Abstract
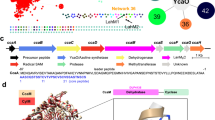
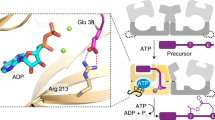
Regioselective modification of amino acids within the context of a peptide is common to a number of biosynthetic pathways, and many of the resulting products have potential as therapeutics. The ATP-dependent enzyme LynD heterocyclizes multiple cysteine residues to thiazolines within a peptide substrate. The enzyme requires the substrate to have a conserved N-terminal leader for full activity. Catalysis is almost insensitive to immediately flanking residues in the substrate, suggesting that recognition occurs distant from the active site. Nucleotide and peptide substrate co-complex structures of LynD reveal that the substrate leader peptide binds to and extends the β-sheet of a conserved domain of LynD, whereas catalysis is accomplished in another conserved domain. The spatial segregation of catalysis from recognition combines seemingly contradictory properties of regioselectivity and promiscuity, and it appears to be a conserved strategy in other peptide-modifying enzymes. A variant of LynD that efficiently processes substrates without a leader peptide has been engineered.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Blunt, J.W., Copp, B.R., Keyzers, R.A., Munro, M.H. & Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 31, 160–258 (2014).
Driggers, E.M., Hale, S.P., Lee, J. & Terrett, N.K. The exploration of macrocycles for drug discovery—an underexploited structural class. Nat. Rev. Drug Discov. 7, 608–624 (2008).
Mayer, A.M., Rodriguez, A.D., Taglialatela-Scafati, O. & Fusetani, N. Marine pharmacology in 2009–2011: marine compounds with antibacterial, antidiabetic, antifungal, anti-inflammatory, antiprotozoal, antituberculosis, and antiviral activities; affecting the immune and nervous systems, and other miscellaneous mechanisms of action. Mar. Drugs 11, 2510–2573 (2013).
Sivonen, K., Leikoski, N., Fewer, D.P. & Jokela, J. Cyanobactins—ribosomal cyclic peptides produced by cyanobacteria. Appl. Microbiol. Biotechnol. 86, 1213–1225 (2010).
Schmidt, E.W. et al. Patellamide A and C biosynthesis by a microcin-like pathway in Prochloron didemni, the cyanobacterial symbiont of Lissoclinum patella. Proc. Natl. Acad. Sci. USA 102, 7315–7320 (2005).
Donia, M.S. et al. Natural combinatorial peptide libraries in cyanobacterial symbionts of marine ascidians. Nat. Chem. Biol. 2, 729–735 (2006).
Long, P.F., Dunlap, W.C., Battershill, C.N. & Jaspars, M. Shotgun cloning and heterologous expression of the patellamide gene cluster as a strategy to achieving sustained metabolite production. ChemBioChem 6, 1760–1765 (2005).
Arnison, P.G. et al. Ribosomally synthesized and post-translationally modified peptide natural products: overview and recommendations for a universal nomenclature. Nat. Prod. Rep. 30, 108–160 (2013).
Milne, B.F., Long, P.F., Starcevic, A., Hranueli, D. & Jaspars, M. Spontaneity in the patellamide biosynthetic pathway. Org. Biomol. Chem. 4, 631–638 (2006).
Schmidt, E.W. & Donia, M.S. Cyanobactin ribosomally synthesized peptides—a case of deep metagenome mining. Methods Enzymol. 458, 575–596 (2009).
Donia, M.S. & Schmidt, E.W. Linking chemistry and genetics in the growing cyanobactin natural products family. Chem. Biol. 18, 508–519 (2011).
Houssen, W.E. et al. An efficient method for the in vitro production of azol(in)ebased cyclic peptides. Angew. Chem. Int. Edn. Engl. 53, 14171–14174 (2014).
Houssen, W.E. et al. The discovery of new cyanobactins from Cyanothece PCC 7425 defines a new signature for processing of patellamides. ChemBioChem 13, 2683–2689 (2012).
Koehnke, J. et al. The mechanism of patellamide macrocyclization revealed by the characterization of the PatG macrocyclase domain. Nat. Struct. Mol. Biol. 19, 767–772 (2012).
