Abstract
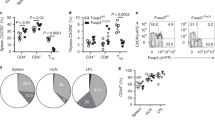
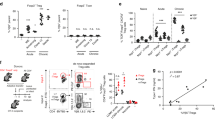
Regulatory T cells (Treg cells) have a critical role in the maintenance of immunological self-tolerance. Here we show that treatment of naive human or mouse T cells with IL-35 induced a regulatory population, which we call 'iTR35 cells', that mediated suppression via IL-35 but not via the inhibitory cytokines IL-10 or transforming growth factor-β (TGF-β). We found that iTR35 cells did not express or require the transcription factor Foxp3, and were strongly suppressive and stable in vivo. Treg cells induced the generation of iTR35 cells in an IL-35- and IL-10-dependent manner in vitro and induced their generation in vivo under inflammatory conditions in intestines infected with Trichuris muris and within the tumor microenvironment (B16 melanoma and MC38 colorectal adenocarcinoma), where they contributed to the regulatory milieu. Thus, iTR35 cells constitute a key mediator of infectious tolerance and contribute to Treg cell–mediated tumor progression. Furthermore, iTR35 cells generated ex vivo might have therapeutic utility.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Fontenot, J.D., Rasmussen, J.P., Gavin, M.A. & Rudensky, A.Y. A function for interleukin 2 in Foxp3-expressing regulatory T cells. Nat. Immunol. 6, 1142–1151 (2005).
Gavin, M.A. et al. Foxp3-dependent programme of regulatory T-cell differentiation. Nature 445, 771–775 (2007).
Hori, S., Nomura, T. & Sakaguchi, S. Control of regulatory T cell development by the transcription factor Foxp3. Science 299, 1057–1061 (2003).
Waldmann, H., Adams, E., Fairchild, P. & Cobbold, S. Infectious tolerance and the long-term acceptance of transplanted tissue. Immunol. Rev. 212, 301–313 (2006).
Selvaraj, R.K. & Geiger, T.L. Mitigation of experimental allergic encephalomyelitis by TGF-B induced Foxp3+ regulatory T lymphocytes through the induction of anergy and infectious tolerance. J. Immunol. 180, 2830–2838 (2008).
Nguyen, K.D., Vanichsarn, C. & Nadeau, K.C. Impaired IL-10-dependent induction of tolerogenic dendritic cells by CD4+CD25hiCD127lo/− natural regulatory T cells in human allergic asthma. Am. J. Respir. Crit. Care Med. 180, 823–833 (2009).
Bluestone, J.A. & Abbas, A.K. Natural versus adaptive regulatory T cells. Nat. Rev. Immunol. 3, 253–257 (2003).
Shevach, E.M. From vanilla to 28 flavors: multiple varieties of T regulatory cells. Immunity 25, 195–201 (2006).
Workman, C.J., Szymczak-Workman, A.L., Collison, L.W., Pillai, M.R. & Vignali, D.A. The development and function of regulatory T cells. Cell. Mol. Life. Sci. 6, 2603–2622 (2009).
Roncarolo, M.G. et al. Interleukin-10-secreting type 1 regulatory T cells in rodents and humans. Immunol. Rev. 212, 28–50 (2006).
Verbsky, J.W. Therapeutic use of T regulatory cells. Curr. Opin. Rheumatol. 19, 252–258 (2007).
Chen, W. et al. Conversion of peripheral CD4+CD25− naive T cells to CD4+CD25+ regulatory T cells by TGF-β induction of transcription factor Foxp3. J. Exp. Med. 198, 1875–1886 (2003).
Groux, H. et al. A CD4+ T-cell subset inhibits antigen-specific T-cell responses and prevents colitis. Nature 389, 737–742 (1997).
Collison, L.W. et al. The inhibitory cytokine IL-35 contributes to regulatory T-cell function. Nature 450, 566–569 (2007).
Sakaguchi, S. Regulatory T cells: key controllers of immunologic self-tolerance. Cell 101, 455–458 (2000).
Huter, E.N. et al. TGF-β-induced Foxp3+ regulatory T cells rescue scurfy mice. Eur. J. Immunol. 38, 1814–1821 (2008).
Workman, C.J. et al. In vivo Treg suppression assays. in Regulatory T cells: Methods and Protocols (Springer-Humana, New York, in the press).
Annacker, O., Pimenta-Araujo, R., Burlen-Defranoux, O. & Bandeira, A. On the ontogeny and physiology of regulatory T cells. Immunol. Rev. 182, 5–17 (2001).
Workman, C.J. et al. Lymphocyte activation gene-3 (CD223) regulates the size of the expanding T cell population following antigen activation in vivo. J. Immunol. 172, 5450–5455 (2004).
Kohm, A.P., Carpentier, P.A., Anger, H.A. & Miller, S.D. Cutting edge: CD4+CD25+ regulatory T cells suppress antigen-specific autoreactive immune responses and central nervous system inflammation during active experimental autoimmune encephalomyelitis. J. Immunol. 169, 4712–4716 (2002).
