Abstract
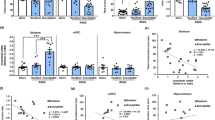
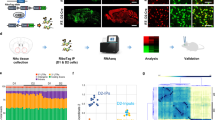
Depression induces structural and functional synaptic plasticity in brain reward circuits, although the mechanisms promoting these changes and their relevance to behavioral outcomes are unknown. Transcriptional profiling of the nucleus accumbens (NAc) for Rho GTPase–related genes, which are known regulators of synaptic structure, revealed a sustained reduction in RAS-related C3 botulinum toxin substrate 1 (Rac1) expression after chronic social defeat stress. This was associated with a repressive chromatin state surrounding the proximal promoter of Rac1. Inhibition of class 1 histone deacetylases (HDACs) with MS-275 rescued both the decrease in Rac1 transcription after social defeat stress and depression-related behavior, such as social avoidance. We found a similar repressive chromatin state surrounding the RAC1 promoter in the NAc of subjects with depression, which corresponded with reduced RAC1 transcription. Viral-mediated reduction of Rac1 expression or inhibition of Rac1 activity in the NAc increases social defeat–induced social avoidance and anhedonia in mice. Chronic social defeat stress induces the formation of stubby excitatory spines through a Rac1-dependent mechanism involving the redistribution of synaptic cofilin, an actin-severing protein downstream of Rac1. Overexpression of constitutively active Rac1 in the NAc of mice after chronic social defeat stress reverses depression-related behaviors and prunes stubby spines. Taken together, our data identify epigenetic regulation of RAC1 in the NAc as a disease mechanism in depression and reveal a functional role for Rac1 in rodents in regulating stress-related behaviors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Krishnan, V. & Nestler, E.J. Linking molecules to mood: new insight into the biology of depression. Am. J. Psychiatry 167, 1305–1320 (2010).
Greenberg, P.E. et al. The economic burden of depression in the United States: how did it change between 1990 and 2000? J. Clin. Psychiatry 64, 1465–1475 (2003).
Kessler, R.C., Chiu, W.T., Demler, O., Merikangas, K.R. & Walters, E.E. Prevalence, severity, and comorbidity of 12-month DSM-IV disorders in the National Comorbidity Survey Replication. Arch. Gen. Psychiatry 62, 617–627 (2005).
Tissot, R. The common pathophysiology of monaminergic psychoses: a new hypothesis. Neuropsychobiology 1, 243–260 (1975).
Smith, R.S. The macrophage theory of depression. Med. Hypotheses 35, 298–306 (1991).
Duman, R.S., Heninger, G.R. & Nestler, E.J. A molecular and cellular theory of depression. Arch. Gen. Psychiatry 54, 597–606 (1997).
Palucha, A. & Pilc, A. The involvement of glutamate in the pathophysiology of depression. Drug News Perspect. 18, 262–268 (2005).
Christoffel, D.J., Golden, S.A. & Russo, S.J. Structural and synaptic plasticity in stress-related disorders. Rev. Neurosci. 22, 535–549 (2011).
Capuron, L. & Miller, A.H. Immune system to brain signaling: neuropsychopharmacological implications. Pharmacol. Ther. 130, 226–238 (2011).
Manji, H.K. et al. Enhancing neuronal plasticity and cellular resilience to develop novel, improved therapeutics for difficult-to-treat depression. Biol. Psychiatry 53, 707–742 (2003).
Vidal, R. et al. New strategies in the development of antidepressants: towards the modulation of neuroplasticity pathways. Curr. Pharm. Des. 17, 521–533 (2011).
Kang, H.J. et al. Decreased expression of synapse-related genes and loss of synapses in major depressive disorder. Nat. Med. 18, 1413–1417 (2012).
Christoffel, D.J. et al. IκB kinase regulates social defeat stress-induced synaptic and behavioral plasticity. J. Neurosci. 31, 314–321 (2011).
Christoffel, D.J. et al. Effects of inhibitor of κB kinase activity in the nucleus accumbens on emotional behavior. Neuropsychopharmacology 37, 2615–2623 (2012).
Nestler, E.J. & Carlezon, W.A. Jr. The mesolimbic dopamine reward circuit in depression. Biol. Psychiatry 59, 1151–1159 (2006).
Krishnan, V. et al. Molecular adaptations underlying susceptibility and resistance to social defeat in brain reward regions. Cell 131, 391–404 (2007).
Covington, H.E. III et al. Antidepressant actions of histone deacetylase inhibitors. J. Neurosci. 29, 11451–11460 (2009).
