Abstract
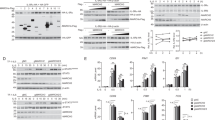
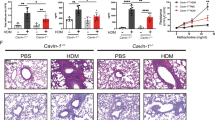
Allergic airway inflammation is associated with activation of innate immune pathways by allergens. Acute exacerbations of asthma are commonly associated with rhinovirus infection. Here we show that, after exposure to house dust mite (HDM) or rhinovirus infection, the E3 ubiquitin ligase midline 1 (MID1) is upregulated in mouse bronchial epithelium. HDM regulates MID1 expression in a Toll-like receptor 4 (TLR4)– and tumor necrosis factor–related apoptosis-inducing ligand (TRAIL)-dependent manner. MID1 decreases protein phosphatase 2A (PP2A) activity through association with its catalytic subunit PP2Ac. siRNA-mediated knockdown of MID1 or pharmacological activation of PP2A using a nonphosphorylatable FTY720 analog in mice exposed to HDM reduces airway hyperreactivity and inflammation, including the expression of interleukin-25 (IL-25), IL-33 and CCL20, IL-5 and IL-13 release, nuclear factor (NF)κB activity, p38 mitogen-activated protein kinase (MAPK) phosphorylation, accumulation of eosinophils, T lymphocytes and myeloid dendritic cells, and the number of mucus-producing cells. MID1 inhibition also limited rhinovirus-induced exacerbation of allergic airway disease. We found that MID1 was upregulated in primary human bronchial epithelial cells upon HDM or rhinovirus exposure, and this correlated with TRAIL and CCL20 expression. Together, these findings identify a key role of MID1 in allergic airway inflammation and links innate immune pathway activation to the development and exacerbation of asthma.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
References
Lemanske, R.F. Jr. & Busse, W.W. Asthma: clinical expression and molecular mechanisms. J. Allergy Clin. Immunol. 125, S95–S102 (2010).
Holgate, S.T., Roberts, G., Arshad, H.S., Howarth, P.H. & Davies, D.E. The role of the airway epithelium and its interaction with environmental factors in asthma pathogenesis. Proc. Am. Thorac. Soc. 6, 655–659 (2009).
Lambrecht, B.N. & Hammad, H. Biology of lung dendritic cells at the origin of asthma. Immunity 31, 412–424 (2009).
Weckmann, M. et al. Critical link between TRAIL and CCL20 for the activation of TH2 cells and the expression of allergic airway disease. Nat. Med. 13, 1308–1315 (2007).
Wills-Karp, M. et al. Interleukin-13: central mediator of allergic asthma. Science 282, 2258–2261 (1998).
Kuperman, D.A. et al. Direct effects of interleukin-13 on epithelial cells cause airway hyperreactivity and mucus overproduction in asthma. Nat. Med. 8, 885–889 (2002).
Mattes, J. et al. IL-13 induces airways hyperreactivity independently of the IL-4R alpha chain in the allergic lung. J. Immunol. 167, 1683–1692 (2001).
Kuperman, D., Schofield, B., Wills-Karp, M. & Grusby, M.J. Signal transducer and activator of transcription factor 6 (Stat6)-deficient mice are protected from antigen-induced airway hyperresponsiveness and mucus production. J. Exp. Med. 187, 939–948 (1998).
Johnston, S.L. et al. Community study of role of viral infections in exacerbations of asthma in 9–11 year old children. Br. Med. J. 310, 1225–1229 (1995).
Kusel, M.M. et al. Role of respiratory viruses in acute upper and lower respiratory tract illness in the first year of life: a birth cohort study. Pediatr. Infect. Dis. J. 25, 680–686 (2006).
Jackson, D.J. et al. Wheezing rhinovirus illnesses in early life predict asthma development in high-risk children. Am. J. Respir. Crit. Care Med. 178, 667–672 (2008).
Wark, P.A. et al. Asthmatic bronchial epithelial cells have a deficient innate immune response to infection with rhinovirus. J. Exp. Med. 201, 937–947 (2005).
Contoli, M. et al. Role of deficient type III interferon-λ production in asthma exacerbations. Nat. Med. 12, 1023–1026 (2006).
Holgate, S.T. A look at the pathogenesis of asthma: the need for a change in direction. Discov. Med. 9, 439–447 (2010).
Crimi, E. et al. Dissociation between airway inflammation and airway hyperresponsiveness in allergic asthma. Am. J. Respir. Crit. Care Med. 157, 4–9 (1998).
Phipps, S. et al. Toll/IL-1 signaling is critical for house dust mite-specific TH1 and TH2 responses. Am. J. Respir. Crit. Care Med. 179, 883–893 (2009).
