Abstract
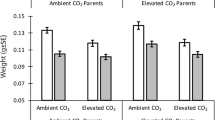
The impact of ocean acidification on marine ecosystems will depend on species capacity to adapt1,2. Recent studies show that the behaviour of reef fishes is impaired at projected CO2 levels3,4; however, individual variation exists that might promote adaptation. Here, we show a clear signature of parental sensitivity to high CO2 in the brain molecular phenotype of juvenile spiny damselfish, Acanthochromis polyacanthus, primarily driven by circadian rhythm genes. Offspring of CO2-tolerant and CO2-sensitive parents were reared at near-future CO2 (754 μatm) or present-day control levels (414 μatm). By integrating 33 brain transcriptomes and proteomes with a de novo assembled genome we investigate the molecular responses of the fish brain to increased CO2 and the expression of parental tolerance to high CO2 in the offspring molecular phenotype. Exposure to high CO2 resulted in differential regulation of 173 and 62 genes and 109 and 68 proteins in the tolerant and sensitive groups, respectively. Importantly, the majority of differences between offspring of tolerant and sensitive parents occurred in high CO2 conditions. This transgenerational molecular signature suggests that individual variation in CO2 sensitivity could facilitate adaptation of fish populations to ocean acidification.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Sunday, J. M. et al. Evolution in an acidifying ocean. Trends Ecol. Evol. 29, 117–125 (2014).
Pespeni, M. H. et al. Evolutionary change during experimental ocean acidification. Proc. Natl Acad. Sci. USA 110, 6937–6942 (2013).
Munday, P. L., Cheal, A. J., Dixson, D. L., Rummer, J. L. & Fabricius, K. E. Behavioural impairment in reef fishes caused by ocean acidification at CO2 seeps. Nature Clim. Change 4, 487–492 (2014).
Munday, P. L. et al. Replenishment of fish populations is threatened by ocean acidification. Proc. Natl Acad. Sci. USA 107, 12930–12934 (2010).
Doney, S. C., Fabry, V. J., Feely, R. A. & Kleypas, J. A. Ocean acidification: the other CO2 problem. Annu. Rev. Mar. Sci. 1, 169–192 (2009).
Nagelkerken, I. & Munday, P. L. Animal behaviour shapes the ecological effects of ocean acidification and warming: moving from individual to community-level responses. Glob. Change Biol. 22, 974–989 (2016).
Clements, J. & Hunt, H. Marine animal behaviour in a high CO2 ocean. Mar. Ecol. Prog. Ser. 536, 259–279 (2015).
Dixson, D. L., Jennings, A. R., Atema, J. & Munday, P. L. Odor tracking in sharks is reduced under future ocean acidification conditions. Glob. Change Biol. 21, 1454–1462 (2015).
Lai, F., Jutfelt, F. & Nilsson, G. E. Altered neurotransmitter function in CO2-exposed stickleback (Gasterosteus aculeatus): a temperate model species for ocean acidification research. Conserv. Physiol. 3, cov018 (2015).
Ou, M. et al. Responses of pink salmon to CO2-induced aquatic acidification. Nature Clim. Change 5, 950–955 (2015).
Holmes, T. H. & McCormick, M. I. Smell, learn and live: the role of chemical alarm cues in predator learning during early life history in a marine fish. Behav. Process. 83, 299–305 (2010).
Welch, M. J., Watson, S.-A., Welsh, J. Q., McCormick, M. I. & Munday, P. L. Effects of elevated CO2 on fish behaviour undiminished by transgenerational acclimation. Nature Clim. Change 4, 1086–1089 (2014).
Ferrari, M. C. O. et al. Intrageneric variation in antipredator responses of coral reef fishes affected by ocean acidification: implications for climate change projections on marine communities. Glob. Change Biol. 17, 2980–2986 (2011).
Chivers, D. P. et al. Impaired learning of predators and lower prey survival under elevated CO2: a consequence of neurotransmitter interference. Glob. Change Biol. 20, 515–522 (2014).
Munday, P. L. et al. Selective mortality associated with variation in CO2 tolerance in a marine fish. Ocean Acidif. 1, 1–5 (2012).
Heuer, R. M. & Grosell, M. Physiological impacts of elevated carbon dioxide and ocean acidification on fish. Am. J. Physiol. Regul. Integr. Comp. Physiol. 307, R1061–R1084 (2014).
Nilsson, G. E. et al. Near-future carbon dioxide levels alter fish behaviour by interfering with neurotransmitter function. Nature Clim. Change 2, 201–204 (2012).
Polakof, S., Panserat, S., Soengas, J. L. & Moon, T. W. Glucose metabolism in fish: a review. J. Comp. Physiol. B 182, 1015–1045 (2012).
Li, Y. et al. Upregulation of cytosolic phosphoenolpyruvate carboxykinase is a critical metabolic event in melanoma cells that repopulate tumors. Cancer Res. 75, 1191–1196 (2015).
