Abstract
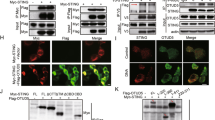
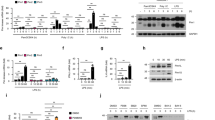
Unbalanced production of proinflammatory cytokines and type I interferons in immune responses may lead to immunopathology; thus, the mechanisms that ensure the beneficial production of proinflammatory cytokines and type I interferons are of particular importance. Here we demonstrate that the phosphatase SHP-1 negatively regulated Toll-like receptor–mediated production of proinflammatory cytokines by inhibiting activation of the transcription factor NF-κB and mitogen-activated protein kinase. Simultaneously, SHP-1 increased the production of type I interferon mediated by Toll-like receptors and the helicase RIG-I by directly binding to and inhibiting activation of the kinase IRAK1. Our data demonstrate that SHP-1 contributes to immune homeostasis by balancing the production of proinflammatory cytokines and type I interferons in the innate immune response.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
Change history
22 December 2010
In the version of this article initially published, the actin loading control in Figure 6b (left) was incorrect. The correct figure is provided here. The error has been corrected in the HTML and PDF versions of the article.
References
Barton, G.M. & Medzhitov, R. Toll-like receptor signaling pathways. Science 300, 1524–1525 (2003).
Takeda, K., Kaisho, T. & Akira, S. Toll-like receptors. Annu. Rev. Immunol. 21, 335–376 (2003).
Liew, F.Y., Xu, D., Brint, E.K. & O'Neill, L.A. Negative regulation of Toll-like receptor-mediated immune responses. Nat. Rev. Immunol. 5, 446–458 (2005).
Hoebe, K. et al. Identification of Lps2 as a key transducer of MyD88-independent TIR signalling. Nature 424, 743–748 (2003).
Yamamoto, M. et al. Role of adaptor TRIF in the MyD88-independent Toll-like receptor signaling pathway. Science 301, 640–643 (2003).
Kawai, T. et al. Interferon-alpha induction through Toll-like receptors involves a direct interaction of IRF7 with MyD88 and TRAF6. Nat. Immunol. 5, 1061–1068 (2004).
Honda, K. et al. IRF-7 is the master regulator of type-I interferon-dependent immune responses. Nature 434, 772–777 (2005).
Kato, H. et al. Differential roles of MDA5 and RIG-I helicases in the recognition of RNA viruses. Nature 441, 101–105 (2006).
Kawai, T. et al. IPS-1, an adaptor triggering RIG-I- and Mda5-mediated type I interferon induction. Nat. Immunol. 6, 981–988 (2005).
Banchereau, J., Pascual, V. & Palucka, A.K. Autoimmunity through cytokine-induced dendritic cell activation. Immunity 20, 539–550 (2004).
An, H. et al. SHP-2 phosphatase negatively regulates the TRIF adaptor protein-dependent type I interferon and proinflammatory cytokine production. Immunity 25, 919–928 (2006).
Wang, N. et al. Antagonism or synergism. Role of tyrosine phosphatases SHP-1 and SHP-2 in growth factor signaling. J. Biol. Chem. 281, 21878–21883 (2006).
Neel, B.G., Gu, H. & Pao, L. The 'Shp'ing news: SH2 domain-containing tyrosine phosphatases in cell signaling. Trends Biochem. Sci. 28, 284–293 (2003).
Rhee, S.H., Jones, B.W., Toshchakov, V., Vogel, S.N. & Fenton, M.J. Toll-like receptors 2 and 4 activate STAT1 serine phosphorylation by distinct mechanisms in macrophages. J. Biol. Chem. 278, 22506–22512 (2003).
Kawai, T. et al. Lipopolysaccharide stimulates the MyD88-independent pathway and results in activation of IFN-regulatory factor 3 and the expression of a subset of lipopolysaccharide-inducible genes. J. Immunol. 167, 5887–5894 (2001).
Fitzgerald, K.A. et al. LPS-TLR4 signaling to IRF-3/7 and NF-κB involves the toll adapters TRAM and TRIF. J. Exp. Med. 198, 1043–1055 (2003).
Honda, K. & Taniguchi, T. IRFs: master regulators of signalling by Toll-like receptors and cytosolic pattern-recognition receptors. Nat. Rev. Immunol. 6, 644–658 (2006).
Janssens, S. & Beyaert, R. Functional diversity and regulation of different interleukin-1 receptor-associated kinase (IRAK) family members. Mol. Cell 11, 293–302 (2003).
Chaudhary, A., Fresquez, T.M. & Naranjo, M.J. Tyrosine kinase Syk associates with Toll-like receptor 4 and regulates signaling in human monocytic cells. Immunol. Cell Biol. 85, 249–256 (2007).
Jefferies, C.A. et al. Bruton's tyrosine kinase is a Toll/interleukin-1 receptor domain-binding protein that participates in nuclear factor κB activation by Toll-like receptor 4. J. Biol. Chem. 278, 26258–26264 (2003).
Noubir, S., Hmama, Z. & Reiner, N.E. Dual receptors and distinct pathways mediate interleukin-1 receptor-associated kinase degradation in response to lipopolysaccharide. Involvement of CD14/TLR4, CR3, and phosphatidylinositol 3-kinase. J. Biol. Chem. 279, 25189–25195 (2004).