Koehnke, J. et al. An enzymatic route to selenazolines. ChemBioChem 14, 564–567 (2013).
Koehnke, J. et al. The cyanobactin heterocyclase enzyme: a processive adenylase that operates with a defined order of reaction. Angew. Chem. Int. Edn Engl. 52, 13991–13996 (2013).
Agarwal, V., Pierce, E., McIntosh, J., Schmidt, E.W. & Nair, S.K. Structures of cyanobactin maturation enzymes define a family of transamidating proteases. Chem. Biol. 19, 1411–1422 (2012).
Bent, A.F. et al. Structure of PatF from Prochloron didemni. Acta Crystallogr. Sect. F Struct. Biol. Cryst. Commun. 69, 618–623 (2013).
Majmudar, J.D. & Gibbs, R.A. Pericyclic prenylation: peptide modification through a Claisen rearrangement. ChemBioChem 12, 2723–2726 (2011).
Melby, J.O., Li, X. & Mitchell, D.A. Orchestration of enzymatic processing by thiazole/oxazole-modified microcin dehydrogenases. Biochemistry 53, 413–422 (2014).
McIntosh, J.A. & Schmidt, E.W. Marine molecular machines: heterocyclization in cyanobactin biosynthesis. ChemBioChem 11, 1413–1421 (2010).
McIntosh, J.A., Donia, M.S. & Schmidt, E.W. Insights into heterocyclization from two highly similar enzymes. J. Am. Chem. Soc. 132, 4089–4091 (2010).
Nielsen, D.S. et al. Improving on nature: making a cyclic heptapeptide orally bioavailable. Angew. Chem. Int. Edn Engl. 53, 12059–12063 (2014).
Regni, C.A. et al. How the MccB bacterial ancestor of ubiquitin E1 initiates biosynthesis of the microcin C7 antibiotic. EMBO J. 28, 1953–1964 (2009).
Dunbar, K.L. & Mitchell, D.A. Insights into the mechanism of peptide cyclodehydrations achieved through the chemoenzymatic generation of amide derivatives. J. Am. Chem. Soc. 135, 8692–8701 (2013).
Dunbar, K.L., Melby, J.O. & Mitchell, D.A. YcaO domains use ATP to activate amide backbones during peptide cyclodehydrations. Nat. Chem. Biol. 8, 569–575 (2012).
Melby, J.O., Dunbar, K.L., Trinh, N.Q. & Mitchell, D.A. Selectivity, directionality, and promiscuity in peptide processing from a Bacillus sp. Al Hakam cyclodehydratase. J. Am. Chem. Soc. 134, 5309–5316 (2012).
Leikoski, N. et al. Highly diverse cyanobactins in strains of the genus Anabaena. Appl. Environ. Microbiol. 76, 701–709 (2010).
Goto, Y., Ito, Y., Kato, Y., Tsunoda, S. & Suga, H. One-pot synthesis of azoline-containing peptides in a cell-free translation system integrated with a posttranslational cyclodehydratase. Chem. Biol. 21, 766–774 (2014).
Dunbar, K.L. et al. Discovery of a new ATP-binding motif involved in peptidic azoline biosynthesis. Nat. Chem. Biol. 10, 823–829 (2014).
Ortega, M.A. et al. Structure and mechanism of the tRNA-dependent lantibiotic dehydratase NisB. Nature 517, 509–512 (2015).
Willey, J.M. & van der Donk, W.A. Lantibiotics: peptides of diverse structure and function. Annu. Rev. Microbiol. 61, 477–501 (2007).
Oman, T.J. & van der Donk, W.A. Follow the leader: the use of leader peptides to guide natural product biosynthesis. Nat. Chem. Biol. 6, 9–18 (2010).
Ruffner, D.E., Schmidt, E.W. & Heemstra, J.R. Assessing the combinatorial potential of the RiPP cyanobactin tru pathway. ACS Synth. Biol. 4, 482–492 (2014).
Oman, T.J., Knerr, P.J., Bindman, N.A., Velasquez, J.E. & van der Donk, W.A. An engineered lantibiotic synthetase that does not require a leader peptide on its substrate. J. Am. Chem. Soc. 134, 6952–6955 (2012).
Studier, F.W. Protein production by auto-induction in high-density shaking cultures. Protein Expr. Purif. 41, 207–234 (2005).
