Abstract
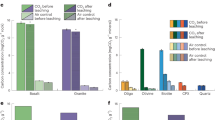
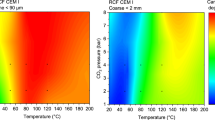
A simple method of CO~2~ capture is by using the calcium looping cycle. The calcium looping cycle uses CaCO~3~ as a CO~2~ carrier, via the reversible reaction CaO(s) + CO~2~(g) = CaCO~3~(s), to extract CO2 from the exhaust stream and provide a pure stream of CO~2~ suitable for sequestration. A problem associated with the technology is that the capacity of the sorbent to absorb CO~2~ reduces significantly with the number of cycles of carbonation and calcination. The energy penalty of the cycle is considerably increased by cycling unreacted sorbent: hydration of unreactive sorbent has emerged as a promising strategy of reducing this penalty by regenerating the reactivity of exhausted sorbent.A small atmospheric pressure fluidised bed reactor has been built and tested, that allows repeated cycling between two temperatures up to 1000 °C. Work presented here focuses on the effects of variation of the calcination temperature before hydration. Hydration has been found to more than double the reactivity of a spent sorbent cycled under the mildest conditions studied (calcination temperature of 840 °C). However, as calcination temperature is increased the observed reactivation decreases until little reactivation is observed for the sorbent cycled at 950 °C. The primary reason for this appears to be a substantial increase in friability of particles, with reactivity normalised for mass losses appearing similar independent of cycling temperature.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Blamey, J., Fennell, P. & Dugwell, D. Reactivation of Limestone-Derived Sorbents using Hydration: Preliminary Results From a Fluidised Bed. Nat Prec (2008). https://doi.org/10.1038/npre.2008.2630.1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/npre.2008.2630.1