McGeachy, M.J., Stephens, L.A. & Anderton, S.M. Natural recovery and protection from autoimmune encephalomyelitis: contribution of CD4+CD25+ regulatory cells within the central nervous system. J. Immunol. 175, 3025–3032 (2005).
Turk, M.J. et al. Concomitant tumor immunity to a poorly immunogenic melanoma is prevented by regulatory T cells. J. Exp. Med. 200, 771–782 (2004).
Zhang, P., Cote, A.L., de Vries, V.C., Usherwood, E.J. & Turk, M.J. Induction of postsurgical tumor immunity and T-cell memory by a poorly immunogenic tumor. Cancer Res. 67, 6468–6476 (2007).
Izcue, A., Coombes, J.L. & Powrie, F. Regulatory T cells suppress systemic and mucosal immune activation to control intestinal inflammation. Immunol. Rev. 212, 256–271 (2006).
Jonuleit, H. et al. Infectious tolerance: human CD25+ regulatory T cells convey suppressor activity to conventional CD4+ T helper cells. J. Exp. Med. 196, 255–260 (2002).
Milojevic, D., Nguyen, K.D., Wara, D. & Mellins, E.D. Regulatory T cells and their role in rheumatic diseases: a potential target for novel therapeutic development. Pediatr Rheumatol Online J 6, 20 (2008).
Waldmann, H. Tolerance can be infectious. Nat. Immunol. 9, 1001–1003 (2008).
Collison, L.W., Pillai, M.R., Chaturvedi, V. & Vignali, D.A. Regulatory T cell suppression is potentiated by target T cells in a cell contact, IL-35- and IL-10-dependent manner. J. Immunol. 182, 6121–6128 (2009).
D'Elia, R., Behnke, J.M., Bradley, J.E. & Else, K.J. Regulatory T cells: a role in the control of helminth-driven intestinal pathology and worm survival. J. Immunol. 182, 2340–2348 (2009).
Di Stasi, A. et al. T lymphocytes coexpressing CCR4 and a chimeric antigen receptor targeting CD30 have improved homing and antitumor activity in a Hodgkin tumor model. Blood 113, 6392–6402 (2009).
Liyanage, U.K. et al. Prevalence of regulatory T cells is increased in peripheral blood and tumor microenvironment of patients with pancreas or breast adenocarcinoma. J. Immunol. 169, 2756–2761 (2002).
North, R.J. & Bursuker, I. Generation and decay of the immune response to a progressive fibrosarcoma. I. Ly-1+2− suppressor T cells down-regulate the generation of Ly-1−2+ effector T cells. J. Exp. Med. 159, 1295–1311 (1984).
Olkhanud, P.B. et al. Breast cancer lung metastasis requires expression of chemokine receptor CCR4 and regulatory T cells. Cancer Res. 69, 5996–6004 (2009).
Zou, W. Immunosuppressive networks in the tumour environment and their therapeutic relevance. Nat. Rev. Cancer 5, 263–274 (2005).
Zou, W. Regulatory T cells, tumour immunity and immunotherapy. Nat. Rev. Immunol. 6, 295–307 (2006).
Kocak, E. et al. Combination therapy with anti-CTL antigen-4 and anti-4–1BB antibodies enhances cancer immunity and reduces autoimmunity. Cancer Res. 66, 7276–7284 (2006).
Peggs, K.S., Quezada, S.A., Chambers, C.A., Korman, A.J. & Allison, J.P. Blockade of CTLA-4 on both effector and regulatory T cell compartments contributes to the antitumor activity of anti-CTLA-4 antibodies. J. Exp. Med. 206, 1717–1725 (2009).
Barrat, F.J. et al. In vitro generation of interleukin 10-producing regulatory CD4+ T cells is induced by immunosuppressive drugs and inhibited by T helper type 1 (Th1)- and Th2-inducing cytokines. J. Exp. Med. 195, 603–616 (2002).
Kemper, C. et al. Activation of human CD4+ cells with CD3 and CD46 induces a T-regulatory cell 1 phenotype. Nature 421, 388–392 (2003).
Andersson, J. et al. CD4+ FoxP3+ regulatory T cells confer infectious tolerance in a TGF-β-dependent manner. J. Exp. Med. 205, 1975–1981 (2008).
Allan, S.E., Song-Zhao, G.X., Abraham, T., McMurchy, A.N. & Levings, M.K. Inducible reprogramming of human T cells into Treg cells by a conditionally active form of FOXP3. Eur. J. Immunol. 38, 3282–3289 (2008).
Bardel, E., Larousserie, F., Charlot-Rabiega, P., Coulomb-L′Hermine, A. & Devergne, O. Human CD4+ CD25+ Foxp3+ regulatory T cells do not constitutively express IL-35. J. Immunol. 181, 6898–6905 (2008).
Horwitz, D.A. et al. Regulatory T cells generated ex vivo as an approach for the therapy of autoimmune disease. Semin. Immunol. 16, 135–143 (2004).
