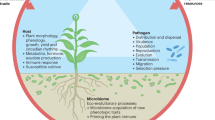
Abstract
The United Nations declared 2016 as the International Year of Pulses (grain legumes) under the banner ‘nutritious seeds for a sustainable future’. A second green revolution is required to ensure food and nutritional security in the face of global climate change. Grain legumes provide an unparalleled solution to this problem because of their inherent capacity for symbiotic atmospheric nitrogen fixation, which provides economically sustainable advantages for farming. In addition, a legume-rich diet has health benefits for humans and livestock alike. However, grain legumes form only a minor part of most current human diets, and legume crops are greatly under-used. Food security and soil fertility could be significantly improved by greater grain legume usage and increased improvement of a range of grain legumes. The current lack of coordinated focus on grain legumes has compromised human health, nutritional security and sustainable food production.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Peoples, M. B., Herridge, D. F. & Ladha, J. K. Biological nitrogen-fixation — an efficient source of nitrogen for sustainable agricultural production. Plant Soil 174, 3–28 (1995).
Siddique, K. H. M., Johansen, C. & Turner, N. C. Innovations in agronomy for food legumes. Agron. Sustain. Dev. 32, 45–64 (2012).
Tilman, D., Cassman, K. G., Matson, P. A., Naylor, R. & Polasky, S. Agricultural sustainability and intensive production practices. Nature 418, 671–677 (2002).
Barton, L., Thamo, T., Engelbrecht, D. & Biswas, W. K. Does growing grain legumes or applying lime cost effectively lower greenhouse gas emissions from wheat production in a semi-arid climate?. J. Clean Prod. 83, 194–203 (2014).
Lassaletta, L., Billen, G., Grizzetti, B., Anglade, J. & Garnier, J. 50 year trends in nitrogen use efficiency of world cropping systems: the relationship between yield and nitrogen input to cropland. Environ. Res. Lett. 9, 105011 (2014).
Herridge, D. F., Peoples, M. B. & Boddey, R. M. Global inputs of biological nitrogen fixation in agricultural systems. Plant Soil 311, 1–18 (2008).
Reeves, T. G., Thomas, G. & Ramsay, G. Save and Grow in Practice: Maize, Rice, Wheat. A Guide to Sustainable Cereal Production (FAO UN, 2016).
Malik, A. I. et al. Relay sowing of lentil (Lens culinaris subsp. culinaris) to intensify rice-based cropping. J. Agr. Sci. 154, 850–857 (2015).
Huang, L.-F. et al. Plant–soil feedbacks and soil sickness: from mechanisms to application in agriculture. J. Chem. Ecol. 39, 232–242 (2013).
Nayyar, A., Hamel, C., Lafond, G., Gossen, B. D. & Hanson, K. Soil microbial quality associated with yield reduction in continuous-pea. Appl. Soil Ecol. 43, 115–121 (2009).
Johansen, C. et al. Integrated crop management of chickpea in environments of Bangladesh prone to botrytis grey mould. Field Crops Res. 108, 238–249 (2008).
Myers, S. S. et al. Increasing CO2 threatens human nutrition. Nature 510, 139–142 (2014).
Mourtzinis, S. et al. Climate-induced reduction in US-wide soybean yields underpinned by region- and in-season-specific responses. Nature Plants 1, 14026 (2015).
Beebe, S. et al. in Crop Adaptation to Climate Change ( eds Yadav, S. S., Redden, R. J., Hatfield, J. L., Lotze-Campen, H. & Hall, A. E. ) Ch. 16 (Wiley-Blackwell, 2011).
Ramirez-Cabral, N. Y. Z., Kumar, L. & Taylor, S. Crop niche modeling projects major shifts in common bean growing areas. Agr. Forest Meteorol. 218–219 102–113 (2016).
Peltonen-Sainio, P., Jauhiainen, L., Hakala, K. & Ojanen, H. Climate change and prolongation of growing season: changes in regional potential for field crop production in Finland. Agr. Food Sci. 18, 171–190 (2009).
Andrews, M. & Hodge, S. in Climate Change and Management of Cool Season Grain Legume Crops (eds Yadav, S. S., McNeil, D. L., Redden, R. & Patil, S. A. ) Ch. 1 (2010).
