Segall, J.E. Imaging tumor cell movement in vivo. Current Protocols in Cell Biology Unit 19.7 (2007).
Wolf, K. et al. Multi-step pericellular proteolysis controls the transition from individual to collective cancer cell invasion. Nat. Cell Biol. 9, 893–904 (2007).
Weiss, P. & Garber, B. Shape and movement of mesenchyme cells as functions of the physical structure of the medium: contributions to a quantitative morphology. Proc. Natl. Acad. Sci. USA 38, 264–280 (1952).
Harrison, R.G. The outgrowth of the nerve fiber as a mode of protoplasmic movement. J. Exp. Zool. 9, 787–846 (1910).
Ehrmann, R.L. & Gey, G.O. The growth of cells on a transparent gel of reconstituted rat-tail collagen. J. Natl. Cancer Inst. 16, 1375–1403 (1956).
Elsdale, T. & Bard, J. Collagen substrata for studies on cell behavior. J. Cell Biol. 54, 626–637 (1972).
Davis, G.E. & Camarillo, C.W. An alpha 2 beta 1 integrin-dependent pinocytic mechanism involving intracellular vacuole formation and coalescence regulates capillary lumen and tube formation in three-dimensional collagen matrix. Exp. Cell Res. 224, 39–51 (1996).
Aplin, A.C., Fogel, E., Zorzi, P. & Nicosia, R.F. The aortic ring model of angiogenesis. Methods Enzymol. 443, 119–136 (2008).
Liu, Y. & Senger, D.R. Matrix-specific activation of Src and Rho initiates capillary morphogenesis of endothelial cells. FASEB. J. 18, 457–468 (2004).
Sweeney, S.M. et al. Angiogenesis in collagen I requires alpha2beta1 ligation of a GFP*GER sequence and possibly p38 MAPK activation and focal adhesion disassembly. J. Biol. Chem. 278, 30516–30524 (2003).
Bayless, K.J. & Davis, G.E. Sphingosine-1-phosphate markedly induces matrix metalloproteinase and integrin-dependent human endothelial cell invasion and lumen formation in three-dimensional collagen and fibrin matrices. Biochem. Biophys. Res. Commun. 312, 903–913 (2003).
Koh, W., Stratman, A.N., Sacharidou, A. & Davis, G.E. In vitro three dimensional collagen matrix models of endothelial lumen formation during vasculogenesis and angiogenesis. Methods Enzymol. 443, 83–101 (2008).
Nakatsu, M.N. et al. Angiogenic sprouting and capillary lumen formation modeled by human umbilical vein endothelial cells (HUVEC) in fibrin gels: the role of fibroblasts and angiopoietin-1. Microvasc. Res. 66, 102–112 (2003).
Nicosia, R.F. & Madri, J.A. The microvascular extracellular matrix. Developmental changes during angiogenesis in the aortic ring-plasma clot model. Am. J. Pathol. 128, 78–90 (1987).
Nicosia, R.F., McCormick, J.F. & Bielunas, J. The formation of endothelial webs and channels in plasma clot culture. Scan. Electron Microsc. (Pt 2): 793–799 (1984).
Nicosia, R.F., Tchao, R. & Leighton, J. Histotypic angiogenesis in vitro: light microscopic, ultrastructural, and radioautographic studies. In vitro 18, 538–549 (1982).
Nehls, V. & Drenckhahn, D. A novel, microcarrier-based in vitro assay for rapid and reliable quantification of three-dimensional cell migration and angiogenesis. Microvasc. Res. 50, 311–322 (1995).
Nehls, V., Schuchardt, E. & Drenckhahn, D. The effect of fibroblasts, vascular smooth muscle cells, and pericytes on sprout formation of endothelial cells in a fibrin gel angiogenesis system. Microvasc. Res. 48, 349–363 (1994).
Bell, S.E. et al. Differential gene expression during capillary morphogenesis in 3D collagen matrices: regulated expression of genes involved in basement membrane matrix assembly, cell cycle progression, cellular differentiation and G-protein signaling. J. Cell Sci. 114 (Pt 15): 2755–2773 (2001).
Su, S.C., Mendoza, E.A., Kwak, H.I. & Bayless, K.J. Molecular profile of endothelial invasion of three-dimensional collagen matrices: insights into angiogenic sprout induction in wound healing. Am. J. Physiol. 295, C1215–C1229 (2008).