Hammad, H. et al. House dust mite allergen induces asthma via Toll-like receptor 4 triggering of airway structural cells. Nat. Med. 15, 410–416 (2009).
Mattes, J., Collison, A., Plank, M., Phipps, S. & Foster, P.S. Antagonism of microRNA-126 suppresses the effector function of TH2 cells and the development of allergic airways disease. Proc. Natl. Acad. Sci. USA 106, 18704–18709 (2009).
Kumar, H., Kawai, T. & Akira, S. Toll-like receptors and innate immunity. Biochem. Biophys. Res. Commun. 388, 621–625 (2009).
Fontanella, B., Russolillo, G. & Meroni, G. MID1 mutations in patients with X-linked Opitz G/BBB syndrome. Hum. Mutat. 29, 584–594 (2008).
Latta, E.J. & Golding, J.P. Regulation of PP2A activity by Mid1 controls cranial neural crest speed and gangliogenesis. Mech Dev. 128, 560–576 (2012).
Aranda-Orgillés, B. et al. Active transport of the ubiquitin ligase MID1 along the microtubules is regulated by protein phosphatase 2A. PLoS ONE 3, e3507 (2008).
McConnell, J.L. et al. α4 is a ubiquitin-binding protein that regulates protein serine/threonine phosphatase 2A ubiquitination. Biochemistry 49, 1713–1718 (2010).
Trockenbacher, A., Suckow, V., Foerster, J., Winter, J. & Krauss, S. MID1, mutation in Opitz syndrome, encodes an ubiquitin ligase that targets phophatase 2A for degradation. Nat. Genet. 29, 287–294 (2001).
Watkins, G.R. et al. Monoubiquitination promotes calpain cleavage of the protein phosphatase 2A (PP2A) regulatory subunit α4, altering PP2A stability and microtubule-associated protein phosphorylation. J. Biol. Chem. 287, 24207–24215 (2012).
Sim, A.T., Ludowyke, R.I. & Verrills, N.M. Mast cell function: regulation of degranulation by serine/threonine phosphatases. Pharmacol. Ther. 112, 425–439 (2006).
Cornell, T.T. et al. Ceramide-dependent PP2A regulation of TNFα-induced IL-8 production in respiratory epithelial cells. Am. J. Physiol. Lung Cell Mol. Physiol. 296, L849–L856 (2009).
Shanley, T.P., Vasi, N., Denenberg, A. & Wong, H.R. The serine/threonine phosphatase, PP2A: endogenous regulator of inflammatory cell signaling. J. Immunol. 166, 966–972 (2001).
Miskolci, V. et al. Okadaic acid induces sustained activation of NFκB and degradation of the nuclear IκBα in human neutrophils. Arch. Biochem. Biophys. 417, 44–52 (2003).
Liu, W. et al. Cell-specific activation profile of extracellular signal-regulated kinase 1/2, Jun N-terminal kinase, and p38 mitogen-activated protein kinases in asthmatic airways. J. Allergy Clin. Immunol. 121, 893–902.e2 (2008).
Griego, S.D., Weston, C.B., Adams, J.L., Tal-Singer, R. & Dillon, S.B. Role of p38 mitogen-activated protein kinase in rhinovirus-induced cytokine production by bronchial epithelial cells. J. Immunol. 165, 5211–5220 (2000).
Duan, W. et al. Inhaled p38α mitogen-activated protein kinase antisense oligonucleotide attenuates asthma in mice. Am. J. Respir. Crit. Care Med. 171, 571–578 (2005).
Wong, W.S. Inhibitors of the tyrosine kinase signaling cascade for asthma. Curr. Opin. Pharmacol. 5, 264–271 (2005).
Das, J. et al. A critical role for NF-κB in GATA3 expression and TH2 differentiation in allergic airway inflammation. Nat. Immunol. 2, 45–50 (2001).
Kobayashi, Y., Mercado, N., Barnes, P.J. & Ito, K. Defects of protein phosphatase 2a causes corticosteroid insensitivity in severe asthma. PLoS ONE 6, e27627 (2011).
Don, A.S. et al. Essential requirement for sphingosine kinase 2 in a sphingolipid apoptosis pathway activated by FTY720 analogues. J. Biol. Chem. 282, 15833–15842 (2007).
Francis, J.N., Sabroe, I., Lloyd, C.M., Durham, S.R. & Till, S.J. Elevated CCR6+ CD4+ T lymphocytes in tissue compared with blood and induction of CCL20 during the asthmatic late response. Clin. Exp. Immunol. 152, 440–447 (2008).