Bhatia, V. S., Singh, P., Wani, S. P., Kesava Rao, A. V. R. & Srinivas, K. Yield Gap Analysis of Soybean, Groundnut, Pigeonpea and Chickpea in India Using Simulation Modeling. Report No. 31 (International Crops Research Institute for the Semi-Arid Tropics, 2006).
Anderson, W., Johansen, C. & Siddique, K. H. M. Addressing the yield gap in rainfed crops: a review. Agron. Sustain. Dev. 36, 18 (2016).
Oldroyd, G. E. D. Speak, friend, and enter: signalling systems that promote beneficial symbiotic associations in plants. Nature Rev. Microbiol. 11, 252–263 (2013).
Li, Y. Z., Green, L. S., Holtzapffel, R., Day, D. A. & Bergersen, F. J. Supply of O2 regulates demand for O2 and uptake of malate by N2-fixing bacteroids from soybean nodules. Microbiology 147, 663–670 (2001).
Reid, D. E., Ferguson, B. J. & Gresshoff, P. M. Inoculation- and nitrate-induced CLE peptides of soybean control NARK-dependent nodule formation. Mol. Plant Microbe In. 24, 606–618 (2011).
Li, X. X., Sorensen, P., Li, F. C., Petersen, S. O. & Olesen, J. E. Quantifying biological nitrogen fixation of different catch crops, and residual effects of roots and tops on nitrogen uptake in barley using in-situ N-15 labelling. Plant Soil 395, 273–287 (2015).
Bouguyon, E. et al. Multiple mechanisms of nitrate sensing by Arabidopsis nitrate transceptor NRT1.1. Nature Plants 1, 15015 (2015).
Leran, S. et al. Nitrate sensing and uptake in Arabidopsis are enhanced by ABI2, a phosphatase inactivated by the stress hormone abscisic acid. Sci. Signal. 8, ra43 (2015).
Parniske, M. Arbuscular mycorrhiza: the mother of plant root endosymbioses. Nature Rev. Microbiol. 6, 763–775 (2008).
Howieson, J. & Ballard, R. Optimising the legume symbiosis in stressful and competitive environments within southern Australia — some contemporary thoughts. Soil Biol. Biochem. 36, 1261–1273 (2004).
Puppo, A. et al. Legume nodule senescence: roles for redox and hormone signalling in the orchestration of the natural aging process. New Phytol. 165, 683–701 (2005).
Cernay, C., Ben-Ari, T., Pelzer, E., Meynard, J. M. & Makowski, D. Estimating variability in grain legume yields across Europe and the Americas. Sci. Rep. 5, 11171 (2015).
Farooq, M. et al. Drought stress in grain legumes during reproduction and grain filling. J. Agron. Crop Sci.http://dx.doi.org/10.1111/jac.12169 (2016).
Daryanto, S., Wang, L. & Jacinthe, P. A. Global synthesis of drought effects on food legume production. PLoS ONE 10, e0127401 (2015).
Sinclair, T. R., Messina, C. D., Beatty, A. & Samples, M. Assessment across the United States of the benefits of altered soybean drought traits. Agron. J. 102, 475–482 (2010).
Devi, M. J., Sinclair, T. R., Beebe, S. E. & Rao, I. M. Comparison of common bean (Phaseolus vulgaris L.) genotypes for nitrogen fixation tolerance to soil drying. Plant Soil 364, 29–37 (2013).
Sinclair, T. R. et al. Variation among cowpea genotypes in sensitivity of transpiration rate and symbiotic nitrogen fixation to soil drying. Crop Sci. 55, 2270–2275 (2015).
Fairbanks, D. J. & Rytting, B. Mendelian controversies: a botanical and historical review. Am. J. Bot. 88, 737–752 (2001).
Abbo, S., Berger, J. & Turner, N. C. Evolution of cultivated chickpea: four bottlenecks limit diversity and constrain adaptation. Funct. Plant Biol. 30, 1081–1087 (2003).
Gizlice, Z., Carter, T. E. & Burton, J. W. Genetic base for north-American public soybean cultivars released between 1947 and 1988. Crop Sci. 34, 1143–1151 (1994).
Cowling, W. A. The challenge of breeding for increased grain production in an era of global climate change and genomics. World Agri. 5, 50–55 (2015).
Varshney, R. K. et al. Analytical and decision support tools for genomics-assisted breeding. Trends Plant Sci.http://dx.doi.org/10.1016/j.tplants.2015.10.018 (2015).
