Abstract
As cannabis use increases, physicians need to be familiar with the effects of both cannabis and tobacco on the lungs. However, there have been very few long-term studies of cannabis smoking, mostly due to legality issues and the confounding effects of tobacco. It was previously thought that cannabis and tobacco had similar long-term effects as both cause chronic bronchitis. However, recent large studies have shown that, instead of reducing forced expiratory volume in 1 s and forced vital capacity (FVC), marijuana smoking is associated with increased FVC. The cause of this is unclear, but acute bronchodilator and anti-inflammatory effects of cannabis may be relevant. Bullous lung disease, barotrauma and cannabis smoking have been recognised in case reports and small series. More work is needed to address the effects of cannabis on lung function, imaging and histological changes.
Similar content being viewed by others
Introduction
As most people are aware, marijuana is the most widely used illicit drug in the world.1 Cannabis is the second most smoked substance, after tobacco. In the past few years, recreational use of cannabis (‘weed’, ‘dope’, ‘grass’, ‘spliff’, ‘toke’, ‘hash’, ‘hemp’, ‘bud’, ‘ganja’ and many others) has had increasing media attention and, with legislation in various countries being relaxed, it appears likely that there will be an increase in exposure generally. However, we still know very little about the long-term effects of smoking cannabis on the respiratory system and on health in general.
Cannabis comes from a flowering plant, native to central Asia and the Indian sub-continent. The genus includes three different species—Cannabis sativa, Cannabis indica and Cannabis ruderalis. They produce two major active compounds, delta-9-tetrahydrocannabinol (d-9-THC) and cannabidiol (CBD); however, they contain 60 cannabinoids and over 400 compounds in total.2 THC is the psychoactive compound, but it is modulated by CBD. The C. sativa-dominant strains have higher THC content, whereas the C. indica-dominant strains have higher CBD content.2 The C. ruderalis-dominant strains contain even lower THC concentrations than the C. indica-dominant strains.3 There is also ‘skunk-like’ cannabis, named for its smell, containing very high THC concentrations.4
Experimentally, the acute bronchodilator effect5,6 and analgesic properties7–9 of inhaled cannabis are well described. Acute bronchodilatation is due to THC.4 However, as cannabinoids can have partial agonist, or even antagonist, effects, little is known about differences in airway effects from different strains of cannabis containing varying concentrations of cannabinols.
Evidence that chronic cannabis users have an increased incidence of respiratory symptoms such as chronic cough, sputum production, dyspnoea, hoarse voice and chest tightness has been reviewed recently.6,10 As marijuana smoke contains many of the same compounds as, and shares similar properties with, cigarette smoke, respiratory symptoms would be expected.11,12
Methodological difficulties
There are obvious inherent difficulties in studying the long-term use of illicit substances. Examining dose–response is confounded by the problem of quantifying cannabis use—the measurement of ‘joint-years’ has inherent difficulties. One joint-year is defined as 365 joints smoked. However, because of the wide variety and strengths of marijuana in a joint,13,14 as well as the varying amounts that individuals put into a joint, this measure differs markedly between individuals. Reproducibility of joints in the same individual is not well defined. Furthermore, most studies use self-reported joint-years to quantify use; this may be inaccurate owing to the legality of cannabis use, recall bias and alteration of behaviour by its observation.
Another major consideration is the variety of ways that cannabis can be smoked, which alter the characteristics of the smoke inhaled. Using a water bong, e.g., decreased the concentration of inhaled carcinogenic compounds.11,15 Recent evidence has suggested that using a vapouriser to smoke cannabis may reduce pulmonary complications.16 Otherwise, there have been no peer-reviewed studies examining the effects of differing methods of smoking cannabis on lung health .
Cannabis and tobacco tend to be smoked differently. Typically, cannabis is smoked without a filter, to a shorter butt length, and the smoke is at a higher temperature. Furthermore, cannabis smokers inhale more deeply, hold their breath for longer and perform a Valsalva manoeuvre at maximal breath-hold.17–19
Some smokers make joints with marijuana leaves alone,20 whereas others usually smoke ‘spliffs’ containing both cannabis and tobacco leaves.21 This varies from one country to another. Cannabis users are also more likely to concurrently smoke tobacco, separately, making it difficult to disentangle the effects of cannabis itself on the lungs.
Effects of tobacco smoking
The effects of cigarette (tobacco) smoking on lung health are well known. They include symptoms of cough, sputum production (which when marked constitutes chronic bronchitis), wheeze and shortness of breath. Spirometric changes such as a progressive, largely irreversible decrease in forced expiratory volume in 1 s (FEV1) and FEV1 divided by forced vital capacity (FVC)−FEV1/FVC ratio occur. This ratio is the most reliable measure of airflow obstruction. These changes may be accompanied by air-trapping within the lungs, measured physiologically (as increased total lung capacity (TLC) or residual volume (RV) or the ratio RV/TLC) or radiologically, on chest X-ray, etc. Chronic persistent, progressive airway narrowing, damage to the alveoli (emphysema) and effects on small airways (investigated with more sophisticated methods, beyond the scope of this review), in varying proportions, comprise the condition chronic obstructive pulmonary disease (COPD). As cannabis was thought to affect the lungs in similar ways to tobacco, it was logical to use spirometric measurements to detect the adverse health effects of marijuana. However, with concurrent tobacco smoking, it is difficult to separate changes due to cannabis from those due to tobacco.
The general paucity of data, the evolving nature of available marijuana (newer, stronger forms, modes of inhalation, etc) and the important confounding factor of tobacco use have led to different interpretations of the health impact of marijuana by the public as well as within the research and medical community. The purpose of this review is to assess current knowledge of the chronic effects of cannabis smoking on respiratory function and the progression of pulmonary disease as well as to identify potential directions for future research.
