Abstract
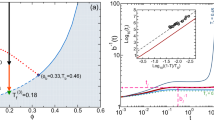
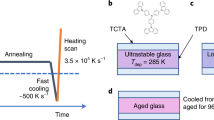
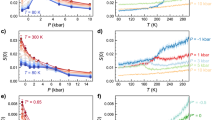
The existence of more than two liquid states in a single-component substance and the ensuing liquid–liquid transitions (LLTs) has attracted considerable attention because of its counterintuitive nature and its importance in the fundamental understanding of the liquid state. Here we report direct experimental evidence for a genuine (isocompositional) LLT without macroscopic phase separation in an aqueous solution of glycerol. We show that liquid I transforms into liquid II by way of two types of kinetics: nucleation and growth, and spinodal decomposition. Although liquid II is metastable against crystallization, we could access both its static and dynamical properties experimentally. We find that liquids I and II differ in density, refractive index, structure, hydrogen bonding state, glass transition temperature and fragility, and that the transition between the two liquids is mainly driven by the local structuring of water rather than of glycerol, suggesting a link to a plausible LLT inpure water.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Mishima, O. & Stanley, H. E. The relationship between liquid, supercooled and glassy water. Nature 396, 329–335 (1998).
Angell, C. A. Formation of glasses from liquids and biopolymers. Science 267, 1924–1935 (1995).
Angell, C. A. Insights into phases of liquid water from study of its unusual glass-forming properties. Science 319, 582–587 (2008).
Debenedetti, P. G. Supercooled and glassy water. J. Phys. Condens. Matter 15, R1669–R1726 (2003).
Tanaka, H. Simple view of waterlike anomalies of atomic liquids with directional bonding. Phys. Rev. B 66, 064202 (2002).
Mishima, O., Calvert, L. D. & Whalley, E. An apparent first-order transition between two amorphous phases of ice induced by pressure. Nature 314, 76–78 (1985).
Sastry, S., Debenedetti, P. G., Sciortino, F. & Stanley, H. E. Singularity-free interpretation of the thermodynamics of supercooled water. Phys. Rev. E 53, 6144–6154 (1996).
Tanaka, H. Thermodynamic anomaly and polyamorphism of water. Europhys. Lett. 50, 340–346 (2000).
Koza, M. M., Schober, H., Fischer, H. E., Hansen, T. & Fujara, F. Kinetics of the high- to low-density amorphous water transition. J. Phys. Condens. Matter 15, 321–332 (2003).
Poole, P. H., Sciortino, F., Essmann, U. & Stanley, H. E. Phase behaviour of metastable water. Nature 360, 324–328 (1992).
Xu, L. et al. Relation between the widom line and the dynamic crossover in systems with a liquid–liquid phase transition. Proc. Natl Acad. Sci. USA 102, 16558–16562 (2005).
Tanaka, H. A new scenario of the apparent fragile-to-strong transition in tetrahedral liquids: water as an example. J. Phys. Condens. Matter 15, L703–L711 (2003).
Poole, P. H., Grande, T., Angell, C. A. & McMillan, P. F. Polymorphic phase transitions in liquids and glasses. Science 275, 322–324 (1997).
Tanaka, H. General view of a liquid–liquid phase. Phys. Rev. E 62, 6968–6976 (2000).
Katayama, Y., Mizutani, T., Utsumi, W., Shimomura, O. & Yamakata, M. A first-order liquid–liquid transition in phosphorus. Nature 403, 170–173 (2000).
Sastry, S. & Angell, C. A. Liquid–liquid phase transition in supercooled silicon. Nature Mater. 2, 739–743 (2003).
Vasisht, V. V., Saw, S. & Sastry, S. Liquid–liquid critical point in supercooled silicon. Nature Phys. 7, 549–553 (2011).
Aasland, S. & McMillan, P. F. Density-driven liquid–liquid phase-separation in the system Al2O3-Y2O3 . Nature 369, 633–639 (1994).
Greaves, G. N. et al. Detection of first-order liquid/liquid phase transitions in yttrium oxide-aluminum oxide melts. Science 322, 566–570 (2008).
Cohen, I. et al. A low temperature amorphous phase in fragile glass-forming substance. J. Phys. Chem 100, 8518–8526 (1996).
Tanaka, H., Kurita, R. & Mataki, H. Liquid–liquid transition in the molecular liquid triphenyl phosphite. Phys. Rev. Lett. 92, 025701 (2004).
Kurita, R. & Tanaka, H. Critical-like phenomena associated with liquid–liquid transition in a molecular liquid. Science 306, 845–848 (2004).
Kurita, R. & Tanaka, H. On the abundance and general nature of the liquid–liquid phase transition in molecular systems. J. Phys. Condens. Matter 17, L293–L302 (2005).
Kurita, R., Murata, K. & Tanaka, H. Control of fluidity and miscibility of a binary liquid mixture by the liquid–liquid transition. Nature Mater. 7, 647–652 (2008).
Murata, K. & Tanaka, H. Surface-wetting effects on the liquid–liquid transition of a single-component molecular liquid. Nature Commun. 1, 16 (2010).
Hedoux, A., Guinet, Y., Descamps, M. & Benabou, A. Raman scattering investigation of the glaciation process in triphenyl phosphite. J. Phys. Chem. B 104, 11774–11780 (2000).
Liu, L., Chen, S. H., Faraone, A., Yen, C. W. & Mou, C. Y. Pressure dependence of fragile-to-strong transition and a possible second critical point in supercooled confined water. Phys. Rev. Lett. 95, 117802 (2005).
Mallamace, F. et al. Transport properties of supercooled confined water. Eur. Phys. J. Special Topics 161, 19–33 (1008).