Varshney, R. K. Exciting journey of 10 years from genomes to fields and markets: some success stories of genomics-assisted breeding in chickpea, pigeonpea and groundnut. Plant Sci. 242, 98–107 (2016).
McCouch, S. et al. Feeding the future. Nature 499, 23–24 (2013).
GENESYS (accessed 21 April 2016); http://www.genesys-pgr.org
https://www.gene.affrc.go.jp/databases_en.php (accessed 21 April 2016).
http://www.nbpgr.ernet.in/Research_Projects/Base_Collection_in_NGB.aspx (accessed 21 April 2016).
Yang, H. et al. Sequencing consolidates molecular markers with plant breeding practice. Theor. Appl. Genet. 128, 779–795 (2015).
Cowling, W. A. Sustainable plant breeding. Plant Breed. 132, 1–9 (2013).
Goddard, M. E. & Hayes, B. J. Mapping genes for complex traits in domestic animals and their use in breeding programmes. Nature Rev. Genet. 10, 381–391 (2009).
Hayes, B. J., Visscher, P. M. & Goddard, M. E. Increased accuracy of artificial selection by using the realized relationship matrix. Genet. Res. 91, 47–60 (2009).
Cowling, W. A. et al. Using the animal model to accelerate response to selection in a self-pollinating crop. Genes Genom. Genet. 5, 1419–1428 (2015).
Granier, C. & Vile, D. Phenotyping and beyond: modelling the relationships between traits. Curr. Opin. Plant Biol. 18, 96–102 (2014).
Fahlgren, N., Gehan, M. A. & Baxter, I. Lights, camera, action: high-throughput plant phenotyping is ready for a close-up. Curr. Opin. Plant Biol. 24, 93–99 (2015).
Ghanem, M. E., Marrou, H. & Sinclair, T. R. Physiological phenotyping of plants for crop improvement. Trends Plant Sci. 20, 139–144 (2015).
Steele, W. M., Allen, D. J. & Summerfield, R. J. in Grain Legume Crops (eds Summerfield, R. J. & Roberts, E. H. ) 520–583 (Collins, 1985).
Beebe, S. E., Rao, I. M., Blair, M. W. & Acosta-Gallegos, J. A. Phenotyping common beans for adaptation to drought. Front. Physiol. 4, 35 (2013).
Nepolo, E., Takundwa, M., Chimwamurombe, P. M., Cullis, C. A. & Kunert, K. A review of geographical distribution of marama bean (Tylosema esculentum (Burchell) Schreiber) and genetic diversity in the Namibian germplasm. Afr. J. Biotechnol. 8, 2088–2093 (2009).
Kouris-Blazos, A. & Belski, R. Health benefits of legumes and pulses with a focus on Australian sweet lupins. Asia Pac. J. Clin. Nutr. 25, 1–17 (2016).
Darmadi-Blackberry, I. et al. Legumes: the most important dietary predictor of survival in older people of different ethnicities. Asia Pac. J. Clin. Nutr. 13, 217–220 (2004).
Chang, W. C. et al. A bean-free diet increases the risk of all-cause mortality among Taiwanese women: the role of the metabolic syndrome. Public Health Nutr. 15, 663–672 (2012).
Trichopoulou, A. et al. Diet and overall survival in elderly people. Brit. Med. J. 311, 1457–1460 (1995).
Kushi, L. H., Meyer, K. A. & Jacobs, D. R. Jr. Cereals, legumes, and chronic disease risk reduction: evidence from epidemiologic studies. Am. J. Clin. Nutr. 70, 451–458 (1999).
Flight, I. & Clifton, P. Cereal grains and legumes in the prevention of coronary heart disease and stroke: a review of the literature. Eur. J. Clin. Nutr. 60, 1145–1159 (2006).
Hashemi, Z. et al. Cooking enhances beneficial effects of pea seed coat consumption on glucose tolerance, incretin, and pancreatic hormones in high-fat-diet-fed rats. Appl. Physiol. Nutr. Metab. 40, 323–333 (2015).
Nothlings, U. et al. Intake of vegetables, legumes, and fruit, and risk for all-cause, cardiovascular, and cancer mortality in a European diabetic population. J. Nutr. 138, 775–781 (2008).
Sievenpiper, J. L. et al. Effect of non-oil-seed pulses on glycaemic control: a systematic review and meta-analysis of randomised controlled experimental trials in people with and without diabetes. Diabetologia 52, 1479–1495 (2009).