Schwartz, R.H. Models of T cell anergy: is there a common molecular mechanism? J. Exp. Med. 184, 1–8 (1996).
Vignali, D.A. & Vignali, K.M. Profound enhancement of T cell activation mediated by the interaction between the TCR and the D3 domain of CD4. J. Immunol. 162, 1431–1439 (1999).
Huang, C.T. et al. Role of LAG-3 in regulatory T cells. Immunity 21, 503–513 (2004).
Rocke, D.M. & Durbin, B. Approximate variance-stabilizing transformations for gene-expression microarray data. Bioinformatics 19, 966–972 (2003).
Koch, M.A. et al. The transcription factor T-bet controls regulatory T cell homeostasis and function during type 1 inflammation. Nat. Immunol. 10, 595–602 (2009).
Mottet, C., Uhlig, H.H. & Powrie, F. Cutting edge: cure of colitis by CD4+CD25+ regulatory T cells. J. Immunol. 170, 3939–3943 (2003).
Artis, D. et al. The IL-27 receptor (WSX-1) is an inhibitor of innate and adaptive elements of type 2 immunity. J. Immunol. 173, 5626–5634 (2004).
Acknowledgements
We thank R. Blumberg and T. Kuo (Brigham and Women's Hospital) for Ebi3−/− mice; A. Rudensky (Memorial Sloan-Kettering Cancer Center) for Foxp3gfp mice; J. Ihle (St. Jude Children's Research Hospital; with permission from A. Rudensky) for Foxp3−/− mice; T. Geiger (St. Jude Children's Research Hospital) for Il10−/− mice; B. Triplett, D. Regan, M. Howard and M. McKenna (St. Louis Cord Blood Bank) for cord blood samples; D. Campana (St. Jude Children's Research Hospital) for the proprietary permeabilization buffer; A. Korman and M. Selby (Medarex–Bristol Myers Squibb) for MC38 colorectal adenocarcinoma cells; and S. Burns, H. Chi, R. Cross, K. Forbes, D. Green, G. Lennon, L. Jones, A. Krause, T. Moore, S. Morgan, A. Szymczak-Workman and K. Vignali for discussions and assistance. Supported by the National Institutes of Health (R01 AI39480 to D.A.A.V.; R01 AI61570 and R01 AI74878 to D.A.; and F32 AI072816 to L.W.C.), the Australian National Health and Medical Research Council Overseas Biomedical Fellowship Program (P.R.G.), the National Cancer Institute Comprehensive Cancer Center (CA21765 subaward to D.A.A.V.) and the American Lebanese Syrian Associated Charities (D.A.A.V.).
Author information
Authors and Affiliations
Contributions
L.W.C. designed (with help from D.A.A.V.) and did all mouse experiments, analyzed data and wrote the manuscript; V.C. did human experiments; A.L.H. (with L.W.C.) did the B16 tumor experiments; J.B. did the MC38 tumor experiments; P.R.G. infected mice with T. muris; C.G. did confocal microscopy; D.F. analyzed Affymetrix data; K.F. and S.A.B (with C.J.W.) generated and screened monoclonal antibodies to IL-35; C.J.W. coordinated the development of monoclonal anti-IL-35 and aided in figure preparation; M.L.J. generated and purified mouse Ebi3 protein for immunization and the development of monoclonal antibodies; H.-T.N. provided reagents and information; J.E.R. created and did histological analyses of Foxp3−/− mice; D.A. designed T. muris experiments and provided input on their interpretation; M.J.T. provided training for the B16 tumor model and provided input to research design and interpretation; and D.A.A.V. conceived of the research, directed the study and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
D.A.A.V. and L.W.C. have submitted a patent based on this work that is now pending. Also, D.A.A.V., L.W.C. and C.J.W. have also submitted a patent on IL-35 and are entitled to a share in net income generated from licensing of these patent rights for commercial development. M.L.J. is employed by Shenandoah Biotechnology and H.-T.N. is employed by R&D Systems.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–18 (PDF 841 kb)
Rights and permissions
About this article
Cite this article
Collison, L., Chaturvedi, V., Henderson, A. et al. IL-35-mediated induction of a potent regulatory T cell population. Nat Immunol 11, 1093–1101 (2010). https://doi.org/10.1038/ni.1952
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1952
This article is cited by
-
Regulatory T cell-mediated immunosuppression orchestrated by cancer: towards an immuno-genomic paradigm for precision medicine
Nature Reviews Clinical Oncology (2024)
-
Tumor-derived interleukin 35 mediates the dissemination of gemcitabine resistance in pancreatic adenocarcinoma
Oncogene (2024)
-
CD19+CD24highCD27+ B cell and interleukin 35 as potential biomarkers of disease activity in systemic lupus erythematosus patients
Advances in Rheumatology (2022)
-
Gut microbiota–derived metabolite 3-idoleacetic acid together with LPS induces IL-35+ B cell generation
Microbiome (2022)
-
STING-induced regulatory B cells compromise NK function in cancer immunity
Nature (2022)