Abstract
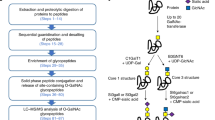
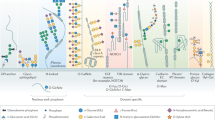
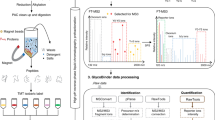
Zinc-finger nuclease (ZFN) gene targeting is emerging as a versatile tool for engineering of multiallelic gene deficiencies. A longstanding obstacle for detailed analysis of glycoproteomes has been the extensive heterogeneities in glycan structures and attachment sites. Here we applied ZFN targeting to truncate the O-glycan elongation pathway in human cells, generating stable 'SimpleCell' lines with homogenous O-glycosylation. Three SimpleCell lines expressing only truncated GalNAcα or NeuAcα2-6GalNAcα O-glycans were produced, allowing straightforward isolation and sequencing of GalNAc O-glycopeptides from total cell lysates using lectin chromatography and nanoflow liquid chromatography–mass spectrometry (nLC-MS/MS) with electron transfer dissociation fragmentation. We identified >100 O-glycoproteins with >350 O-glycan sites (the great majority previously unidentified), including a GalNAc O-glycan linkage to a tyrosine residue. The SimpleCell method should facilitate analyses of important functions of protein glycosylation. The strategy is also applicable to other O-glycoproteomes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
08 January 2015
In the version of this article initially published, the T3M4 cell line used for the experiments was mislabeled as Capan-1. The error has been corrected in the HTML and PDF versions of the article.
References
Gill, D., Clausen, H. & Bard, F. Location, location, location: new insights into O-GalNAc protein glycosylation. Trends Cell Biol. 23, 149–158 (2010).
Gram Schjoldager, K.T. et al. O-glycosylation modulates proprotein convertase activation of angiopoietin-like protein 3 – possible role of polypeptide GalNAc-transferase-2 in regulation of concentrations of plasma lipids. J. Biol. Chem. 285, 36293–36303 (2010).
Kato, K. et al. Polypeptide GalNAc-transferase T3 and familial tumoral calcinosis. Secretion of fibroblast growth factor 23 requires O-glycosylation. J. Biol. Chem. 281, 18370–18377 (2006).
Hassan, H., Bennett, E.P., Mandel, U., Hollingsworth, M.A. & Clausen, H. Control of mucin-type O-glycosylation: O-glycan occupancy is directed by substrate specificities of polypeptide GalNAc transferases. in Carbohydrates in Chemistry and Biology. A Comprehension Handbook (eds. Ernst, B., Hart, G.W. and Sinay, P.) 273–292 (Wiley-VCH, New York, 2000).
Schwientek, T. et al. Functional conservation of subfamilies of putative UDP-N-acetylgalactosamine:polypeptide N-acetylgalactosaminyltransferases in Drosophila, Caenorhabditis elegans, and mammals. One subfamily composed of l(2)35Aa is essential in Drosophila. J. Biol. Chem. 277, 22623–22638 (2002).
Chalkley, R.J., Thalhammer, A., Schoepfer, R. & Burlingame, A.L. Identification of protein O-GlcNAcylation sites using electron transfer dissociation mass spectrometry on native peptides. Proc. Natl. Acad. Sci. USA 106, 8894–8899 (2009).
Darula, Z. & Medzihradszky, K.F. Affinity enrichment and characterization of mucin core-1 type glycopeptides from bovine serum. Mol. Cell. Proteomics 8, 2515–2526 (2009).
Iwai, T. et al. Core 3 synthase is down-regulated in colon carcinoma and profoundly suppresses the metastatic potential of carcinoma cells. Proc. Natl. Acad. Sci. USA 102, 4572–4577 (2005).
Wang, Y. et al. Cosmc is an essential chaperone for correct protein O-glycosylation. Proc. Natl. Acad. Sci. USA 107, 9228–9233 (2010).
Ju, T. et al. Human tumor antigens Tn and sialyl Tn arise from mutations in Cosmc. Cancer Res. 68, 1636–1646 (2008).
Santiago, Y. et al. Targeted gene knockout in mammalian cells by using engineered zinc-finger nucleases. Proc. Natl. Acad. Sci. USA 105, 5809–5814 (2008).
Charlier, E. et al. SHIP-1 inhibits CD95/APO-1/Fas-induced apoptosis in primary T lymphocytes and T leukemic cells by promoting CD95 glycosylation independently of its phosphatase activity. Leukemia 24, 821–832 (2010).
Nilsson, J. et al. Enrichment of glycopeptides for glycan structure and attachment site identification. Nat. Methods 6, 809–811 (2009).
Rudd, P.M. et al. The glycosylation of the complement regulatory protein, human erythrocyte CD59. Adv. Exp. Med. Biol. 435, 153–162 (1998).
Hilvo, M. et al. Biochemical characterization of CA IX, one of the most active carbonic anhydrase isozymes. J. Biol. Chem. 283, 27799–27809 (2008).
Hayes, G.R., Enns, C.A. & Lucas, J.J. Identification of the O-linked glycosylation site of the human transferrin receptor. Glycobiology 2, 355–359 (1992).