Lee, Y. P. et al. Lupin-enriched bread increases satiety and reduces energy intake acutely. Am. J. Clin. Nutr. 84, 975–980 (2006).
Lee, Y. P. et al. Effects of lupin kernel flour-enriched bread on blood pressure: a controlled intervention study. Am. J. Clin. Nutr. 89, 766–772 (2009).
Belski, R. et al. Effects of lupin-enriched foods on body composition and cardiovascular disease risk factors: a 12-month randomized controlled weight loss trial. Int. J. Obesity 35, 810–819 (2011).
Dove, E. R. et al. Lupin and soya reduce glycaemia acutely in type 2 diabetes. Brit. J. Nutr. 106, 1045–1051 (2011).
Singh, J. & Basu, P. S. Non-nutritive bioactive compounds in pulses and their impact on human health: an overview. Food Nutr. Sci. 3, 1664–1672 (2012).
Singh, B. B., Ajeigbe, H. A., Tarawali, S. A., Fernandez-Rivera, S. & Abubakar, M. Improving the production and utilization of cowpea as food and fodder. Field Crop Res. 84, 169–177 (2003).
Lambot, C. in Challenges and Opportunities for Enhancing Sustainable Cowpea Production (eds Fatokun, C. A., Tarawali, S. A., Singh, B. B., Kormawa, P. M. & Tamò, M. ) 367–375 (International Institute of Tropical Agriculture, 2002).
Nadaraja, D., Weintraub, S. T., Hakala, K. W., Sherman, N. E. & Starcher, B. Isolation and partial sequence of a Kunitz-type elastase specific inhibitor from marama bean (Tylosema esculentum). J. Enzym. Inhib. Med. Chem. 25, 377–382 (2010).
Sulieman, S. & Tran, L. S. P. Phosphorus homeostasis in legume nodules as an adaptive strategy to phosphorus deficiency. Plant Sci. 239, 36–43 (2015).
http://www.most.gov.cn/ztzl/kjzykfgx/kjzygjjctjpt/kjzyptml/201407/t20140716_114275.htm (accessed 21 April 2016).
Stoutjesdijk, P. Plant genetic resources for food and agriculture: second national report Technical Report 13.11 (ABARES, 2013)
Bennett, M. D. & Leitch I. J. Plant DNA C-values database (v.6.0, December 2012; accessed 21 April 2016); http://www.kew.org/cvalues/
Simmonds, N. W. & Smartt, J. Principles of Crop Improvement 2nd edn (Blackwell Science, 1999).
Singh, R. J. et al. Landmark research in legumes. Genome 50, 525–537 (2007).
FAOSTAT (accessed 5 January 2016); http://faostat3.fao.org/compare/E
South, A. rworldmap: a new R package for mapping global data. The R Journal 3/1 35–43 (2011).
Neuwirth, E. RColorBrewer: ColorBrewer Palettes. R package version 1.1–2 (2014); http://CRAN.R-project.org/package=RColorBrewer
Lavin, M., Herendeen, P. S. & Wojciechowski, M. F. Evolutionary rates analysis of Leguminosae implicates a rapid diversification of lineages during the tertiary. Syst. Biol. 54, 575–594 (2005).
Gepts, P. et al. Legumes as a model plant family. Genomics for food and feed report of the cross-legume advances through genomics conference. Plant Physiol. 137, 1228–1235 (2005).
Katoh, K. & Standley, D. M. MAFFT Multiple sequence alignment software version 7: improvements in performance and usability. Mol. Biol. Evol. 30, 772–780 (2013).
Lemey, P., Rambaut, A., Drummond, A. J. & Suchard, M. A. Bayesian phylogeography finds its roots. PLoS Comput. Biol. 5, e1000520 (2009).
Erisman, J. W., Sutton, M. A., Galloway, J., Klimont, Z. & Winiwarter, W. How a century of ammonia synthesis changed the world. Nature Geosci. 1, 636–639 (2008).
Heffer, P. & Prud'homme, M. Fertilizer Outlook 2015–2019 (2015); www.fertilizer.org
US Energy Information Administration Total Primary Energy Consumption (2015); www.eia.gov
Storkey, J. et al. Grassland biodiversity bounces back from long-term nitrogen addition. Nature 528, 401–404 (2015).