Methods
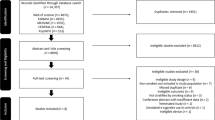
MEDLINE/PubMed (NLM), Scopus (Elsevier) and Science Citation Index Expanded (Web of Science) databases were searched for English-language peer-reviewed articles from 1 January 1968 to 6 June 2015. These were refined by the following search terms: ‘marijuana smoking lung’, ‘cannabis smoking lung’, ‘marijuana smoking pulmonary’ and ‘cannabis smoking pulmonary’. This method yielded 256 articles.
These initial 256 results were reduced to 114 peer-reviewed articles, as the other 142 were not published in peer-reviewed journals. The remaining 114 articles were individually screened by title and abstract, looking for measurements of pulmonary function and long-term cannabis use in humans. Case reports and case series were omitted. The final count contained 19 articles fitting all the above criteria. Each article was then individually appraised, with the main findings and conclusions tabulated.
An additional search was also conducted for English-language peer-reviewed articles, as above, containing the following search terms: ‘cannabis bullous lung’, ‘cannabis bullae lung’, ‘cannabis pneumothorax’ ‘bong lung’, ‘marijuana bullous lung’, ‘marijuana bullae lung’ and ‘marijuana pneumothorax’. This yielded 69 articles, which were individually screened by title and abstract for articles relating to bullous lung disease in marijuana smokers. Case reports and case series were included in this search, and the final count contained 18 articles.
Results
Simple lung function measurements
The results of these 19 studies are summarised in Table 1. A total of 11 studies were cross-sectional,20,22–31 and 8 were observational cohort studies.21,32–38 Eighteen out of 19 studies included spirometric measurements in chronic marijuana smokers.20–29,31–35,37–39 The results from these studies varied; eight studies found no significant changes in FEV1/FVC ratio,23,24,26–29,32,35 whereas six studies found a significant decrease in FEV1/FVC in chronic marijuana-only smokers compared with that in controls, with 0.5–1.9% reduction.20–22,34,37,38 The remaining studies also varied in their findings, but suggested that simply measuring the FEV1/FVC ratio does not accurately reflect the pulmonary effects of chronic marijuana use.
All studies reporting a significant decrease, ~1.5%, in FEV1/FVC ratio in marijuana smokers published incomplete data; in particular, most omitted the absolute results for FVC alone. However, two of these studies reported no significant effect on FEV1 in chronic cannabis use.20,22 Sherrill et al.34 in a follow-up survey of a random, stratified, cluster sample of the Tucson population, aged 15–60 years (n=856 who had at least two measurements), found a reduction in FEV1/FVC of −1.9±0.7% and in FEV1 of −142±44 ml only in previous marijuana smokers, with a nonsignificant decrease of −0.5±0.6% in current marijuana smokers.22 Surprisingly, in 1,239 of the same subjects, tested on at least one occasion, FEV1/FVC was reduced by 0.5±0.6% and FEV1 increased by 58 ml. However, in a population-based cohort born in 1972 and 1973 in Dunedin, New Zealand (n=1,037), Hancox et al. showed no significant association between chronic marijuana use and change in FEV1 or FEV1/FVC at age 32 but found an increased FVC.32 Subsequently, both Pletcher et al., in a longitudinal study over 20 years, recruited in 1985, examining Coronary Artery Risk Development In 5115 young Adults, and Kempker et al., in a cross-sectional study of 7,716 US adults from the National Health And Nutrition Examination Study cohort 2007–2008 and 2009–2010 surveys, reported similar findings as had Tilles et al. previously, although with a much smaller sample size (n=15 women).25,31,33 Pletcher et al., reported that FEV1 and FVC were 36 and 59 ml greater in cannabis users, with >10 joint-years’ smoking history, than in non-smoking controls.33 Kempker27 showed no effect on FEV1/FVC up to 20 joint-years but over 20 joint-years was associated with a 2.1-fold risk for FEV1/FVC ratio <70%, accounted for by a significant increase in FVC and no significant reduction in FEV1.25 Interestingly, Tashkin et al.29 reported a nonsignificant increase in both FEV1 and FVC in a convenience sample of 144 heavy marijuana smokers compared with other smoking groups, and in another study, in the same subjects,39 found that heavy habitual marijuana use, over a period of 8 years, was not associated with a decline in FEV1. Taken together, this information suggests that, although in some cases FEV1/FVC decreases by ~1.5% in chronic users, this may relate more to an increase in FVC rather than to a reduction in FEV1. This represents a major difference from the effects of tobacco smoking. The possibility that the effect on FEV1 is due to selection of people with higher FEV1 (because those with lower values do not smoke or do not continue to smoke cannabis) cannot be excluded in some of the studies.
