Abstract
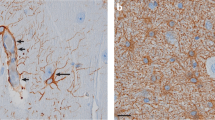
Reactive astrocytosis develops in many neurologic diseases, including epilepsy. Astrocytotic contributions to pathophysiology are poorly understood. Studies examining this are confounded by comorbidities accompanying reactive astrocytosis. We found that high-titer transduction of astrocytes with enhanced green fluorescent protein (eGFP) via adeno-associated virus induced reactive astrocytosis without altering the intrinsic properties or anatomy of neighboring neurons. We examined the consequences of selective astrocytosis induction on synaptic transmission in mouse CA1 pyramidal neurons. Neurons near eGFP-labeled reactive astrocytes had reduced inhibitory, but not excitatory, synaptic currents. This inhibitory postsynaptic current (IPSC) erosion resulted from a failure of the astrocytic glutamate-glutamine cycle. Reactive astrocytes downregulated expression of glutamine synthetase. Blockade of this enzyme normally induces rapid synaptic GABA depletion. In astrocytotic regions, residual inhibition lost sensitivity to glutamine synthetase blockade, whereas exogenous glutamine administration enhanced IPSCs. Astrocytosis-mediated deficits in inhibition triggered glutamine-reversible hyperexcitability in hippocampal circuits. Thus, reactive astrocytosis could generate local synaptic perturbations, leading to broader functional deficits associated with neurologic disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Haydon, P.G. & Carmignoto, G. Astrocyte control of synaptic transmission and neurovascular coupling. Physiol. Rev. 86, 1009–1031 (2006).
Pascual, O. et al. Astrocytic purinergic signaling coordinates synaptic networks. Science 310, 113–116 (2005).
Volterra, A. & Meldolesi, J. Astrocytes, from brain glue to communication elements: the revolution continues. Nat. Rev. Neurosci. 6, 626–640 (2005).
Bak, L.K., Schousboe, A. & Waagepetersen, H.S. The glutamate/GABA-glutamine cycle: aspects of transport, neurotransmitter homeostasis and ammonia transfer. J. Neurochem. 98, 641–653 (2006).
de Melo Reis, R.A., Ventura, A.L., Schitine, C.S., de Mello, M.C. & de Mello, F.G. Müller glia as an active compartment modulating nervous activity in the vertebrate retina: neurotransmitters and trophic factors. Neurochem. Res. 33, 1466–1474 (2008).
Syková, E. & Nicholson, C. Diffusion in brain extracellular space. Physiol. Rev. 88, 1277–1340 (2008).
Chaudhry, F.A., Reimer, R.J. & Edwards, R.H. The glutamine commute: take the N line and transfer to the A. J. Cell Biol. 157, 349–355 (2002).
Liang, S.L., Carlson, G.C. & Coulter, D.A. Dynamic regulation of synaptic GABA release by the glutamate-glutamine cycle in hippocampal area CA1. J. Neurosci. 26, 8537–8548 (2006).
Fricke, M.N., Jones-Davis, D.M. & Mathews, G.C. Glutamine uptake by System A transporters maintains neurotransmitter GABA synthesis and inhibitory synaptic transmission. J. Neurochem. 102, 1895–1904 (2007).
Kam, K. & Nicoll, R. Excitatory synaptic transmission persists independently of the glutamate-glutamine cycle. J. Neurosci. 27, 9192–9200 (2007).
Eid, T. et al. Loss of glutamine synthetase in the human epileptogenic hippocampus: possible mechanism for raised extracellular glutamate in mesial temporal lobe epilepsy. Lancet 363, 28–37 (2004).
Eid, T., Williamson, A., Lee, T.S., Petroff, O.A. & de Lanerolle, N.C. Glutamate and astrocytes—key players in human mesial temporal lobe epilepsy? Epilepsia 49, 42–52 (2008).
Sepkuty, J.P. et al. A neuronal glutamate transporter contributes to neurotransmitter GABA synthesis and epilepsy. J. Neurosci. 22, 6372–6379 (2002).
Robinson, S.R. Changes in the cellular distribution of glutamine synthetase in Alzheimer's disease. J. Neurosci. Res. 66, 972–980 (2001).
Tilleux, S. & Hermans, E. Neuroinflammation and regulation of glial glutamate uptake in neurological disorders. J. Neurosci. Res. 85, 2059–2070 (2007).
Seifert, G., Schilling, K. & Steinhauser, C. Astrocyte dysfunction in neurological disorders: a molecular perspective. Nat. Rev. Neurosci. 7, 194–206 (2006).
Pekny, M. & Nilsson, M. Astrocyte activation and reactive gliosis. Glia 50, 427–434 (2005).
Wilhelmsson, U. et al. Absence of glial fibrillary acidic protein and vimentin prevents hypertrophy of astrocytic processes and improves post-traumatic regeneration. J. Neurosci. 24, 5016–5021 (2004).
Wetherington, J., Serrano, G. & Dingledine, R. Astrocytes in the epileptic brain. Neuron 58, 168–178 (2008).
Steffens, M., Huppertz, H.J., Zentner, J., Chauzit, E. & Feuerstein, T.J. Unchanged glutamine synthetase activity and increased NMDA receptor density in epileptic human neocortex: implications for the pathophysiology of epilepsy. Neurochem. Int. 47, 379–384 (2005).