UNEP and WHRC Reactive Nitrogen in the Environment: Too Much or Too Little of a Good Thing? (The United Nations Environment Program, 2007); www.unep.org/pdf/dtie/Reactive_Nitrogen.pdf
Tilman, D., Cassman, K. G., Matson, P. A., Naylor, R. & Polasky, S. Agricultural sustainability and intensive production practices. Nature 418, 671–677 (2002).
Ladha, J. K. et al. Global nitrogen budgets in cereals: a 50-year assessment for maize, rice, and wheat production systems. Sci. Rep. 6, 19355 (2016).
Acknowledgements
We thank the World University Network (WUN) and research collaboration awards (UWA and the University of Leeds) for financial support. C.H.F. thanks the Biotechnology and Biological Sciences Research Council (BBSRC UK, BB/M009130/1) and the European Union (KBBE-2012-6-311840; ECOSEED) for financial support. J.W.C. thanks BBSRC UK and Wherry and Sons (UK) for an industrial CASE studentship (BB/K501839/1). H.-M.L. was supported by the Hong Kong RGC Collaborative Research Fund (CUHK3/CRF/11G), the Lo Kwee-Seong Biomedical Research Fund and Lee Hysan Foundation. K.-M. Fung, Q. Wang and L. K.-W. Siu of The Chinese University of Hong Kong assisted in the production of Fig. 3, Table 1 and the associated webpage (http://legumecrops.wildsoydb.org/). We thank H. Upadhyaya for the images shown in Fig. 4. T.A.M. and J.M.H. thank the Western Australian Government, Department of Industry and Resources for financial support. The authors thank Bodhi's Bakery, Fremantle, Western Australia, for baking the bread and biscuits and Belmar Foods, Balcatta, Western Australia, for manufacturing and providing the pasta. B.N.K., H.B. and T.D.C. are supported by the Australian Research Council (ARC), ITRH—Legumes for Sustainable Agriculture (IH140100013). M.J.C. and C.H.F. thank the ARC (DP150103211) for financial support. A.J.M. is supported by grant funding (BB/JJ004553/1 and BB/L010305/1) from the BBSRC and the John Innes Foundation.
Author information
Authors and Affiliations
Contributions
C.H.F. co-created the network, discussed the idea, organized the content, wrote the abstract, introduction and ‘Conclusions and perspectives’ sections, and edited the final article before submission. H.-M.L. discussed and contributed to the content, prepared Fig. 3, Table 1 and the associated webpage (http://legumecrops.wildsoydb.org/). R.V. provided information concerning genetics and breeding. H.T.N. discussed and contributed to the content and coordinated the genetic and breeding topics including the figures and Table 1. K.H.M.S. co-created the network, discussed the idea, contributed to various sections and figures, and edited the final version. T.D.C. discussed the content, contributed to the section on sustainable agriculture, contributed edits to several sections, and gave suggestions on figures. W.A.C. wrote parts of the text, contributed citations and edited Table 1 and figures. H.B. discussed the content, prepared Fig. 1 and Fig. 2, and contributed to the section ‘Sustainable agriculture’. T.A.M. discussed the idea and contributed to the section ‘Nutrition and health’. J.M.H. contributed to the section ‘Nutrition and health’. J.W.C. produced Fig. 4 and finalized the references. A.J.M. contributed to the section ‘Symbiotic nitrogen fixation’. K.K. and J.V. contributed to the section ‘Bringing in orphans’. C.C. organized the section ‘Bringing in orphans’ and reviewed the final version of the section. J.A.O. discussed the idea and provided information for the section ‘Nutrition and health’. M.L.W. contributed to the section ‘Nutrition and health’. Y.L. discussed the idea and contributed to the section ‘Sustainable agriculture’. H.S., K.S. and J.Y. discussed the idea and helped to edit the content before submission. N.F. contributed to the section ‘Mitigating climate change’. B.N.K. contributed to sections focussed on legume nitrogen fixation and helped to edit the content before submission. F.-L.W. produced Table 1 and the associated webpage (http://legumecrops.wildsoydb.org/). B.V. contributed to the section and citations on abiotic stress, and Fig. 6. M.C. co-created the network, prepared Box 1, discussed the idea and edited the figures before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Foyer, C., Lam, HM., Nguyen, H. et al. Neglecting legumes has compromised human health and sustainable food production. Nature Plants 2, 16112 (2016). https://doi.org/10.1038/nplants.2016.112
Published:
DOI: https://doi.org/10.1038/nplants.2016.112