Effects of dose and duration of exposure
Four studies found a dose-related response to marijuana exposure.20,21,25,33 Using ANCOVA, Aldington et al. (n=75 with a mean of 54.2 joint-years) found that, for every joint-year of smoking, there was a decrease in FEV1/FVC of 0.019%. Changes due to chronic cannabis use (per joint-year) were also found in specific airway conductance (−0.0017%), functional residual capacity (+0.0013%) and TLC (+0.002%) but not in FEV1 (unfortunately FVC was not reported). From this, they estimated that one pack-year of tobacco was equivalent to 4.1–7.9 joint-years of cannabis smoking (alternatively 1 joint of cannabis was equivalent to 2.5–5 cigarettes) in causing airflow obstruction.20 This was echoed by MacLeod et al., who found a 0.3% increase in the prevalence of COPD (defined by FEV1/FVC<0.70) for each additional joint-year in marijuana and tobacco concurrent smokers (n=252) in their sample of 500 subjects recruited from a general practice in Edinburgh, Scotland. It is important to note that subjects in this study were eligible for recruitment only if they reported significant tobacco or cannabis use, defined as at least 5 pack-years and/or 1 joint-year, with none of their participants smoking exclusively cannabis.21 Contrary to previous studies, Pletcher et al. (n=795 in Coronary Artery Risk Development In 5115 young Adults) noted that lifetime marijuana exposure was associated with an increase in FEV1 up to 7 joint-years with a decline thereafter at a slope of −2.2 ml/joint-year, although FEV1 was still nonsignificantly higher than in controls at all exposure levels (by 36 ml at >10 joint-years lifetime exposure).33 FVC, even after 20 joint-years, was still significantly raised by 59 ml. In both cases, however, FVC and FEV1 had a non-linear relationship with marijuana, which again differs from that of tobacco smoking.33 Kempker et al. calculated in 855 cannabis smokers that, for each additional joint-year smoked, there was no significant change in FEV1 %predicted (0.02+0.02), whereas FEV1/FVC decreased by −0.03±0.01% (P=0.02), accounted for by a significant increase in FVC 0.07+0.02% (P=0.004).25
There is evidence that current use of marijuana may influence spirometric measurements. Sherrill et al.34 reported that only former cannabis smokers (n=856) had decreased FEV1/FVC ratios, with no significant change in current smokers. In support, Kempker et al. found that use of cannabis in the past month was associated with an increased FVC for each additional day, with no FEV1 decrease.25 Eight of the 19 studies reported no criteria for abstinence from marijuana before spirometric testing.21,22,24,34,37–39 Strictly this confounds interpretation, making it difficult to distinguish acute from chronic effects of marijuana as acute bronchodilator effects of d-9-THC can be seen 2–3 h after inhalation6,40 and up to 6 h after oral ingestion.5
Other measures of lung function
Very few studies have examined more sophisticated measurements of lung physiology. Aldington et al. found a very small increase in plethysmographic TLC of 0.14 l in cannabis users in a convenience sample from the Greater Wellington region, New Zealand, which was supported by similar findings by Hancox et al., who reported an increase of just 0.03 l with an increase of 0.01 l in functional residual capacity and RV.32 Tilles et al. also found that marijuana smokers (n=15) had a TLC of 108±15% of predicted, which was significantly raised.31 Increased RV may indicate early signs of small airway dysfunction and air-trapping; however, it is not a very specific measure of small airway disease.41 Conversely, results from Tashkin et al.28,29 (n=74 and n=144) showed no significant changes in multiple measures of small airway function (FEV25–75%, closing volume, closing capacity, RV) between chronic cannabis smokers and non-smoking controls.
Three studies also reported an association of chronic cannabis smoking with increased airway resistance (0.03–0.38 cm H2O/l/s)28,29,32 and four studies found reduced specific airway conductance (0.007–0.07 ml/s/cm H2O/l).20,28,29,32 There was no association of these changes with change in lung volume.32 Central airway secretions or inflammation or oedema would be a potential explanation.
In studies measuring carbon monoxide transfer factor (TLco), two small studies (n=28 and n=15) found a significant decrease in TLco with chronic concurrent marijuana and tobacco use down to 65±15% of predicted.27,31 However, these studies did not find a significant reduction in TLco with marijuana-only use. Three other larger studies also found no significant decrease.20,29,32
Airway responsiveness was measured in three studies.24,30,38 Hernandez et al. studied 23 subjects from Texas, including six marijuana smokers, using histamine to measure bronchial responsiveness but found no difference in PD50 sRaw (dose of histamine causing a 50% increase in specific resistance, sRaw) in marijuana smokers compared with controls, whereas asthmatics showed significant hyper-reactivity to histamine. Larger studies by Tashkin et al., in a convenience sample of 113 out of 542 subjects, and by Taylor et al., in a sample of cannabis-dependent 21-year olds (n=91), from 943 members of the Dunedin birth cohort (n=1037), measured metacholine responsiveness as the provocative dose causing a 20% reduction in FEV1 (PC20). They found no significant difference in the number of marijuana-only smokers who had a positive PC20 metacholine compared with that in non-smokers. However, it is important to note that, after logistic regression to control for the effect of tobacco and cocaine, Tashkin et al. found that concurrent smokers of marijuana and tobacco had a significant association with hyper-responsiveness to metacholine.