Lambrecht, B.N. et al. Myeloid dendritic cells induce TH2 responses to inhaled antigen, leading to eosinophilic airway inflammation. J. Clin. Invest. 106, 551–559 (2000).
Ehrle, C. et al. Overexpressing mouse model demonstrates the protective role of Muc5ac in the lungs. Proc. Natl. Acad. Sci. USA. 109, 16528–16533 (2012).
Xatzipsalti, M. & Papadopoulos, N.G. Cellular and animals models for rhinovirus infection in asthma. Contrib. Microbiol. 14, 33–41 (2007).
Bartlett, N.W. et al. Mouse models of rhinovirus-induced disease and exacerbation of allergic airway inflammation. Nat. Med. 14, 199–204 (2008).
Collison, A., Mattes, J., Plank, M. & Foster, P.S. Inhibition of house dust mite-induced allergic airways disease by antagonism of microRNA-145 is comparable to glucocorticoid treatment. J. Allergy Clin. Immunol. 128, 160–167 (2011).
Sim, A.T., Collins, E., Mudge, L.M. & Rostas, J.A. Developmental regulation of protein phosphatase types 1 and 2A in post-hatch chicken brain. Neurochem. Res. 23, 487–491 (1998).
Acknowledgements
This study was supported by the National Health and Medical Research Council (NH&MRC 631075 and 1011153) (J.M., P.S.F., N.V.), the Hunter Medical Research Institute (J.M., P.A.B.W., P.S.F.), the Hunter Children's Research Foundation (J.M., P.S.F., A.P.d.S.) and an NH&MRC Health Practitioner Research Fellowship to J.M. (455623). N.W.B. was supported by a project grant from Asthma UK (06-050) and S.L.J. by a Chair from Asthma UK (CH1155). This work was supported in part by MRC Centre grant G1000758 and ERC FP7 Advanced grant 233015 (to S.L.J.). We would like to thank M. Smyth, Peter MacCallum Cancer Centre, and J. Peschon, Amgen, for providing Tnfsf10−/− mice and S. Akira, Osaka University, for providing Tlr4−/− and Myd88−/− mice. We appreciate technical assistance from C. Cesar de Souza Alves, F. Eyers, J. Girkin, J. Grehan, H. MacDonald, M. Morten, K. Parsons, S. Reeves, L. Sokulsky and the staff from the animal care facilities of the contributing institutes.
Author information
Authors and Affiliations
Contributions
A.C. and L.H. performed and designed mouse and cell culture experiments, analyzed data, generated figures and edited the manuscript. P.A.B.W. and M.T. performed and supervised studies on healthy subjects and subjects with asthma and performed cell culture experiments. N.V. and H.C. performed and analyzed PP2Ac measurements and immunoprecipitation and designed experiments. N.V. edited the manuscript. A.S.D. and J.C.M. synthesized AAL(S) for use as an activator of PP2A and developed the dosing regiment. N.Z. and M.E.R. coordinated and assisted in microarray array analysis. N.W.B. and S.L.J. assisted in design of experiments, provided RV1B for further propagation and cDNAs and edited the manuscript. A.P.d.S. coordinated and supervised mouse and human studies. P.S.F. supervised mouse studies, interpreted data and edited the manuscript. J.M. conceptualized, coordinated, designed and supervised mouse and human studies, interpreted and analyzed data, and drafted and edited the manuscript. All authors contributed to data discussion and revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
J.M., A.C, L.H. and P.S.F. have filed a patent application under the Patent Cooperation Treaty that relates to the described data on MID1 function in this manuscript.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7, Supplementary Table 1 and Supplementary Methods (PDF 1229 kb)
Rights and permissions
About this article
Cite this article
Collison, A., Hatchwell, L., Verrills, N. et al. The E3 ubiquitin ligase midline 1 promotes allergen and rhinovirus-induced asthma by inhibiting protein phosphatase 2A activity. Nat Med 19, 232–237 (2013). https://doi.org/10.1038/nm.3049
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3049
This article is cited by
-
Rhinovirus infection induces secretion of endothelin-1 from airway epithelial cells in both in vitro and in vivo models
Respiratory Research (2023)
-
TRIM18 is a critical regulator of viral myocarditis and organ inflammation
Journal of Biomedical Science (2022)
-
The kinase p38α functions in dendritic cells to regulate Th2-cell differentiation and allergic inflammation
Cellular & Molecular Immunology (2022)
-
Targeting protein phosphatases for the treatment of inflammation-related diseases: From signaling to therapy
Signal Transduction and Targeted Therapy (2022)
-
GITRL on dendritic cells aggravates house dust mite-induced airway inflammation and airway hyperresponsiveness by modulating CD4+ T cell differentiation
Respiratory Research (2021)