Abstract
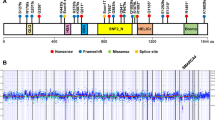
Small cell carcinoma of the ovary of hypercalcemic type (SCCOHT) is an extremely rare, aggressive cancer affecting children and young women. We identified germline and somatic inactivating mutations in the SWI/SNF chromatin-remodeling gene SMARCA4 in 69% (9/13) of SCCOHT cases in addition to SMARCA4 protein loss in 82% (14/17) of SCCOHT tumors but in only 0.4% (2/485) of other primary ovarian tumors. These data implicate SMARCA4 in SCCOHT oncogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Change history
13 June 2014
In the version of this article initially published, there were errors in the numbers of tumors reported in the text. The abstract reports SMARCA4 mutations in 75% (9/12) of tumors. The correct numbers are 69% (9/13) of tumors. The main text reports immunohistochemical analysis of 15 tumors, with 6 tumors overlapping with the 12 cases sequenced. These should have been reported as 17 tumors, 8 of which were overlapping. The errors do not affect any of the analyses or conclusions in the paper. In addition, a secondary affiliation for author Pilar Ramos was erroneously omitted and should read "School of Life Sciences, Arizona State University, Tempe, Arizona, USA." The errors have been corrected in the HTML and PDF versions of the article.
References
Young, R.H., Oliva, E. & Scully, R.E. Am. J. Surg. Pathol. 18, 1102–1116 (1994).
Florell, S.R., Bruggers, C.S., Matlak, M., Young, R.H. & Lowichik, A. Med. Pediatr. Oncol. 32, 304–307 (1999).
Shrimali, R.K., Correa, P.D. & Reed, N.S. Med. Oncol. 28, 766–770 (2011).
Estel, R., Hackethal, A., Kalder, M. & Munstedt, K. Arch. Gynecol. Obstet. 284, 1277–1282 (2011).
McCluggage, W.G., Oliva, E., Connolly, L.E., McBride, H.A. & Young, R.H. Int. J. Gynecol. Pathol. 23, 330–336 (2004).
Gamwell, L.F. et al. Orphanet J. Rare Dis. 8, 33 (2013).
Kupryjańczyk, J. et al. Pol. J. Pathol. 64, 238–246 (2013).
Martinez-Borges, A.R. et al. Pediatr. Blood Cancer 53, 1334–1336 (2009).
McDonald, J.M. et al. J. Pediatr. Surg. 47, 588–592 (2012).
Eaton, K.W., Tooke, L.S., Wainwright, L.M., Judkins, A.R. & Biegel, J.A. Pediatr. Blood Cancer 56, 7–15 (2011).
Serber, D.W. et al. PLoS ONE 7, e31346 (2012).
Wilson, B.G. et al. Mol. Cell. Biol. 34, 1136–1144 (2014).
Oike, T. et al. Cancer Res. 73, 5508–5518 (2013).
Hoffman, G.R. et al. Proc. Natl. Acad. Sci. USA 111, 3128–3133 (2014).
Kononen, J. et al. Nat. Med. 4, 844–847 (1998).
Acknowledgements
We particularly thank the families and patients who joined this institutional review board (IRB)-approved study for their critical contributions. We also thank the faculty and staff at TGen from the Macromolecular Analysis & Processing Center (G. Hostetter and J. LoBello) and the Office of Research Compliance & Quality Management (L. Nordstrom, S. Althoff and S. Buchholtz); the Children's Oncology Group for their samples; and B. Vanderhyden (University of Ottawa and Ottawa Hospital Research Institute) for BIN-67 cells. This study was supported by grants from the Marsha Rivkin Center for Ovarian Cancer Research, the Anne Rita Monahan Foundation, the Ovarian Cancer Alliance of Arizona, the Small Cell Ovarian Cancer Foundation and philanthropic support to the TGen Foundation. Further support was provided to A.N.K. and D.G.H. by the Terry Fox Research Initiative New Frontiers Program in Cancer and to A.N.K. by the Molecular Oncologic Pathology Fellowship of the Canadian Institutes of Health Research.
