Abstract
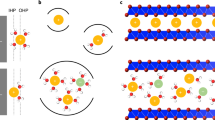
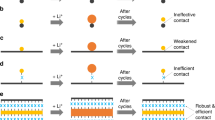
Two-dimensional (2D) materials provide slit-shaped ion diffusion channels that enable fast movement of lithium and other ions. However, electronic conductivity, the number of intercalation sites, and stability during extended cycling are also crucial for building high-performance energy storage devices. While individual 2D materials, such as graphene, show some of the required properties, none of them can offer all properties needed to maximize energy density, power density, and cycle life. Here we argue that stacking different 2D materials into heterostructured architectures opens an opportunity to construct electrodes that would combine the advantages of the individual building blocks while eliminating the associated shortcomings. We discuss characteristics of common 2D materials and provide examples of 2D heterostructured electrodes that showed new phenomena leading to superior electrochemical performance. We also consider electrode fabrication approaches and finally outline future steps to create 2D heterostructured electrodes that could greatly expand current energy storage technologies.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Tarascon, J.-M. & Armand, M. Issues and challenges facing rechargeable lithium batteries. Nature 414, 359–367 (2001).
Armand, M. & Tarascon, J.-M. Building better batteries. Nature 451, 652–657 (2008).
Larcher, D. & Tarascon, J.-M. Towards greener and more sustainable batteries for electrical energy storage. Nat. Chem. 7, 19–29 (2015).
Lee, S. & Cho, J. Critical requirements for rapid charging of rechargeable Al- and Li-ion batteries. Angew. Chem. Int. Edn 54, 9452–9455 (2015).
Whittingham, M. S. Lithium batteries and cathode materials. Chem. Rev. 104, 4271–4302 (2004).
Ebner, M., Marone, F., Stampanoni, M. & Wood, V. Visualization and quantification of electrochemical and mechanical degradation in Li-ion batteries. Science 342, 716–720 (2013).
Chhowalla, M. et al. The chemistry of two-dimensional layered transition metal dichalcogenide nanosheets. Nat. Chem. 5, 263–275 (2013).
Pumera, M., Sofer, Z. & Ambrosi, A. Layered transition metal dichalcogenides for electrochemical energy generation and storage. J. Mater. Chem. A 2, 8981–8987 (2014).
Naguib, M., Mochalin, V. N., Barsoum, M. W. & Gogotsi, Y. 25th anniversary article: MXenes: a new family of two-dimensional materials. Adv. Mater. 26, 992–1005 (2014).
He, P., Yu, H. J., Li, D. & Zhou, H. Layered lithium transition metal oxide cathodes towards high energy lithium-ion batteries. J. Mater. Chem. 22, 3680–3695 (2012).
Kubota, K., Yabuuchi, N., Yoshida, H., Dahbi, M. & Komaba, S. Layered oxides as positive electrode materials for Na-ion batteries. MRS Bull. 39, 416–422 (2014).
Kim, D. et al. Enabling sodium batteries using lithium-substituted sodium layered transition metal oxide cathodes. Adv. Energy Mater. 1, 333–336 (2011).
Stoller, M. D., Park, S., Zhu, Y., An, J. & Ruoff, R. S. Graphene-based ultracapacitors. Nano Lett. 8, 3498–3502 (2008).
Peng, L., Zhu, Y., Chen, D., Ruoff, R. S. & Yu, G. Two-dimensional materials for beyond-lithium-ion batteries. Adv. Energy Mater. 6, 1600025 (2016).
Liu, J. & Liu, X.-W. Two-dimensional nanoarchitectures for lithium storage. Adv. Mater. 24, 4097–4111 (2012).
Yan, K. et al. Ultrathin two-dimensional atomic crystals as stable interfacial layer for improvement of lithium metal anode. Nano Lett. 14, 6016–6022 (2014).
Nicolosi, V., Chhowalla, M., Kanatzidis, M. G., Strano, M. S. & Coleman, J. N. Liquid exfoliation of layered materials. Science 340, 1226419 (2013).