The relationship between tobacco and marijuana together on lung function is complex and somewhat controversial. Sherman et al. reported a reduction in FEV1/FVC and TLco in a marijuana and tobacco-smoking group (n=13) but no change in the marijuana-alone group (n=19).27 Tan et al. in a sample of 878 people over 40 years of age in Vancouver, Canada, also found a significant increase in respiratory symptoms in the marijuana and tobacco-smoking group but not in the marijuana-only group.35 However, three other studies have reported no additive effects on lung function of chronically smoking both marijuana and tobacco.30,37,39
Respiratory symptoms
Eight out of nine studies, which quantified respiratory symptoms, reported an increase in respiratory complaints with odds ratios (ORs) of up to 2.98 compared with non-smoking controls.20–22,26,28,29,34,38 Respiratory symptoms recorded included increased incidence of cough, sputum production, shortness of breath and wheeze. Taylor et al. also reported significant increases in exercise-related shortness of breath (OR 1.65), nocturnal wakening with chest tightness (OR 1.72) and morning sputum production (OR 2.44) in a birth cohort of 1,037 subjects at 21 years of age.38 These ORs were even higher if casual cannabis users were excluded. However, in a cross-sectional study of 6,728 subjects gathered from the National Health And Nutrition Examination Study survey, which was conducted between 1988 and 1994, there was no association with shortness of breath (OR 1.29, P=0.26) compared with non-smoking controls after adjusting for age, although they did find an increase in cough, phlegm, wheezing and chronic bronchitis symptoms with similar OR to tobacco users, which is interesting as in this sample marijuana users were, on average, 10 years younger than tobacco smokers.26 Marijuana users (n=414) were included in the sample if they had smoked more than 100 cannabis cigarettes in total and had at least 1 day of use in the previous month; they were not asked about the frequency of use or overall exposure above 100 cigarettes. In addition to these nine studies, Hancox et al. also found an association between cannabis and morning cough, sputum production and wheeze compared with non-smokers.42
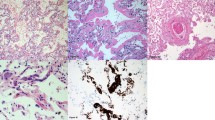
It was observed that cannabis smoke produces large airway epithelial damage, oedema, erythema and increased secretions with goblet cell hyperplasia,43 loss of ciliated epithelium and squamous metaplasia on biopsy. There is also evidence of additive bronchial epithelial damage in combined cannabis and tobacco smokers.44
Effect of quitting cannabis smoking
Examining the effect of quitting is another approach to determining the effect of smoking cannabis. The effect of quitting cigarette (tobacco) smoking is well known.45–47 Unfortunately, there are only two studies available of quitting cannabis. Tashkin reported that in a convenience sample of 299 young adults, including 95 regular cannabis-only smokers, 71 cannabis and tobacco concurrent smokers and 49 tobacco-only smokers, those who became non-smokers of both substances had no increased risk for chronic bronchitis compared with never smokers at a mean of 9.8 years of follow-up.29,48 However, those who continued smoking either substance had an increased risk for chronic bronchitis over the follow-up period. Hancox et al. analysed data from a well-characterised, population birth cohort of 1,037 subjects followed from birth to age 38 years with symptomatic assessment at 18, 26, 32 and 38 years of age.42 Frequent cannabis users numbered 157, 162, 138 and 99 compared with 540, 487, 608 and 703 non-users at different time points. There were significant reductions in morning cough, sputum production and wheeze, but not shortness of breath on exertion in the 26, 52, 54 and 50 quitters compared with continuing smokers, whether of cannabis alone or cannabis and tobacco. Furthermore, symptoms in the quitters were reduced to levels similar to those in never users. Sherill et al. reported that the risk for respiratory symptoms in previous non-tobacco smokers reduced to normal after quitting, but a significant reduction in FEV1, FEV1/FVC and Vmax50 persisted in previous cannabis smokers, although it was not present in current cannabis smokers.34
Bullous lung disease and emphysema
Characteristic, peripheral, cystic changes on high-resolution computed tomography scan are often found in patients who are (probably heavy) cannabis smokers,49 although the specificity and rate of occurrence of these findings are unclear. Only one cross-sectional study, recruited from the Greater Wellington area, measured radiological changes, reporting an increase in rates of macroscopic emphysema in tobacco smokers (16% in tobacco±cannabis n=183), but not in cannabis-only smokers (1.3% of 75).20 However, the cannabis smokers showed a significant increase in low-density lung regions compared with tobacco smokers, and this was interpreted as a result of airflow obstruction and hyperinflation rather than microscopic emphysema.
Aside from the above-mentioned study, 7 case series and 10 case reports were also found, including a total of 56 marijuana smokers presenting with bullous lung disease (often with pneumothorax) with concurrent tobacco smoking present in all but 3 cases. The results of these studies are summarised in Table 2. One case report was excluded because of being included in a subsequent case series.50,51 The majority of subjects in these cases were heavy smokers of marijuana, although it is quite difficult to compare usage as the units of measurement varied. Seven case series and six case reports had predominantly upper lobe involvement (2 with Vanishing Lung Syndrome, VLS), and one case report had predominantly lower lobe bullae. For those with upper lobe involvement, the majority had peripheral emphysema. Lung function was measured in four case series, with the majority of cases having normal lung function results.
Cannabis and lung cancer
Currently, the evidence regarding an association of cannabis smoking and lung cancer is inconclusive.52,53 Some studies have found precancerous histological changes in bronchial biopsies of marijuana smokers.44,54 However, epidemiological studies have not found a definite association. A pooled analysis of 6 case–control studies with a total of 2,159 lung cancer cases and 2,958 controls found little or no association between cannabis smoking and lung cancer; the overall pooled OR was 0.96 for habitual versus non-habitual smokers.55 Another large retrospective cohort study of 64,855 subjects found no increased risk for cancer after 8.6 years, although their subjects were relatively young even after follow-up.56 Other epidemiological studies have reported associations with lung cancer with heavier marijuana use. A 40-year longitudinal cohort study in 49,321 Swedish conscripts found that those who smoked cannabis >50 times had a twofold risk of developing lung cancer.57 Although this study is large, there are significant flaws in its methodology that limit the conclusions that can be drawn. The subjects were only assessed for tobacco and cannabis use at the time of conscription with no information on use patterns before conscription and the 40 years after conscription. A small case–control study in 403 subjects (79 lung cancer patients including 14 heavy cannabis users) also found, after adjusting for tobacco, an increased risk for lung cancer for each joint-year smoked, although not in the 2 lowest tertiles.58 However, owing to the small number of cases (n=14) and controls (n=4), it is highly likely that relative risk estimates are inflated.