Wei, Q. et al. Early-life forebrain glucocorticoid receptor overexpression increases anxiety behavior and cocaine sensitization. Biol. Psychiatry 71, 224–231 (2012).
Andrus, B.M. et al. Gene expression patterns in the hippocampus and amygdala of endogenous depression and chronic stress models. Mol. Psychiatry 17, 49–61 (2012).
Tolias, K.F., Duman, J.G. & Um, K. Control of synapse development and plasticity by Rho GTPase regulatory proteins. Prog. Neurobiol. 94, 133–148 (2011).
Kiraly, D.D., Eipper-Mains, J.E., Mains, R.E. & Eipper, B.A. Synaptic plasticity, a symphony in GEF. ACS Chem. Neurosci. 1, 348–365 (2010).
Berton, O. et al. Essential role of BDNF in the mesolimbic dopamine pathway in social defeat stress. Science 311, 864–868 (2006).
Nestler, E.J. & Hyman, S.E. Animal models of neuropsychiatric disorders. Nat. Neurosci. 13, 1161–1169 (2010).
Golden, S.A., Covington, H.E. III, Berton, O. & Russo, S.J. A standardized protocol for repeated social defeat stress in mice. Nat. Protoc. 6, 1183–1191 (2011).
Wilkinson, M.B. et al. Imipramine treatment and resiliency exhibit similar chromatin regulation in the mouse nucleus accumbens in depression models. J. Neurosci. 29, 7820–7832 (2009).
Kumar, A. et al. Chromatin remodeling is a key mechanism underlying cocaine-induced plasticity in striatum. Neuron 48, 303–314 (2005).
Feng, J. et al. Cocaine induced transcriptome and epigenome changes in mouse nucleus accumbens. Soc. Neurosci. Abstr. 458.12/T7 (2012).
Dietz, D.M. et al. Rac1 is essential in cocaine-induced structural plasticity of nucleus accumbens neurons. Nat. Neurosci. 15, 891–896 (2012).
Jiang, Y., Matevossian, A., Huang, H.-S., Straubhaar, J. & Akbarian, S. Isolation of neuronal chromatin from brain tissue. BMC Neurosci. 9, 42 (2008).
Chen, L., Melendez, J., Campbell, K., Kuan, C.Y. & Zheng, Y. Rac1 deficiency in the forebrain results in neural progenitor reduction and microcephaly. Dev. Biol. 325, 162–170 (2009).
Pontrello, C.G. et al. Cofilin under control of β-arrestin-2 in NMDA-dependent dendritic spine plasticity, long-term depression (LTD), and learning. Proc. Natl. Acad. Sci. USA 109, E442–E451 (2012).
Bongmba, O.Y., Martinez, L., Elhardt, M., Butler, K. & Tejada-Simon, M. Modulation of dendritic spines and synaptic function by Rac1: a possible link to Fragile X syndrome pathology. Brain Res. 1399, 79–95 (2011).
Chen, L.Y. et al. Physiological activation of synaptic Rac1 PAK (p-21 activated kinase) signaling is defective in a mouse model of fragile X syndrome. J. Neurosci. 30, 10977–10984 (2010).
Hayashi-Takagi, A. et al. Disrupted-in-Schizophrenia 1 (DISC1) regulates spines of the glutamate synapse via Rac1. Nat. Neurosci. 13, 327–332 (2010).
Golden, S.A. & Russo, S.J. Mechanisms of psychostimulant-induced structural plasticity. Cold Spring Harb. Perspect. Med. 2, a011957 (2012).
de Curtis, I. Functions of Rac GTPases during neuronal development. Dev. Neurosci. 30, 47–58 (2008).
Koh, C.-G. Rho GTPases and their regulators in neuronal functions and development. Neurosignals 15, 228–237 (2006).
Martinez, L.A. & Tejada-Simon, M. Pharmacological inactivation of the small GTPase Rac1 impairs long-term plasticity in the mouse hippocampus. Neuropharmacology 61, 305–312 (2011).
Holtmaat, A. & Svoboda, K. Experience-dependent structural synaptic plasticity in the mammalian brain. Nat. Rev. Neurosci. 10, 647–658 (2009).
Yoshihara, Y., De Roo, M. & Muller, D. Dendritic spine formation and stabilization. Curr. Opin. Neurobiol. 19, 146–153 (2009).
Luo, L. et al. Differential effects of the Rac GTPase on Purkinje cell axons and dendritic trunks and spines. Nature 379, 837–840 (1996).
Nakayama, A.Y., Harms, M.B. & Luo, L. Small GTPases Rac and Rho in the maintenance of dendritic spines and branches in hippocampal pyramidal neurons. J. Neurosci. 20, 5329–5338 (2000).