Dustin, L.B. et al. Expression of dominant-negative src-homology domain 2-containing protein tyrosine phosphatase-1 results in increased Syk tyrosine kinase activity and B cell activation. J. Immunol. 162, 2717–2724 (1999).
Cao, Z., Henzel, W.J. & Gao, X. IRAK: a kinase associated with the interleukin-1 receptor. Science 271, 1128–1131 (1996).
Kollewe, C. et al. Sequential autophosphorylation steps in the interleukin-1 receptor-associated kinase-1 regulate its availability as an adapter in interleukin-1 signaling. J. Biol. Chem. 279, 5227–5236 (2004).
Wu, T.R. et al. SHP-2 is a dual-specificity phosphatase involved in Stat1 dephosphorylation at both tyrosine and serine residues in nuclei. J. Biol. Chem. 277, 47572–47580 (2002).
Khaled, A.R., Butfiloski, E.J., Sobel, E.S. & Schiffenbauer, J. Functional consequences of the SHP-1 defect in motheaten viable mice: role of NF-κB. Cell. Immunol. 185, 49–58 (1998).
Uematsu, S. et al. Interleukin-1 receptor-associated kinase-1 plays an essential role for Toll-like receptor (TLR)7- and TLR9-mediated interferon-α induction. J. Exp. Med. 201, 915–923 (2005).
Zhande, R. et al. FADD negatively regulates lipopolysaccharide signaling by impairing interleukin-1 receptor-associated kinase 1-MyD88 interaction. Mol. Cell. Biol. 27, 7394–7404 (2007).
Nguyen, H. et al. IRAK-dependent phosphorylation of Stat1 on serine 727 in response to interleukin-1 and effects on gene expression. J. Interferon Cytokine Res. 23, 183–192 (2003).
Hamerman, J.A., Tchao, N.K., Lowell, C.A. & Lanier, L.L. Enhanced Toll-like receptor responses in the absence of signaling adaptor DAP12. Nat. Immunol. 6, 579–586 (2005).
Nandan, D., Lo, R. & Reiner, N.E. Activation of phosphotyrosine phosphatase activity attenuates mitogen-activated protein kinase signaling and inhibits c-FOS and nitric oxide synthase expression in macrophages infected with Leishmania donovani. Infect. Immun. 67, 4055–4063 (1999).
Zhang, Z., Jimi, E. & Bothwell, A.L. Receptor activator of NF-κB ligand stimulates recruitment of SHP-1 to the complex containing TNFR-associated factor 6 that regulates osteoclastogenesis. J. Immunol. 171, 3620–3626 (2003).
Frearson, J.A. & Alexander, D.R. The phosphotyrosine phosphatase SHP-2 participates in a multimeric signaling complex and regulates T cell receptor (TCR) coupling to the Ras/mitogen-activated protein kinase (MAPK) pathway in Jurkat T cells. J. Exp. Med. 187, 1417–1426 (1998).
Zhang, M. et al. Splenic stroma drives mature dendritic cells to differentiate into regulatory dendritic cells. Nat. Immunol. 5, 1124–1133 (2004).
Li, X. et al. Mutant cells that do not respond to interleukin-1 (IL-1) reveal a novel role for IL-1 receptor-associated kinase. Mol. Cell. Biol. 19, 4643–4652 (1999).
Acknowledgements
We thank X. Ma, S. Li and S. Xia for technical assistance; C. Kollewe (Justus-Liebig-University Giessen) for providing IRAK1 expression plasmids; K. Fitzgerald (The University of Massachusetts Medical School) for providing IRF3 and IRF7 reporter plasmids; X. Li (Cleveland Clinic Foundation) for providing IRAK1-deficient and control HEK293 cells; H. Shu (Wuhan University) for providing the RIG-I-N expression plasmid; G.-Sh. Feng (The Burnham Institute) for providing SHP-2–deficient MEFs; and W. Pan (Second Military Medical University) for providing VSV. Supported by the National Natural Science Foundation of China (30490240, 30721091 and 30731160623), the National Key Basic Research Program of China (2007CB512403) and the Shuguang Program of the Shanghai Municipal Education Committee.
Author information
Authors and Affiliations
Contributions
H.A., J.H., J.Z., W.Z., H.X., Y.Z., Y.Y. and S.L. did the experiments; H.A. and X.C. designed the experiments and wrote the paper.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 and Methods (PDF 275 kb)
Rights and permissions
About this article
Cite this article
An, H., Hou, J., Zhou, J. et al. Phosphatase SHP-1 promotes TLR- and RIG-I-activated production of type I interferon by inhibiting the kinase IRAK1. Nat Immunol 9, 542–550 (2008). https://doi.org/10.1038/ni.1604
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1604
This article is cited by
-
Lysine methyltransferase SMYD2 inhibits antiviral innate immunity by promoting IRF3 dephosphorylation
Cell Death & Disease (2023)
-
Electroacupuncture Alleviates CFA-Induced Inflammatory Pain via PD-L1/PD-1-SHP-1 Pathway
Molecular Neurobiology (2023)
-
C-type lectin receptor CLEC4A2 promotes tissue adaptation of macrophages and protects against atherosclerosis
Nature Communications (2022)
-
Phosphatases in toll-like receptors signaling: the unfairly-forgotten
Cell Communication and Signaling (2021)
-
E3 ligase Nedd4l promotes antiviral innate immunity by catalyzing K29-linked cysteine ubiquitination of TRAF3
Nature Communications (2021)