Discussion
Although there are clear inconsistencies between these 19 studies, which may relate to subjects studied as well as methodology, we can nevertheless draw various conclusions regarding the effects of cannabis on lung function. Cannabis smokers usually also smoke tobacco, either concomitantly or separately. This makes it difficult to disentangle the effects of the two. Quantification of cannabis use compounds the difficulty of dose–response studies. Although studies consistently show the effects of cannabis on symptoms (chronic bronchitis), there are mixed reports regarding airflow obstruction. The larger cross-sectional and observational cohort studies have found no association with reduced FEV1/FVC. Self-selection (where subjects with a tendency to airway narrowing reject cannabis smoking) cannot be excluded. However, more recent, larger studies have found that chronic cannabis users have an increased FVC alone, or in conjunction with an increased FEV1.32,33 A larger rise in FVC compared with FEV1 could also contribute to previous observations of a decreased FEV1/FVC ratio.33
This contrasts with the reduced FEV1 and FEV1/FVC ratio (indicating airflow obstruction) consistently associated with cigarette smoking and COPD. The cause of the raised FVC in chronic marijuana smokers is unclear. It has been suggested that it might be because of training of respiratory muscles by the characteristic inhalation techniques employed by marijuana smokers.19,59,60 However, other evidence that training can increase FVC by this amount in humans is scant.61 Bronchodilatation of small airways can increase FVC, e.g., by up to 300 ml or 8.6% of predicted after inhaled salbutamol 100 μg.62 An acute bronchodilator effect of cannabis is well described; in three studies, marijuana was shown to increase FEV1 by 150–250 ml above baseline.6 Relative preservation of FEV1 (36 ml greater in marijuana smokers, with >10 joint-years of exposure, compared with that in controls) as found by Pletcher could relate to a bronchodilator effect, as suggested by Kempker et al.25
The lack of a defined marijuana abstinence period complicates interpretation of spirometric results. Current cannabis smokers had higher FVC values in studies of Kempker25 and Sherrill et al.34 Against this, tachyphylaxis to the bronchodilator effect would be anticipated, and in studies in asthmatics acute bronchodilatation lasted only about 2–6 h. The small but consistent increases in airway resistance, and reduction in airway conductance,28,29,32 suggest small effects on central airways, which may be explained by airway secretions, inflammation or oedema.
Another speculative, potential explanation for the absence of chronic airflow obstruction with cannabis smoking may relate to the well-documented anti-inflammatory and immunomodulatory effect of THC63—e.g., impairment of functional activity of stimulated alveolar macrophages (antimicrobial and respiratory burst, impaired cytokine production and nitric oxide production)27,64–66 thought to be critical in COPD pathogenesis.67–69
The clinical relevance of all this is unclear. COPD resulting from an inflammatory response in the airways to tobacco smoking is a major epidemic, currently the sixth leading cause of death worldwide and projected to be the fourth leading cause of death by 2030. Chronic marijuana smokers, who often also smoke tobacco, present with similar chronic respiratory symptoms but do not appear to develop airflow obstruction and COPD.
Future studies
In general, more information is required on cannabis smoking and the lung. The difficulties with joint-years as the measure of exposure have been mentioned. The observation that chronic marijuana smokers generally buy and keep track of their supply in grams suggests that grams per year may have advantages for both clinicians and researchers. It has been reported that the amount of cannabis purchased each month, and the intensity of the high afterwards, predicted respiratory symptoms independently of frequency of use.70 Although this will not overcome the problems with self-reporting illegal substance use, it may serve as a better measure of standardisation than joint-years.
Further research is necessary to clarify the relationship between respiratory symptoms and lung function. Further studies are needed to address acute bronchodilatation as a possible confounding factor in studies on chronic airflow obstruction. Serial examination of the bronchodilator effect of cannabis to look for tachyphylaxis would be of interest. More research into the effects of cannabis on the pathogenesis of COPD in relation to small airway inflammation, cytokine production and macrophage involvement is needed as well.
Other measures of airflow obstruction, including airway resistance, plethysmographic lung volumes and particularly measurements of small airways function, including imaging studies, are required. The priority is to understand why tobacco and cannabis smoking both cause chronic bronchitis yet have different effects on lung physiology. The pharmacological and pathophysiological basis of this needs to be established.
More work is needed in quitters of cannabis smoking, including motivations to quit, the effect on respiratory symptoms and lung function and bronchoscopic biopsy studies, to examine the effects on goblet cell hyperplasia and other histological findings.
Conclusions
This review clearly shows that chronic marijuana smoking is associated with respiratory symptoms and increase in FVC. The mechanisms for these effects and the differences from the effects of tobacco remain unclear. More work needs to accurately measure cannabis use as well as measure all aspects of respiratory health, particularly breathlessness and exercise tolerance. More importantly, however, there needs to be larger, longer-term studies with marijuana smokers who do not smoke tobacco.
There is clear evidence that marijuana causes similar symptoms to tobacco smoking (chronic bronchitis) and produces similar large airway pathological features. There is some evidence that the combination of tobacco and marijuana is additive. Tobacco unequivocally causes chronic airflow obstruction and COPD but only in a minority of smokers. Cannabis smoking, however, produces an increase in FVC and the reason(s) for this are unclear and require elucidation. Taking a more detailed history with regard to cannabis smoking and other illicit inhalational drugs should be part of the standard respiratory assessment of all patients, which would also support better epidemiological data collection for future studies, particularly in the primary care population.
References
UNODC. World Drug Report, Sales No. E.12.XI.1 (UNO, 2012).
Atakan, Z. Cannabis, a complex plant: different compounds and different effects on individuals. Ther. Adv. Psychopharmacol. 2, 241–254 (2012).