Tashiro, A. & Yuste, R. Regulation of dendritic spine motility and stability by Rac1 and Rho kinase: evidence for two forms of spine motility. Mol. Cell. Neurosci. 26, 429–440 (2004).
Kuhn, T.B. et al. Regulating actin dynamics in neuronal growth cones by ADF/cofilin and rho family GTPases. J. Neurobiol. 44, 126–144 (2000).
Rex, C.S. et al. Different Rho GTPase–dependent signaling pathways initiate sequential steps in the consolidation of long-term potentiation. J. Cell Biol. 186, 85–97 (2009).
Dubos, A. et al. Alteration of synaptic network dynamics by the intellectual disability protein PAK3. J. Neurosci. 32, 519–527 (2012).
Li, N. et al. mTOR-dependent synapse formation underlies the rapid antidepressant effects of NMDA antagonists. Science 329, 959–964 (2010).
LaPlant, Q. et al. Dnmt3a regulates emotional behavior and spine plasticity in the nucleus accumbens. Nat. Neurosci. 13, 1137–1143 (2010).
Stolzenburg, S., Bilsland, A., Keith, W.N. & Rots, M.G. Modulation of gene expression using zinc finger–based artificial transcription factors. Methods Mol. Biol. 649, 117–132 (2010).
Russo, S.J., Murrough, J.W., Han, M.H., Charney, D.S. & Nestler, E.J. Neurobiology of resilience. Nat. Neurosci. 15, 1475–1484 (2012).
Tsankova, N. et al. Sustained hippocampal chromatin regulation in a mouse model of depression and antidepressant action. Nat. Neurosci. 9, 519–544 (2006).
Stan, A.D. et al. Human postmortem tissue: what quality markers matter? Brain Res. 1123, 1–11 (2006).
Radley, J.J. et al. Repeated stress induces dendritic spine loss in the rat medial prefrontal cortex. Cereb. Cortex 16, 313–320 (2006).
Rodriguez, A., Ehlenberger, D.B., Dickstein, D.L., Hof, P.R. & Wearne, S.L. Automated three-dimensional detection and shape classification of dendritic spines from fluorescence microscopy images. PLoS One 3, e1997 (2008).
Russo, S.J. et al. Nuclear factor κB signaling regulates neuronal morphology and cocaine reward. J. Neurosci. 29, 3529–3537 (2009).
Acknowledgements
We thank Y. Zeng (University of Cincinnati) for providing Rac1flox/flox mice. This research was supported by US National Institute of Mental Health grant R01 MH090264, the Johnson and Johnson International Mental Health Research Organization Rising Star Award (S.J.R.), US National Institute of Mental Health grants P50 MH66172 and P50MH096890 (C.A.T.) and US National Institutes of Health grant T32 GM062754 (K.D.).
Author information
Authors and Affiliations
Contributions
S.A.G. and S.J.R. contributed to study design, data collection, analysis and writing. D.J.C., M.H., G.E.H., J.M., K.D., M.E.C., C.D., E.R., A.J.R., P.J.K. and J.L.A. contributed to data collection. J.G.-M. contributed to data analysis. R.L.N. contributed to viral design and packaging. G.T., S.G. and C.A.T. collected human brain samples and contributed to data analysis.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–10 and Supplementary Tables 1–6 (PDF 6780 kb)
Rights and permissions
About this article
Cite this article
Golden, S., Christoffel, D., Heshmati, M. et al. Epigenetic regulation of RAC1 induces synaptic remodeling in stress disorders and depression. Nat Med 19, 337–344 (2013). https://doi.org/10.1038/nm.3090
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3090
This article is cited by
-
Circulating myeloid-derived MMP8 in stress susceptibility and depression
Nature (2024)
-
α-MSH-catabolic enzyme prolylcarboxypeptidase in nucleus accumbens shell ameliorates stress susceptibility in mice through regulating synaptic plasticity
Acta Pharmacologica Sinica (2023)
-
Aging impairs recovery from stress-induced depression in male rats possibly by alteration of microRNA-101 expression and Rac1/RhoA pathway in the prefrontal cortex
Biogerontology (2023)
-
S-Ketamine Exerts Antidepressant Effects by Regulating Rac1 GTPase Mediated Synaptic Plasticity in the Hippocampus of Stressed Rats
Cellular and Molecular Neurobiology (2023)
-
Neuroadaptations and TGF-β signaling: emerging role in models of neuropsychiatric disorders
Molecular Psychiatry (2022)