Author information
Authors and Affiliations
Contributions
Study concept and design: P.R., A.N.K., H.E.C., J.M.T. and D.G.H. Acquisition of data: P.R., A.N.K., M.L.R., J.J.C., B.S. and H.E.C. Sample contribution: J.P., E.D., B.A.C., J.G.P., J.H.F., S.P.A. and R.B.S.R. Data analysis: P.R., A.N.K., A.S., W.P.D.H., M.T.B., J.K., V.L.Z., B.S., T.A.M., J.M.T. and D.G.H. Bioinformatics data analysis: D.W.C., M.L.R., J.J.C., K.S., Y.Y., S.P.S., L.M.P. and M.A.M. Drafting of the manuscript: P.R., A.N.K., A.S., W.P.D.H., J.M.T. and D.G.H.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 LOH analysis in SCCOHT tumors with SMARCA4 mutations.
LOH analysis was performed using bcbio-nextgen 0.7.7a-e91123c to map reads with BWA 0.7.5a to GRCh37 and freebayes v0.9.10-11 for joint variant calling restricted to chr. 19: 10,667,750–11,554,548. The heat map shows SNPs with a call rate of 0.2. No evidence of LOH was observed.
Supplementary Figure 2 Genome view of SMARCA4 450K methylation data.
The top panel is a scatter plot displaying differential β values for all 44 SMARCA4 CpG probes in eight SCCOHT samples. Only two probes demonstrate differential hypomethylation (indicated by a red box). The upper limit of the y axis does not exceed 0.11, indicative of a lack of differential hypermethylation for any probe.
Supplementary Figure 3 SMARCA4 methylation status in SCCOHT tumors and normal fallopian tube tissues.
Vertical scatter plot of the average β values for 44 SMARCA4 450K CpG methylation probes in 8 SCCOHTs and 2 pools of normal fallopian tissue. There is no significant difference in SMARCA4 methylation between normal tissue and SCCOHT.
Supplementary Figure 4 SMARCA4 immunohistochemistry in normal premenopausal ovary and fallopian tube.
H&E-stained sections (A, C, E) and SMARCA4 immunohistochemistry (B, D, F) demonstrating strong, uniform positive nuclear staining in oocytes (B), granulosa cells of primordial (B) and secondary (D) follicles, theca cells (D) and secretory and ciliated cells throughout the fallopian tube ampulla (F) and fimbria (data not shown). In contrast, the ovarian stroma stains weakly (B) or is negative (data not shown). 400× magnification. Scale bars, 50 μm.
Supplementary information
Supplementary Text and Figures
Supplementary Note, Supplementary Figures 1–4 and Supplementary Tables 1 and 3 (PDF 1937 kb)
Supplementary Table 2
Summary of whole-genome and exome sequencing metrics. (XLS 499 kb)
Supplementary Table 4
SMARCA4 immunohistochemical analysis in primary ovarian and adnexal epithelial, sex cord stromal and germ cell tumors. (XLS 23 kb)
Rights and permissions
About this article
Cite this article
Ramos, P., Karnezis, A., Craig, D. et al. Small cell carcinoma of the ovary, hypercalcemic type, displays frequent inactivating germline and somatic mutations in SMARCA4. Nat Genet 46, 427–429 (2014). https://doi.org/10.1038/ng.2928
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.2928
This article is cited by
-
Pathological characteristics and immune microenvironment of SMARCA4-deficient undifferentiated uterine sarcoma
Diagnostic Pathology (2023)
-
Thoracic SMARCA4-deficient undifferentiated tumor
Discover Oncology (2023)
-
Pitfalls of whole exome sequencing in undefined clinical conditions with a suspected genetic etiology
Genes & Genomics (2023)
-
Alanine supplementation exploits glutamine dependency induced by SMARCA4/2-loss
Nature Communications (2023)
-
Primary cutaneous SMARCA4-deficient undifferentiated malignant neoplasm: first two cases with clinicopathologic and molecular comparison to eight visceral counterparts
Modern Pathology (2022)