Xie, Y. et al. Prediction and characterization of MXene nanosheet anodes for non-lithium-ion batteries. ACS Nano 8, 9606–9615 (2014).
Wang, X. et al. Atomic-scale recognition of surface structure and intercalation mechanism of Ti3C2X. J. Am. Chem. Soc. 137, 2715–2721 (2015).
Brezesinski, T., Wang, J., Tolbert, S. H. & Dunn, B. Ordered mesoporous α-MoO3 with iso-oriented nanocrystalline walls for thin-film pseudocapacitors. Nat. Mater. 9, 146–151 (2010).
Bruce, P. G., Scrosati, B. & Tarascon, J.-M. Nanomaterials for rechargeable lithium batteries. Angew. Chem. Int. Edn 47, 2930–2946 (2008).
Lukatskaya, M. R. et al. Cation intercalation and high volumetric capacitance of two-dimensional titanium carbide. Science 341, 1502–1505 (2013).
Kondrat, S., Wu, P., Qiao, R. & Kornyshev, A. A. Accelerating charging dynamics in subnanometre pores. Nat. Mater. 13, 387–393 (2014).
Dimitrakakis, G. K., Tylianakis, E. & Froudakis, G. E. Pillared graphene: a new 3D network nanostructure for enhanced hydrogen storage. Nano Lett. 8, 3166–3170 (2008).
Anasori, B., Lukatskaya, M. R. & Gogotsi, Y. 2D metal carbides and nitrides (MXenes) for energy storage. Nat. Rev. Mater. 2, 16098 (2016).
Come, J. et al. Controlling the actuation properties of MXene paper electrodes upon cation intercalation. Nano Energy 17, 27–35 (2015).
Tepavcevic, S. et al. Nanostructured layered cathode for rechargeable Mg-ion batteries. ACS Nano 9, 8194–8205 (2015).
Zhao, Y. et al. Stable alkali metal ion intercalation compounds as optimized metal oxide nanowire cathodes for lithium batteries. Nano Letters 15, 2180–2185 (2015).
David, L., Bhandavat, R. & Singh, G. MoS2/graphene composite paper for sodium-ion battery electrodes. ASC Nano 8, 1759–1770 (2014).
Zhao, C. Y. et al. Self-assembly-induced alternately stacked single-layer MoS2 and N-doped graphene: a novel van der Waals heterostructure for lithium-ion batteries. ACS Appl. Mater. Inter. 8, 2372–2379 (2016).
Xie, X., Ao, Z., Su, D., Zhang, J. & Wang, G. MoS2/graphene composite anodes with enhanced performance for sodium-ion batteries: the role of the two-dimensional heterointerface. Adv. Funct. Mater. 25, 1393–1403 (2015).
Fang, X. P. et al. Lithium storage performance in ordered mesoporous MoS2 electrode material. Microporous Mesoporous Mater. 151, 418–423 (2012).
Liu, H. et al. Highly ordered mesoporous MoS2 with expanded spacing of the (002) crystal plane for ultrafast lithium ion storage. Adv. Energy Mater. 2, 970–975 (2012).
Liang, Y. et al. Interlayer-expanded molybdenum disulfide nanocomposites for electrochemical magnesium storage. Nano Letters 15, 2194–2202 (2015).
Oakes, L. et al. Interface strain in vertically stacked two-dimensional heterostructured carbon-MoS2 nanosheets controls electrochemical reactivity. Nat. Commun. 7, 11796 (2016).
Geim, A. K. & Grigorieva, I. V. Van der Waals heterostructures. Nature 499, 419–425 (2013).
Withers, F. et al. Light-emitting diodes by band-structure engineering in van der Waals heterostructures. Nat. Mater. 14, 301–306 (2015).
Zhou, K.-G. et al. Raman modes of MoS2 used as fingerprint of van der Waals interactions in 2D crystal-based heterostructures. ACS Nano 8, 9914–9924 (2014).