Beutler, J. A. & Marderosian, A. H. Chemotaxonomy of Cannabis I. Crossbreeding between Cannabis sativa and C. ruderalis, with analysis of cannabinoid content. Econ. Bot. 32, 387–394 (1978).
Di Forti, M. et al. High-potency cannabis and the risk of psychosis. Br. J. Psychiatry 195, 488–491 (2009).
Tashkin, D. P., Shapiro, B. J. & Frank, I. M. Acute pulmonary physiologic effects of smoked marijuana and oral 9 -tetrahydrocannabinol in healthy young men. N. Engl. J. Med. 289, 336–341 (1973).
Tetrault, J. M. et al. Effects of marijuana smoking on pulmonary function and respiratory complications: a systematic review. Arch. Intern. Med. 167, 221–228 (2007).
Ellis, R. J. et al. Smoked medicinal cannabis for neuropathic pain in HIV: a randomized, crossover clinical trial. Neuropsychopharmacology 34, 672–680 (2009).
Russo, E. Cannabinoids in pain management. Study was bound to conclude that cannabinoids had limited efficacy. BMJ 323, 1249–1250 (2001).
Russo, E. B. Cannabinoids in the management of difficult to treat pain. Ther. Clin. Risk Manag. 4, 245–259 (2008).
Howden, M. L. & Naughton, M. T. Pulmonary effects of marijuana inhalation. Expert Rev. Respir. Med. 5, 87–92 (2011).
Hoffmann, D., Rathkamp, G. & Wynder, E. L. Comparison of the yields of several selected components in the smoke from different tobacco products. J. Natl Cancer Inst. 31, 627–637 (1963).
Sparacino, C. M., Hyldburg, P. A. & Hughes, T. J. Chemical and biological analysis of marijuana smoke condensate. NIDA Res. Monogr. 99, 121–140 (1990).
Cascini, F., Aiello, C. & Di Tanna, G. Increasing delta-9-tetrahydrocannabinol (Delta-9-THC) content in herbal cannabis over time: systematic review and meta-analysis. Curr. Drug Abuse Rev. 5, 32–40 (2012).
Potter, D. J., Clark, P. & Brown, M. B. Potency of delta 9-THC and other cannabinoids in cannabis in England in 2005: implications for psychoactivity and pharmacology. J. Forensic Sci. 53, 90–94 (2008).
Savaki, H. E., Cunha, J., Carlini, E. A. & Kephalas, T. A. Pharmacological activity of three fractions obtained by smoking cannabis through a water pipe. Bull. Narc. 28, 49–56 (1976).
Van Dam, N. T. & Earleywine, M. Pulmonary function in cannabis users: Support for a clinical trial of the vaporizer. Int. J. Drug Policy 21, 511–513 (2010).
Tashkin, D. P. et al. Tar, CO and delta 9THC delivery from the 1st and 2nd halves of a marijuana cigarette. Pharmacol. Biochem. Behav. 40, 657–661 (1991).
Birrer, R. B. & Calderon, J. Pneumothorax, pneumomediastinum, and pneumopericardium following Valsalva's maneuver during marijuana smoking. NY State J. Med. 84, 619–620 (1984).
Wu, T. C., Tashkin, D. P., Djahed, B. & Rose, J. E. Pulmonary hazards of smoking marijuana as compared with tobacco. N. Engl. J. Med. 318, 347–351 (1988).
Aldington, S. et al. Effects of cannabis on pulmonary structure, function and symptoms. Thorax 62, 1058–1063 (2007).
Macleod, J. et al. Cannabis, tobacco smoking, and lung function: a cross-sectional observational study in a general practice population. Br. J. Gen. Pract. 65, e89–e95 (2015).
Bloom, J. W., Kaltenborn, W. T., Paoletti, P., Camilli, A. & Lebowitz, M. D. Respiratory effects of non-tobacco cigarettes. Br. Med. J. 295, 1516–1518 (1987).
Cruickshank, E. K. Physical assessment of 30 chronic cannabis users and 30 matched controls. Ann. NY Acad. Sci. 282, 162–167 (1976).
Hernandez, M. J., Martinez, F., Blair, H. T. & Miller, W. C. Airway response to inhaled histamine in asymptomatic long-term marijuana smokers. J. Allergy Clin. Immunol. 67, 153–155 (1981).
Kempker, J. A., Honig, E. G. & Martin, G. S. The effects of marijuana exposure on expiratory airflow. A study of adults who participated in the U.S. National Health and Nutrition Examination Study. Ann. Am. Thorac. Soc. 12, 135–141 (2015).
Moore, B. A., Augustson, E. M., Moser, R. P. & Budney, A. J. Respiratory effects of marijuana and tobacco use in a U.S. sample. J. Gen. Intern. Med. 20, 33–37 (2005).
Sherman, M. P., Roth, M. D., Gong, H. Jr & Tashkin, D. P. Marijuana smoking, pulmonary function, and lung macrophage oxidant release. Pharmacol. Biochem. Behav. 40, 663–669 (1991).
Tashkin, D. P., Calvarese, B. M., Simmons, M. S. & Shapiro, B. J. Respiratory status of seventy-four habitual marijuana smokers. Chest 78, 699–706 (1980).
Tashkin, D. P. et al. Respiratory symptoms and lung function in habitual heavy smokers of marijuana alone, smokers of marijuana and tobacco, smokers of tobacco alone, and nonsmokers. Am. Rev. Respir. Dis. 135, 209–216 (1987).
Tashkin, D. P., Simmons, M. S., Chang, P., Liu, H. & Coulson, A. H. Effects of smoked substance abuse on nonspecific airway hyperresponsiveness. Am. Rev. Respir. Dis. 147, 97–103 (1993).