Villeneuve, L. et al. Altered gene expression in the brain and ovaries of zebrafish (Danio rerio) exposed to the aromatase inhibitor fadrozole: microarray analysis and hypothesis generation. Environ. Toxicol. Chem. 28, 1767–1782 (2009).
Harshman, S. W., Young, N. L., Parthun, M. R. & Freitas, M. A. H1 histones: current perspectives and challenges. Nucleic Acids Res. 41, 9593–9609 (2013).
Kim, S., You, S. & Hwang, D. Aminoacyl-tRNA synthetases and tumorigenesis: more than housekeeping. Nature Rev. Cancer 11, 708–718 (2011).
Pan, T. Adaptive translation as a mechanism of stress response and adaptation. Annu. Rev. Genet. 47, 121–137 (2013).
Haschemeyer, A. E. V. Multiple aminoacyl-tRNA synthetases (translases) in temperature acclimation of eurythermal fish. J. Exp. Mar. Biol. Ecol. 87, 191–198 (1985).
Ishimatsu, A., Hayashi, M. & Kikkawa, T. Fishes in high-CO2, acidified oceans. Mar. Ecol. Prog. Ser. 373, 295–302 (2008).
Balment, R. J., Lu, W., Weybourne, E. & Warne, J. M. Arginine vasotocin a key hormone in fish physiology and behaviour: a review with insights from mammalian models. Gen. Comp. Endocrinol. 147, 9–16 (2006).
Dmitriev, A. V. & Mangel, S. C. A circadian clock regulates the pH of the fish retina. J. Physiol. 522, 77–82 (2000).
Peterson, M. S. & Gilmore, R. G. Hematocrit, osmolality, and ion concentration in fishes: consideration of circadian patterns in the experimental design. J. Exp. Mar. Biol. Ecol. 121, 73–78 (1988).
Dunlap, J. C. Molecular bases for circadian clocks. Cell 96, 271–290 (1999).
Zhang, E. E. & Kay, S. A. Clocks not winding down: unravelling circadian networks. Nature Rev. Mol. Cell Biol. 11, 764–776 (2010).
López-Patiño, M. A., Rodríguez-Illamola, A., Gesto, M., Soengas, J. L. & Míguez, J. M. Changes in plasma melatonin levels and pineal organ melatonin synthesis following acclimation of rainbow trout (Oncorhynchus mykiss) to different water salinities. J. Exp. Biol. 214, 928–936 (2011).
Acknowledgements
This study was supported by the Australian Research Council (ARC) and the ARC Centre of Excellence for Coral Reef Studies (P.L.M.), the Office of Competitive Research Funds OCRF-2014-CRG3-62140408 from the King Abdullah University of Science and Technology (T.R., M.L.B., P.L.M., T.Ryu, C.S.), and the University of Oslo (G.E.N.). This project was completed under James Cook University (JCU) ethics permit A1828. We thank the Marine and Aquaculture Research Facilities Unit (JCU), Integrative Systems Biology Laboratory (KAUST), and Biosciences Core Laboratory (KAUST) for support and assistance.
Author information
Authors and Affiliations
Contributions
M.J.W. and P.L.M. designed and managed the fish rearing experiments. M.J.W. performed the adult fish behavioural phenotyping. C.S. prepared the samples for RNA sequencing, and together with H.Z. protein samples for mass spectrometry. T.Ryu performed the genome assembly and gene annotation and wrote the corresponding part. C.S. analysed transcriptome expression data, and performed quantitative real-time PCR expression validation and variant analysis. C.S. analysed mass spectrometry data and integrated the data sets. G.E.N. assisted in interpreting the expression data. C.S., P.L.M., T.Ravasi and G.E.N. wrote the paper and all authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 1298 kb)
Supplementary Information
Supplementary Information (XLSX 189 kb)
Rights and permissions
About this article
Cite this article
Schunter, C., Welch, M., Ryu, T. et al. Molecular signatures of transgenerational response to ocean acidification in a species of reef fish. Nature Clim Change 6, 1014–1018 (2016). https://doi.org/10.1038/nclimate3087
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nclimate3087
This article is cited by
-
Neuromolecular responses in disrupted mutualistic cleaning interactions under future environmental conditions
BMC Biology (2023)
-
Epigenetic plasticity enables copepods to cope with ocean acidification
Nature Climate Change (2022)
-
Phenotypic responses in fish behaviour narrow as climate ramps up
Climatic Change (2022)
-
Circadian Rhythm Disturbances Due to Exposure to Acidified Conditions and Different Photoperiods in Juvenile Olive Flounder (Paralichthys olivaceus)
Ocean Science Journal (2021)
-
Ocean acidification promotes broad transcriptomic responses in marine metazoans: a literature survey
Frontiers in Zoology (2020)