Srivastava, S. & Kotov, N. A. Composite layer-by-layer (LBL) assembly with inorganic nanoparticles and nanowires. Acc. Chem. Res. 41, 1831–1841 (2008).
Hammond, P. T. Engineering materials layer-by-layer: challenges and opportunities in multilayer assembly. Am. Inst. Chem. Eng. J. 57, 2928–2940 (2011).
Yang, X., Cheng, C., Wang, Y., Qiu, L. & Li, D. Liquid-mediated dense integration of graphene materials for compact capacitive energy storage. Science 341, 534–537 (2013).
Wang, X., Weng, Q., Yang, Y., Bando, Y. & Golberg, D. Hybrid two-dimensional materials in rechargeable battery applications and their microscopic mechanisms. Chem. Soc. Rev. 45, 4042–4073 (2016).
Lukatskaya, M. R., Dunn, B. & Gogotsi, Y. Multidimensional materials and device architectures for future hybrid energy storage. Nat. Commun. 7, 12647 (2016).
Xu, K. et al. Charging/discharging dynamics in two-dimensional ditanium carbide (MXene) slit nanopore: insights from molecular dynamic study. Electrochimica Acta 196, 75–83 (2016).
Withers, F. et al. Heterostructures produced from nanosheet-based inks. Nano Lett. 14, 3987–3992 (2014).
Zhao, M.-Q. et al. 2D titanium carbide and transition metal oxides hybrid electrodes for Li-ion storage. Nano Energy 30, 603–613 (2016).
Xie, X. et al. Porous heterostructured MXene/carbon nanotube composite paper with high volumetric capacity for sodium-based energy storage devices. Nano Energy 26, 513–523 (2016).
Backes, C. et al. Guidelines for exfoliation, characterization and processing of layered materials produced by liquid exfoliation. Chem. Mater. 29, 243–255 (2017).
Xu, Y. et al. Holey graphene frameworks for highly efficient capacitive energy storage. Nat. Commun. 5, 4554 (2014).
Xu, Y. et al. Solution processable holey graphene oxide and its derived macrostructures for high-performance supercapacitors. Nano Lett. 15, 4605–4610 (2015).
Bonaccorso, F. et al. Graphene, related two-dimensional crystals, and hybrid systems for energy conversion and storage. Science 347, 1246501 (2015).
Lin, D. et al. Layered reduced graphene oxide with nanoscale interlayer gaps as a stable host for lithium metal anodes. Nat. Nanotechnol. 11, 626–632 (2016).
Wee, A. T. S., Hersam, M. C., Chhowalla, M. & Gogotsi, Y. An update from Flatland. ACS Nano 10, 8121–8123 (2016).
Wang, X. et al. Pseudocapacitance of MXene nanosheets for high-power sodium-ion hybrid capacitors. Nat. Commun. 6, 6544 (2015).
Fukano, M. et al. Vertically oriented propylene carbonate molecules and tetraethyl ammonium ions in carbon slit pores. J. Phys. Chem. C 117, 5752–5757 (2013).
Acknowledgements
Y.G. acknowledges funding from the Fluid Interface Reactions, Structures and Transport (FIRST) Center, an Energy Frontier Research Center funded by the US Department of Energy, Office of Science, Office of Basic Energy Sciences.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Pomerantseva, E., Gogotsi, Y. Two-dimensional heterostructures for energy storage. Nat Energy 2, 17089 (2017). https://doi.org/10.1038/nenergy.2017.89
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/nenergy.2017.89
This article is cited by
-
MXenes for multispectral electromagnetic shielding
Nature Reviews Electrical Engineering (2024)
-
Metal telluride nanosheets by scalable solid lithiation and exfoliation
Nature (2024)
-
Self-supporting multi-functional two-dimensional nanofilms for flexible perceptual devices: review
Journal of Materials Science: Materials in Electronics (2024)
-
MXene/graphene oxide nanocomposites for friction and wear reduction of rough steel surfaces
Scientific Reports (2023)
-
In-situ atomic level observation of the strain response of graphene lattice
Scientific Reports (2023)