Hahn, C.N. et al. Expression profiling reveals functionally important genes and coordinately regulated signaling pathway genes during in vitro angiogenesis. Physiol. Genomics 22, 57–69 (2005).
Kahn, J. et al. Gene expression profiling in an in vitro model of angiogenesis. Am. J. Pathol. 156, 1887–1900 (2000).
Koh, W., Mahan, R.D. & Davis, G.E. Cdc42- and Rac1-mediated endothelial lumen formation requires Pak2, Pak4 and Par3, and PKC-dependent signaling. J. Cell Sci. 121 (Pt 7): 989–1001 (2008).
Koh, W. et al. Formation of endothelial lumens requires a coordinated PKC{epsilon}-, Src-, Pak- and Raf-kinase-dependent signaling cascade downstream of Cdc42 activation. J. Cell Sci. 122 (Pt 11): 1812–1822 (2009).
Saunders, W.B. et al. Coregulation of vascular tube stabilization by endothelial cell TIMP-2 and pericyte TIMP-3. J. Cell Biol. 175, 179–191 (2006).
Stratman, A.N. et al. Endothelial cell lumen and vascular guidance tunnel formation requires MT1-MMP-dependent proteolysis in 3D collagen matrices. Blood 114, 237–247 (2009).
Sainson, R.C. et al. Cell-autonomous notch signaling regulates endothelial cell branching and proliferation during vascular tubulogenesis. FASEB. J. 19, 1027–1029 (2005).
Sainson, R.C. et al. TNF primes endothelial cells for angiogenic sprouting by inducing a tip cell phenotype. Blood 111, 4997–5007 (2008).
Bayless, K.J. & Davis, G.E. The Cdc42 and Rac1 GTPases are required for capillary lumen formation in three-dimensional extracellular matrices. J. Cell Sci. 115 (Pt 6): 1123–1136 (2002).
Kamei, M., Saunders, W.B., Bayless, K.J., Dye, L., Davis, G.E. & Weinstein, B.M. Endothelial tubes assemble from intracellular vacuoles in vivo. Nature 442, 453–456 (2006).
Kang, H., Bayless, K.J. & Kaunas, R. Fluid shear stress modulates endothelial cell invasion into three-dimensional collagen matrices. Am. J. Physiol. Heart Circ. Physiol. 295, H2087–H2097 (2008).
Lee, P.F., Yeh, A.T. & Bayless, K.J. Nonlinear optical microscopy reveals invading endothelial cells anisotropically alter three-dimensional collagen matrices. Exp. Cell Res. 315, 396–410 (2009).
Emonard, H. et al. Interactions between fibroblasts and a reconstituted basement membrane matrix. J. Invest. Dermatol. 89, 156–163 (1987).
Bikfalvi, A., Cramer, E.M., Tenza, D. & Tobelem, G. Phenotypic modulations of human umbilical vein endothelial cells and human dermal fibroblasts using two angiogenic assays. Biol. Cell 72, 275–278 (1991).
Vukicevic, S., Kleinman, H.K., Luyten, F.P., Roberts, A.B., Roche, N.S. & Reddi, A.H. Identification of multiple active growth factors in basement membrane matrigel suggests caution in interpretation of cellular activity related to extracellular matrix components. Exp. Cell Res. 202, 1–8 (1992).
Hoying, J.B., Boswell, C.A. & Williams, S.K. Angiogenic potential of microvessel fragments established in three-dimensional collagen gels. In Vitro Cell Dev. Biol. Anim. 32, 409–419 (1996).
Hughes, C.C. Endothelial-stromal interactions in angiogenesis. Curr. Opin. Hematol. 15, 204–209 (2008).
Bornstein, M.B. Reconstituted rattail collagen used as substrate for tissue cultures on coverslips in Maximow slides and roller tubes. Lab. Invest. 7, 134–137 (1958).
Rajan, N., Habermehl, J., Cote, M.F., Doillon, C.J. & Mantovani, D. Preparation of ready-to-use, storable and reconstituted type I collagen from rat tail tendon for tissue engineering applications. Nat. Protoc. 1, 2753–2758 (2006).
Dunlap, K.A. et al. Progesterone and placentation increase secreted phosphoprotein one (SPP1 or osteopontin) in uterine glands and stroma for histotrophic and hematotrophic support of ovine pregnancy. Biol. Reprod. 79, 983–990 (2008).