Rosati, A. et al. Epilepsy in glioblastoma multiforme: correlation with glutamine synthetase levels. J. Neurooncol. 93, 319–324 (2009).
Barberis, A., Petrini, E.M. & Cherubini, E. Presynaptic source of quantal size variability at GABAergic synapses in rat hippocampal neurons in culture. Eur. J. Neurosci. 20, 1803–1810 (2004).
Parpura, V. et al. Glutamate-mediated astrocyte-neuron signaling. Nature 369, 744–747 (1994).
Rizzoli, S.O. & Betz, W.J. Synaptic vesicle pools. Nat. Rev. Neurosci. 6, 57–69 (2005).
Burrone, J., Li, Z. & Murthy, V.N. Studying vesicle cycling in presynaptic terminals using the genetically encoded probe synaptopHluorin. Nat. Protoc. 1, 2970–2978 (2006).
Oliva, A.A. Jr., Jiang, M., Lam, T., Smith, K.L. & Swann, J.W. Novel hippocampal interneuronal subtypes identified using transgenic mice that express green fluorescent protein in GABAergic interneurons. J. Neurosci. 20, 3354–3368 (2000).
Ang, C.W., Carlson, G.C. & Coulter, D.A. Hippocampal CA1 circuitry dynamically gates direct cortical inputs preferentially at theta frequencies. J. Neurosci. 25, 9567–9580 (2005).
Ang, C.W., Carlson, G.C. & Coulter, D.A. Massive and specific dysregulation of direct cortical input to the hippocampus in temporal lobe epilepsy. J. Neurosci. 26, 11850–11856 (2006).
Bushong, E.A., Martone, M.E., Jones, Y.Z. & Ellisman, M. Protoplasmic astrocytes in CA1 stratum radiatum occupy separate anatomical domains. J. Neurosci. 22, 183–192 (2002).
Mozrzymas, J.W. et al. GABA transient sets the susceptibility of mIPSCs to modulation by benzodiazepine receptor agonists in rat hippocampal neurons. J. Physiol. (Lond.) 585, 29–46 (2007).
Cohen, A.S., Lin, D.D. & Coulter, D.A. Protracted postnatal development of inhibitory synaptic transmission in rat hippocampal area CA1 neurons. J. Neurophysiol. 84, 2465–2476 (2000).
Sun, H.Y., Lyons, S.A. & Dobrunz, L.E. Mechanisms of target-cell specific short-term plasticity at Schaffer collateral synapses onto interneurones versus pyramidal cells in juvenile rats. J. Physiol. (Lond.) 568, 815–840 (2005).
Jakubs, K. et al. Inflammation regulates functional integration of neurons born in adult brain. J. Neurosci. 28, 12477–12488 (2008).
Galic, M.A. et al. Postnatal inflammation increases seizure susceptibility in adult rats. J. Neurosci. 28, 6904–6913 (2008).
Binder, D.K. & Steinhauser, C. Functional changes in astroglial cells in epilepsy. Glia 54, 358–368 (2006).
Cameron, H.A. Quantitative analysis of in vivo cell proliferation. in Current Protocols in Neuroscience Ch 3, Unit 3.9 (2006).
Ortinski, P.I., Lu, C., Takagaki, K., Fu, Z. & Vicini, S. Expression of distinct alpha subunits of GABAA receptor regulates inhibitory synaptic strength. J. Neurophysiol. 92, 1718–1727 (2004).
Acknowledgements
This work was supported by US National Institutes of Health grants P01 NS054900 and P20 MH071705 to D.A.C., P01NS054900, R01NS054770 and R01NS037585 to P.G.H., R01NS040978 to D.J.W. and by an Epilepsy Foundation Research Fellowship to P.I.O.
Author information
Authors and Affiliations
Contributions
P.I.O. and J.D. conducted and analyzed all of the experiments. A.M. assisted with viral vector production. C.Y. contributed to VSD data collection and analysis. H.T. assisted with confocal and multiphoton microscope data acquisition and processing. D.J.W. contributed to initial generation of the AAV-injected mice. P.I.O. and D.A.C. wrote the manuscript with help from P.G.H. and J.D. D.A.C. and P.G.H. designed the experiments with P.I.O. and J.D. and supervised the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–11 and Supplementary Tables 1 and 2 (PDF 16834 kb)
Rights and permissions
About this article
Cite this article
Ortinski, P., Dong, J., Mungenast, A. et al. Selective induction of astrocytic gliosis generates deficits in neuronal inhibition. Nat Neurosci 13, 584–591 (2010). https://doi.org/10.1038/nn.2535
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2535
This article is cited by
-
Serum protein S-100B as a novel biomarker of diagnosis and prognosis of childhood epilepsy
The Egyptian Journal of Neurology, Psychiatry and Neurosurgery (2023)
-
Adenosine-independent regulation of the sleep–wake cycle by astrocyte activity
Cell Discovery (2023)
-
Astrocyte-oligodendrocyte interaction regulates central nervous system regeneration
Nature Communications (2023)
-
Astrocyte reactivity and inflammation-induced depression-like behaviors are regulated by Orai1 calcium channels
Nature Communications (2023)
-
Astrocytes in human central nervous system diseases: a frontier for new therapies
Signal Transduction and Targeted Therapy (2023)