Pisano, A., Redmond, J.W., Williams, K.L. & Gooley, A.A. Glycosylation sites identified by solid-phase Edman degradation: O-linked glycosylation motifs on human glycophorin A. Glycobiology 3, 429–435 (1993).
Oxvig, C., Haaning, J., Hojrup, P. & Sottrup-Jensen, L. Location and nature of carbohydrate groups in proform of human major basic protein isolated from pregnancy serum. Biochem. Mol. Biol. Int. 33, 329–336 (1994).
Nilsson, J., Nilsson, J., Larson, G. & Grahn, A. Characterization of site-specific O-glycan structures within the mucin-like domain of α-dystroglycan from human skeletal muscle. Glycobiology 20, 1160–1169 (2010).
Stalnaker, S.H. et al. Site mapping and characterization of O-glycan structures on α-dystroglycan isolated from rabbit skeletal muscle. J. Biol. Chem. 285, 24882–24891 (2010).
Perdivara, I. et al. Elucidation of O-glycosylation structures of the β-amyloid precursor protein by liquid chromatography-mass spectrometry using electron transfer dissociation and collision induced dissociation. J. Proteome Res. 8, 631–642 (2009).
Halim, A. et al. Site-specific characterization of threonine, serine, and tyrosine glycosylations of amyloid precursor protein/amyloid β-peptides in human cerebrospinal fluid. Proc. Natl. Acad. Sci. USA 108, 11848–11853 (2011).
Julenius, K., Molgaard, A., Gupta, R. & Brunak, S. Prediction, conservation analysis, and structural characterization of mammalian mucin-type O-glycosylation sites. Glycobiology 15, 153–164 (2005).
Mandel, U. et al. Expression of polypeptide GalNAc-transferases in stratified epithelia and squamous cell carcinomas: immunohistological evaluation using monoclonal antibodies to three members of the GalNAc-transferase family. Glycobiology 9, 43–52 (1999).
Wandall, H.H. et al. Substrate specificities of three members of the human UDP-N-acetyl-α-D-galactosamine:polypeptide N-acetylgalactosaminyltransferase family, GalNAc-T1, -T2, and -T3. J. Biol. Chem. 272, 23503–23514 (1997).
Tenno, M. et al. Initiation of protein O-glycosylation by the polypeptide GalNAcT-1 in vascular biology and humoral immunity. Mol. Cell. Biol. 27, 8783–8796 (2007).
Kingsley, D.M., Kozarsky, K.F., Segal, M. & Krieger, M. Three types of low density lipoprotein receptor-deficient mutant have pleiotropic defects in the synthesis of N-linked, O-linked, and lipid-linked carbohydrate chains. J. Cell Biol. 102, 1576–1585 (1986).
Rottger, S. et al. Localization of three human polypeptide GalNAc-transferases in HeLa cells suggests initiation of O-linked glycosylation throughout the Golgi apparatus. J. Cell Sci. 111, 45–60 (1998).
Serafini-Cessi, F., Dall'Olio, F., Malagolini, N. & Campadelli-Fiume, G. Temporal aspects of O-glycosylation of glycoprotein C from herpes simplex virus type-1. Biochem. J. 262, 479–484 (1989).
Rauschert, N. et al. A new tumor-specific variant of GRP78 as target for antibody-based therapy. Lab. Invest. 88, 375–386 (2008).
Acknowledgements
We thank R. Chalkley, K. Medzihradszky, and T. Schwientek for helpful discussion and advice, M.A. Hollingsworth for critical reading of the manuscript, and T. Ju and R. Cummings (Emory University School of Medicine) for providing the C1GalT1 enzyme for generation of the monoclonal antibody, S. Itzkowitz (Mount Sinai Medical Center) for cell lines, and R. Viner for valuable advice on implementing the HCD-triggered ETD experiment. This work was supported by Kirsten og Freddy Johansen Fonden, A.P. Møller og Hustru Chastine Mc-Kinney Møllers Fond til Almene Formaal, The Carlsberg Foundation, The Novo Nordisk Foundation, The Alfred Benzon Foundation, The Danish Research Councils and a program of excellence from the University of Copenhagen.
Author information
Authors and Affiliations
Contributions
C.S., S.Y.V., S.B.L. and H.C. designed and performed experiments, analyzed data and wrote the paper. K.T.-B.G.S. and H.W. designed experiments. M.B.V.-C., Y.K., E.P.B. and U.M. performed experiments.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–3, Supplementary Tables 1–2 and Supplementary Results (PDF 2293 kb)
Rights and permissions
About this article
Cite this article
Steentoft, C., Vakhrushev, S., Vester-Christensen, M. et al. Mining the O-glycoproteome using zinc-finger nuclease–glycoengineered SimpleCell lines. Nat Methods 8, 977–982 (2011). https://doi.org/10.1038/nmeth.1731
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth.1731
This article is cited by
-
Endoplasmic reticulum stress: molecular mechanism and therapeutic targets
Signal Transduction and Targeted Therapy (2023)
-
Identification of global inhibitors of cellular glycosylation
Nature Communications (2023)
-
Glycoproteomics
Nature Reviews Methods Primers (2022)
-
StrucGP: de novo structural sequencing of site-specific N-glycan on glycoproteins using a modularization strategy
Nature Methods (2021)
-
The clinical impact of glycobiology: targeting selectins, Siglecs and mammalian glycans
Nature Reviews Drug Discovery (2021)