Doster, W. et al. Dynamical transition of protein-hydration water. Phys. Rev. Lett. 104, 098101 (2010).
Mancinelli, R. The effect of confinement on water structure. J. Phys. Condens. Matter 22, 404213 (2010).
Morineau, D. & Alba-Simionesco, C. Does molecular self-association survive in nanochannels? J. Phys. Chem. Lett. 1, 1155–1159 (2010).
Findenegg, G. H., Jähnert, S., Akcakayiran, D. & Schreiber, A. Freezing and melting of water confined in silica nanopores. ChemPhysChem. 9, 2651–2659 (2008).
Davis-Searles, P. R., Saunders, A. J., Erie, D. A., Winzor, D. J. & Pielak, G. J. Interpreting the effects of small uncharged solutes on protein-folding equilibria. Annu. Rev. Biophys. Biomol. Struct. 30, 271–306 (2001).
Chatterjee, S. & Debenedetti, P. G. Fluid-phase behavior of binary mixtures in which one component can have two critical point. J. Chem. Phys. 124, 154503 (2006).
Mishima, O. Application of polyamorphism in water to spontaneous crystallization of emulsified LiCl–H2O solution. J. Chem. Phys. 123, 154506 (2005).
Mishima, O. Phase separation in dilute LiCl–H2O solution related to the polyamorphism of liquid water. J. Chem. Phys. 126, 244507 (2007).
Le, L. & Molinero, V. Nanophase segregation in supercooled aqueous solutions and their glasses driven by the polyamorphism of water. J. Phys. Chem. A 115, 5900–5907 (2011).
Angell, C. A., Borick, S. & Grabow, M. Glass transition and first order liquid-metal-to-semiconductor transitions in 4-5-6 covalent systems. J. Non-Cryst. Solids 205–207, 463–471 (1996).
Ito, K., Moynihan, C. T. & Angell, C. A. Thermodynamic determination of fragility in liquids and a fragile-to-strong liquid transition in water. Nature 398, 492–495 (1999).
Kurita, R. & Tanaka, H. Control of the fragility of a glass-forming liquid using the liquid–liquid phase transition. Phys. Rev. Lett. 95, 065701 (2005).
Kurita, R. & Tanaka, H. Kinetics of the liquid–liquid transition of triphenyl phosphite. Phys. Rev. B 73, 104202 (2006).
Suzuki, Y. & Tominaga, Y. Polarized Raman spectroscopic study of relaxed high density amorphous ices under pressure. J. Chem. Phys. 133, 164508 (2010).
Mudalige, A. & Pemberton, J. E. Raman spectroscopy of glycerol/D2O solutions. Vib. Spectrosc. 45, 27–35 (2007).
Hansen, T. C., Koza, M. M. & Kuhs, W. F. Formation and annealing of cubic ice: I. Modelling of stacking fault. J. Phys. Condens. Matter 20, 285104 (2008).
Johari, G. P. Water’s size-dependent freezing to cubic ice. J. Chem. Phys. 122, 194504 (2005).
Jenniskens, P. & Blake, D. F. Crystallization of amorphous water ice in the solar system. Astrophys. J. 473, 1104–1113 (1996).
Moore, E. B. & Molinero, V. Ice crystallization in water’s ‘no-man’s land’. J. Chem. Phys. 132, 244504 (2010).
Kivelson, D. & Tarjus, G. H2O below 277 K: A novel picture. J. Phys. Chem. B 105, 6620–6627 (2001).
Kobayashi, M. & Tanaka, H. Possible link of the V-shaped phase diagram to the glass-forming ability and fragility in a water-salt mixture. Phys. Rev. Lett. 106, 125703 (2011).
Mishima, O. Volume of supercooled water under pressure and liquid–liquid critical point. J. Chem. Phys. 133, 144503 (2010).
Miyata, K. & Kanno, H. Supercooling behavior of aqueous solutions of alcohols and saccharides. J. Mol. Liq. 119, 189–193 (2005).
Rasmussen, D. H. & MacKenzie, A. P. Water Structure at the Water-Polymer Interface (Plenum, 1972).
Olsen, N. B. Scaling of β-relaxation in the equilibrium liquid state of sorbitol. J. Non-Cryst. Solids 235–237, 399–405 (1998).
Dyre, J. C. & Olsen, N. B. Minimal model for beta relaxation in viscous liquids. Phys. Rev. Lett. 91, 155703 (2003).
Lane, L. B. Freezing points of glycerol and its aqueous solutions. Ind. Eng. Chem. 17, 924 (1925).
Acknowledgements
This work was partially supported by a grant-in-aid from the Ministry of Education, Culture, Sports, Science and Technology, Japan.
Author information
Authors and Affiliations
Contributions
H.T. conceived the research, K.M. performed experiments and analysed data, and K.M. and H.T. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 1470 kb)
Rights and permissions
About this article
Cite this article
Murata, Ki., Tanaka, H. Liquid–liquid transition without macroscopic phase separation in a water–glycerol mixture. Nature Mater 11, 436–443 (2012). https://doi.org/10.1038/nmat3271
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat3271
This article is cited by
-
Problems of human spermatozoa cryopreservation: research methods, solutions
Biophysical Reviews (2023)
-
Partial freezing of rat livers extends preservation time by 5-fold
Nature Communications (2022)
-
Designing cryo-enzymatic reactions in subzero liquid water by lipidic mesophase nanoconfinement
Nature Nanotechnology (2021)
-
Pressure Induced Liquid-to-Liquid Transition in Zr-based Supercooled Melts and Pressure Quenched Glasses
Scientific Reports (2017)
-
Solidification of Immiscible Alloys: A Review
Acta Metallurgica Sinica (English Letters) (2017)