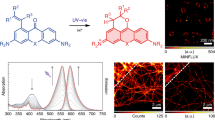
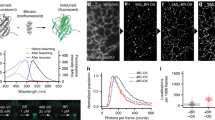
Abstract
Fluorescence nanoscopy has revolutionized our ability to visualize biological structures not resolvable by conventional microscopy. However, photodamage induced by intense light exposure has limited its use in live specimens. Here we describe Kohinoor, a fast-switching, positively photoswitchable fluorescent protein, and show that it has high photostability over many switching repeats. With Kohinoor, we achieved super-resolution imaging of live HeLa cells using biocompatible, ultralow laser intensity (0.004 J/cm2) in reversible saturable optical fluorescence transition (RESOLFT) nanoscopy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Accession codes
References
Hell, S.W. & Wichmann, J. Opt. Lett. 19, 780–782 (1994).
Tiwari, D.K. & Nagai, T. Dev. Growth Differ. 55, 491–507 (2013).
Klar, T.A., Jakobs, S., Dyba, M., Egner, A. & Hell, S.W. Proc. Natl. Acad. Sci. USA 97, 8206–8210 (2000).
Gustafsson, M.G. J. Microsc. 198, 82–87 (2000).
Betzig, E. et al. Science 313, 1642–1645 (2006).
Chen, B.C. et al. Science 346, 1257998 (2014).
Wu, Y. et al. Nat. Biotechnol. 31, 1032–1038 (2013).
Hell, S.W. Phys. Lett. A 326, 140–145 (2004).
Hofmann, M., Eggeling, C., Jakobs, S. & Hell, S.W. Proc. Natl. Acad. Sci. USA 102, 17565–17569 (2005).
Grotjohann, T. et al. eLife 1, e00248 (2012).
Grotjohann, T. et al. Nature 478, 204–208 (2011).
Chmyrov, A. et al. Nat. Methods 10, 737–740 (2013).
Brakemann, T. et al. Nat. Biotechnol. 29, 942–947 (2011).
Andresen, M. et al. Nat. Biotechnol. 26, 1035–1040 (2008).
Andresen, M. et al. Proc. Natl. Acad. Sci. USA 102, 13070–13074 (2005).
Schwentker, M.A. et al. Microsc. Res. Tech. 70, 269–280 (2007).
Ando, R., Mizuno, H. & Miyawaki, A. Science 306, 1370–1373 (2004).
Zhao, H., Giver, L., Shao, Z., Affholter, J.A. & Arnold, F.H. Nat. Biotechnol. 16, 258–261 (1998).
Brakemann, T. et al. J. Biol. Chem. 285, 14603–14609 (2010).
Kao, Y.-T., Zhu, X., Xu, F. & Min, W. Biomed. Opt. Express 3, 1955–1963 (2012).
Hafi, N. et al. Nat. Methods 11, 579–584 (2014).
Stiel, A.C. et al. Biochem. J. 402, 35–42 (2007).
Matsuda, T., Miyawaki, A. & Nagai, T. Nat. Methods 5, 339–345 (2008).
Tomosugi, W. et al. Nat. Methods 6, 351–353 (2009).
Shaner, N.C. et al. Nat. Methods 10, 407–409 (2013).
Gayda, S., Nienhaus, K. & Nienhaus, G.U. Biophys. J. 103, 2521–2531 (2012).
Pfennig, N. & Wagener, S. J. Microbiol. Methods 4, 303–306 (1986).
Acknowledgements
This work was supported by a grant-in-aid for Scientific Research on Innovative Areas, 'Spying minority in biological phenomena (no. 3306)', from the Ministry of Education, Culture, Sports, Science and Technology, Japan (no. 23115003) (to T.N.), Cooperative Research Program of 'Network Joint Research Center for Materials and Devices' (to K.F.) and a postdoctoral fellowship from the Japan Society for the Promotion of Science (to D.K.T.).
Author information
Authors and Affiliations
Contributions
T.N. conceived the project. D.K.T. constructed Kohinoor and characterized it with contributions from T.M. and M.N. M.Y. and K.F. set up the RESOLFT microscopy. D.K.T., M.Y. and Y.A. performed RESOLFT imaging. M.A. and Y.A. performed two-photon imaging. All authors discussed and commented on the results and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors have filed an international patent application for Kohinoor (no. PCT-JP2014-074121).
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–17 and Supplementary Tables 1–4 (PDF 12752 kb)
Repeated photoswitching of Kohinoor-β-actin in a live HeLa cell
Photoswitching was repeated 500 times by the alternative excitation of 405-nm and 488-nm lasers in a confocal laserscanning microscope. The cell did not show any morphological changes during photoswitching, indicating the lack of phototoxicity of the 405-nm light. (MP4 15346 kb)
Rights and permissions
About this article
Cite this article
Tiwari, D., Arai, Y., Yamanaka, M. et al. A fast- and positively photoswitchable fluorescent protein for ultralow-laser-power RESOLFT nanoscopy. Nat Methods 12, 515–518 (2015). https://doi.org/10.1038/nmeth.3362
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth.3362
This article is cited by
-
Super-resolution imaging of fluorescent dipoles via polarized structured illumination microscopy
Nature Communications (2019)
-
Super-resolution fluorescence microscopy studies of human immunodeficiency virus
Retrovirology (2018)
-
Specialty probes give super-res imaging that special blink
Nature Methods (2018)
-
Fast reversibly photoswitching red fluorescent proteins for live-cell RESOLFT nanoscopy
Nature Methods (2018)
-
Novel reversibly switchable fluorescent proteins for RESOLFT and STED nanoscopy engineered from the bacterial photoreceptor YtvA
Scientific Reports (2018)