Winter, G. xia2: an expert system for macromolecular crystallography data reduction. J. Appl. Crystallogr. 43, 186–190 (2009).
McCoy, A.J., Grosse-Kunstleve, R.W., Storoni, L.C. & Read, R.J. Likelihood-enhanced fast translation functions. Acta Crystallogr. D Biol. Crystallogr. 61, 458–464 (2005).
Storoni, L.C., McCoy, A.J. & Read, R.J. Likelihood-enhanced fast rotation functions. Acta Crystallogr. D Biol. Crystallogr. 60, 432–438 (2004).
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D Biol. Crystallogr. 60, 2126–2132 (2004).
Murshudov, G.N. et al. REFMAC5 for the refinement of macromolecular crystal structures. Acta Crystallogr. D Biol. Crystallogr. 67, 355–367 (2011).
Adams, P.D. et al. Recent developments in the PHENIX software for automated crystallographic structure determination. J. Synchrotron Radiat. 11, 53–55 (2004).
DeLano, W.L. The PyMOL Molecular Graphics System (DeLano Scientific, San Carlos, California, USA, 2002).
Acknowledgements
This work was supported by grants from the European Research Council (no. 339367, J.H.N. and M.J.), the UK Biotechnology and Biological Sciences Research Council (no. BB/K015508/1, J.H.N. and M.J.) and the Wellcome Trust, which funded the purchase of the TripleTOF 5600 mass spectrometer at the Biomedical Sciences Research Complex Mass Spectrometry and Proteomics Facility, University of St. Andrews (grant no. 094476/Z/10/Z), and the MALDI TOF-TOF Analyser (grant no. WT079272AIA). We acknowledge use of the Diamond (beamlines I02 and I24) and ESRF (beamline ID29) synchrotrons. J.H.N. is a Royal Society Wolfson Merit Award Holder and a 1000 talent scholar of the Chinese Government at Sichuan University.
Author information
Authors and Affiliations
Contributions
J.K. carried out crystallization and X-ray crystallography experiments, interpreted data and wrote the paper. G.M. carried out protein expression and purification, heterocyclization reactions, ITC measurements and selective 18O labeling experiments; interpreted data; and wrote the paper. A.F.B. carried out site-directed mutagenesis, protein expression and purification, heterocyclization reactions and MALDI-TOF/MS data acquisition experiments; interpreted data; and wrote the paper. H.L. carried out protein expression and purification, crystallization and X-ray crystallography experiments, and interpreted data. S.S. carried out MALDI-TOF/MS, LC/MS and MS/MS data acquisition experiments and interpreted data. C.B. carried out MALDI-TOF/MS, LC/MS and MS/MS data acquisition experiments and interpreted data. T.L. carried out 31P NMR experiments and interpreted data. W.E.H. interpreted data and wrote the paper. M.J. interpreted data and wrote the paper. J.H.N. carried out X-ray crystallography experiments, interpreted data and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The Universities of St. Andrews and Aberdeen have filed a patent application with the UK Patent Office on the use of modified LynD enzyme. J.K., A.F.B., G.M., W.E.H., M.J. and J.H.N. are among those listed as inventors.
Supplementary information
Supplementary Text and Figures
Supplementary Results, Supplementary Figures 1–9 and Supplementary Tables 1–3 (PDF 4919 kb)
Rights and permissions
About this article
Cite this article
Koehnke, J., Mann, G., Bent, A. et al. Structural analysis of leader peptide binding enables leader-free cyanobactin processing. Nat Chem Biol 11, 558–563 (2015). https://doi.org/10.1038/nchembio.1841
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.1841
This article is cited by
-
Crystal structure and catalytic mechanism of the MbnBC holoenzyme required for methanobactin biosynthesis
Cell Research (2022)
-
Structures and function of a tailoring oxidase in complex with a nonribosomal peptide synthetase module
Nature Communications (2022)
-
Biosynthesis and characterization of fuscimiditide, an aspartimidylated graspetide
Nature Chemistry (2022)
-
The core of the matter
Nature Chemical Biology (2021)
-
Functional elucidation of TfuA in peptide backbone thioamidation
Nature Chemical Biology (2021)