Tilles, D. S. et al. Marijuana smoking as cause of reduction in single-breath carbon monoxide diffusing capacity. Am. J. Med. 80, 601–606 (1986).
Hancox, R. J. et al. Effects of cannabis on lung function: a population-based cohort study. Eur. Respir. J. 35, 42–47 (2010).
Pletcher, M. J. et al. Association between marijuana exposure and pulmonary function over 20 years. JAMA 307, 173–181 (2012).
Sherrill, D. L., Krzyzanowski, M., Bloom, J. W. & Lebowitz, M. D. Respiratory effects of non-tobacco cigarettes: a longitudinal study in general population. Int. J. Epidemiol. 20, 132–137 (1991).
Tan, W. C. et al. Marijuana and chronic obstructive lung disease: a population-based study. CMAJ 180, 814–820 (2009).
Tashkin, D. P. et al. Effects of 'crack' cocaine on pulmonary alveolar permeability. Chest 112, 327–335 (1997).
Taylor, D. R. et al. A longitudinal study of the effects of tobacco and cannabis exposure on lung function in young adults. Addiction 97, 1055–1061 (2002).
Taylor, D. R., Poulton, R., Moffitt, T. E., Ramankutty, P. & Sears, M. R. The respiratory effects of cannabis dependence in young adults. Addiction 95, 1669–1677 (2000).
Tashkin, D. P., Simmons, M. S., Sherrill, D. L. & Coulson, A. H. Heavy habitual marijuana smoking does not cause an accelerated decline in FEV1 with age. Am. J. Respir. Crit. Care Med. 155, 141–148 (1997).
Tashkin, D. P. et al. Bronchial effects of aerosolized delta 9-tetrahydrocannabinol in healthy and asthmatic subjects. Am. Rev. Respir. Dis. 115, 57–65 (1977).
McNulty, W. & Usmani, O. S. Techniques of assessing small airways dysfunction. Eur. Clin. Respir. J. 1, 1–17 (2014).
Hancox, R. J., Shin, H. H., Gray, A. R., Poulton, R. & Sears, M. R. Effects of quitting cannabis on respiratory symptoms. Eur. Respir. J. 46, 80–87 (2015).
Roth, M. D. et al. Airway inflammation in young marijuana and tobacco smokers. Am. J. Respir. Crit. Care Med. 157, 928–937 (1998).
Fligiel, S. E. et al. Tracheobronchial histopathology in habitual smokers of cocaine, marijuana, and/or tobacco. Chest 112, 319–326 (1997).
Kanner, R. E., Connett, J. E., Williams, D. E. & Buist, A. S. Effects of randomized assignment to a smoking cessation intervention and changes in smoking habits on respiratory symptoms in smokers with early chronic obstructive pulmonary disease: the Lung Health Study. Am. J. Med. 106, 410–416 (1999).
US Department of Health and Human Services. The Health Benefits of Smoking Cessation. DHHS Publication No. (CDC) 90-8416 (US Department of Health and Human Services, Public Health Service, Centers for Disease Control, Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health, 1990).
Tashkin, D. P. The respiratory health benefits of quitting cannabis use. Eur. Respir. J. 46, 1–4 (2015).
Tashkin, D. P., Simmons, M. S. & Tseng, C. H. Impact of changes in regular use of marijuana and/or tobacco on chronic bronchitis. COPD 9, 367–374 (2012).
Johnson, M. K., Smith, R. P., Morrison, D., Laszlo, G. & White, R. J. Large lung bullae in marijuana smokers. Thorax 55, 340–342 (2000).
Hii, S., Naughton, M. T. & Young, A. Marijuana lung. Int. Med. J. 36, 270–271 (2006).
Hii, S. W., Tam, J. D. C., Thompson, B. R. & Naughton, M. T. Bullous lung disease due to marijuana. Respirology 13, 122–127 (2008).
Biehl, J. R. & Burnham, E. L. Cannabis smoking in 2015: a concern for lung health? Chest 148, 596–606 (2015).
Tashkin, D. P. Increasing cannabis use: what we still need to know about its effects on the lung. Respirology 19, 619–620 (2014).
Barsky, S. H., Roth, M. D., Kleerup, E. C., Simmons, M. & Tashkin, D. P. Histopathologic and molecular alterations in bronchial epithelium in habitual smokers of marijuana, cocaine, and/or tobacco. J. Natl Cancer Inst. 90, 1198–1205 (1998).
Zhang, L. R. et al. Cannabis smoking and lung cancer risk: pooled analysis in the International Lung Cancer Consortium. Int. J. Cancer 136, 894–903 (2015).
Sidney, S., Quesenberry Jr C. P., Friedman, G. D. & Tekawa, I. S. Marijuana use and cancer incidence (California, United States). Cancer Causes Control 8, 722–728 (1997).
Callaghan, R. C., Allebeck, P. & Sidorchuk, A. Marijuana use and risk of lung cancer: a 40-year cohort study. Cancer Causes Control 24, 1811–1820 (2013).
Aldington, S. et al. Cannabis use and risk of lung cancer: a case-control study. Eur. Respir. J. 31, 280–286 (2008).
Lee, M. H. & Hancox, R. J. Effects of smoking cannabis on lung function. Expert Rev. Respir. Med. 5, 537–546 (2011).
Tashkin, D. P. Does marijuana pose risks for chronic airflow obstruction? Ann. Am. Thorac. Soc. 12, 235–236 (2015).
Freitas, D. A. et al. Breathing exercises for adults with asthma. Cochrane Database Syst. Rev. 10, CD001277 (2013).