Engelse, M.A., Laurens, N., Verloop, R.E., Koolwijk, P. & van Hinsbergh, V.W. Differential gene expression analysis of tubule forming and non-tubule forming endothelial cells: CDC42GAP as a counter-regulator in tubule formation. Angiogenesis 11, 153–167 (2008).
Fisher, K.E., Pop, A., Koh, W., Anthis, N.J., Saunders, W.B. & Davis, G.E. Tumor cell invasion of collagen matrices requires coordinate lipid agonist-induced G-protein and membrane-type matrix metalloproteinase-1-dependent signaling. Mol. Cancer 5, 69 (2006).
Kwak, H.I., Mendoza, E.A. & Bayless, K.J. ADAM17 co-purifies with TIMP-3 and modulates endothelial invasion responses in three-dimensional collagen matrices. Matrix Biol. 28, 470–479 (2009).
Kim, A., Lakshman, N. & Petroll, W.M. Quantitative assessment of local collagen matrix remodeling in 3-D culture: the role of Rho kinase. Exp. Cell Res. 312, 3683–3692 (2006).
Maciag, T., Cerundolo, J., Ilsley, S., Kelley, P.R. & Forand, R. An endothelial cell growth factor from bovine hypothalamus: identification and partial characterization. Proc. Natl. Acad. Sci. USA 76, 5674–5678 (1979).
Davis, G.E., Black, S.M. & Bayless, K.J. Capillary morphogenesis during human endothelial cell invasion of three-dimensional collagen matrices. In Vitro Cell Dev. Biol. Anim. 36, 513–519 (2000).
Iruela-Arispe, M.L. & Davis, G.E. Cellular and molecular mechanisms of vascular lumen formation. Dev. Cell 16, 222–231 (2009).
Acknowledgements
This work was supported by American Heart Association Scientist Development Grant 053020N (K.J.B.) and start up funds from Texas A&M HSC. We thank Dr. George Davis (University of Missouri-Columbia) for reading the manuscript and Pei-Hsing Chen and Chih-Po Su for assistance with illustrations in Figure 1.
Author information
Authors and Affiliations
Contributions
K.J.B., S.-C.S. and H.-I.K. designed the study; K.J.B. and H.-I.K. carried out the experiments; K.J.B., S.-C.S and H.-I.K collected and analyzed data; K.J.B., S.-C.S. and H.-I.K wrote the manuscript.
Corresponding author
Supplementary information
Supplementary Movie 1
Differential interference contrast (DIC) analyses of invading endothelial cells stably expressing green fluorescent protein with time. Experiments were established as described in Box 2. The movie is compiled from 87 frames captured at 5 minute intervals. Movies were initiated at 12 hours of invasion and were allowed to proceed until 19.25 hours. Retracting processes, extending processes, individual endothelial cells, and large vacuoles are indicated in Figure 7. (WMV 2162 kb)
Supplementary Movie 2
Time-lapse analyses of invading endothelial cells stably expressing green fluorescent protein (GFP) using epifluorescence with time. Movie is compiled from 87 frames captured at 5 minute intervals. Movies were initiated at 12 hours of invasion and were allowed to proceed until 19.25 hours. Retracting processes, extending processes, individual endothelial cells, and vacuoles are indicated in Figure 7. (WMV 701 kb)
Supplementary Movie 3
Combination of Differential interference contrast (DIC; movie 1) and epifluorescence (movie 2) analyses of invading endothelial cells stably expressing green fluorescent protein with time. (WMV 2154 kb)
Rights and permissions
About this article
Cite this article
Bayless, K., Kwak, HI. & Su, SC. Investigating endothelial invasion and sprouting behavior in three-dimensional collagen matrices. Nat Protoc 4, 1888–1898 (2009). https://doi.org/10.1038/nprot.2009.221
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2009.221
This article is cited by
-
Integrins and their potential roles in mammalian pregnancy
Journal of Animal Science and Biotechnology (2023)
-
FGF-2 promotes angiogenesis through a SRSF1/SRSF3/SRPK1-dependent axis that controls VEGFR1 splicing in endothelial cells
BMC Biology (2021)
-
FOXK2 transcriptionally activating VEGFA induces apatinib resistance in anaplastic thyroid cancer through VEGFA/VEGFR1 pathway
Oncogene (2021)
-
NIK promotes metabolic adaptation of glioblastoma cells to bioenergetic stress
Cell Death & Disease (2021)
-
T-Plastin reinforces membrane protrusions to bridge matrix gaps during cell migration
Nature Communications (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.