Richter, D. C., Joubert, J. R., Nell, H., Schuurmans, M. M. & Irusen, E. M. Diagnostic value of post-bronchodilator pulmonary function testing to distinguish between stable, moderate to severe COPD and asthma. Int. J. Chron. Obstruct. Pulmon. Dis. 3, 693–699 (2008).
Tashkin, D. P. Does cannabis use predispose to chronic airflow obstruction? Eur. Respir. J. 35, 3–5 (2010).
Klein, T. W., Newton, C. A., Nakachi, N. & Friedman, H. Delta 9-tetrahydrocannabinol treatment suppresses immunity and early IFN-gamma, IL-12, and IL-12 receptor beta 2 responses to Legionella pneumophila infection. J. Immunol. 164, 6461–6466 (2000).
Zhu, W., Friedman, H. & Klein, T. W. Delta9-tetrahydrocannabinol induces apoptosis in macrophages and lymphocytes: involvement of Bcl-2 and caspase-1. J. Pharmacol. Exp. Ther. 286, 1103–1109 (1998).
Shay, A. H. et al. Impairment of antimicrobial activity and nitric oxide production in alveolar macrophages from smokers of marijuana and cocaine. J. Infect. Dis. 187, 700–704 (2003).
Saetta, M. et al. CD8+ T-lymphocytes in peripheral airways of smokers with chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 157, 822–826 (1998).
Turato, G. et al. Airway inflammation in severe chronic obstructive pulmonary disease: relationship with lung function and radiologic emphysema. Am. J. Respir. Crit. Care Med. 166, 105–110 (2002).
Hogg, J. C. et al. The nature of small-airway obstruction in chronic obstructive pulmonary disease. N. Engl. J. Med. 350, 2645–2653 (2004).
Walden, N. & Earleywine, M. How high: quantity as a predictor of cannabis-related problems. Harm Reduct. J. 5, 20 (2008).
Feldman, A. L., Sullivan, J. T., Passero, M. A. & Lewis, D. C. Pneumothorax in polysubstance-abusing marijuana and tobacco smokers: three cases. J. Subst. Abuse 5, 183–186 (1993).
Rawlins, R., Carr, C. S., Brown, K. M., Cameron, C. R. & Dussek, J. E. Quoted in Minerva. Bmj 323, 1012 (2001).
Thompson, C. S. & White, R. J. Lung bullae and marijuana. Thorax 57, 563 (2002).
Phan, T. D., Lau, K. K. P. & Li, X. Lung bullae and pulmonary fibrosis associated with marijuana smoking. Australas. Radiol. 49, 411–414 (2005).
Beshay, M., Kaiser, H., Niedhart, D., Reymond, M. A. & Schmid, R. A. Emphysema and secondary pneumothorax in young adults smoking cannabis. Eur. J. Cardiothorac. Surg. 32, 834–838 (2007).
Reece, A. S. Severe multisystem dysfunction in a case of high level exposure to smoked cannabis. BMJ Case Rep. 2009, bcr08.2008.0798 (2009).
Gao, Z. et al. ‘Bong lung’ in cystic fibrosis: a case report. J. Med. Case Rep. 4, 371 (2010).
Allen, R. K. A. Bullectomy for ‘bong lung’ in an 18 year-old male presenting with spontaneous pneumothorax. Pneumon 23, 301–303 (2010).
Shah, A. & Paramlal, M. The importance of an illicit drug history in the evaluation of suspected spontaneous pneumothorax. BMJ Case Rep. 2011, bcr0120113693 (2011).
Sood, N. & Sood, N. A rare case of vanishing lung syndrome. Case Rep. Pulmonol. 2011, 957463 (2011).
Gargani, Y., Bishop, P. & Denning, D. W. Too many mouldy joints—marijuana and chronic pulmonary aspergillosis. Mediterr. J. Hematol. Infect. Dis. 3, e2011005 (2011).
Golwala, H. Marijuana abuse and bullous emphysema. Lung India 29, 56–58 (2012).
Tashtoush B., Gonzalez-Ibarra F., Memarpour R., Hadeh A. & Smolley L. Vanishing lung syndrome in a patient with HIV infection and heavy marijuana use. Case Rep. Pulmonol. 2014 285208 (2014).
Fiorelli, A. et al. Does cannabis smoking predispose to lung bulla formation? Asian Cardiovasc. Thorac. Ann. 22, 65–71 (2014).
Cary, R. M., Bragg, C. & Mukherjee, J. Pleuritic chest pain and fluid levels on imaging in a heavy cannabis smoker. BMJ Case Rep. 2015, bcr2014208064 (2015).
Acknowledgements
Funding
The authors declare that no funding was received.
Author information
Authors and Affiliations
Contributions
LIGR is the main author who contributed to the design and writing of the study and performed the systematic literature search. PWI supervised the whole work and assisted in the writing and design of the study.
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Ribeiro, L., Ind, P. Effect of cannabis smoking on lung function and respiratory symptoms: a structured literature review. npj Prim Care Resp Med 26, 16071 (2016). https://doi.org/10.1038/npjpcrm.2016.71
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/npjpcrm.2016.71
This article is cited by
-
Cannabis use prevalence, patterns, and reasons for use among patients with cancer and survivors in a state without legal cannabis access
Supportive Care in Cancer (2023)
-
Cannabis and Cannabinoids in Reproduction and Fertility: Where We Stand
Reproductive Sciences (2022)
-
Cannabis and Lung Health: Does the Bad Outweigh the Good?
Pulmonary Therapy (2021)
-
Cannabis-Associated Asthma and Allergies
Clinical Reviews in Allergy & Immunology (2019)