Abstract
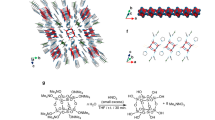
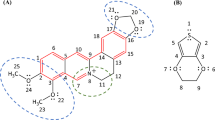
The consequence of unpaired electrons in organic molecules has fascinated and confounded chemists for over a century. The study of open-shell molecules has been rekindled in recent years as new synthetic methods, improved spectroscopic techniques and powerful computational tools have been brought to bear on this field. Nonetheless, it is the intrinsic instability of the biradical species that limits the practicality of this research. Here we report the synthesis and characterization of a molecule based on the diindeno[b,i]anthracene framework that exhibits pronounced open-shell character yet possesses remarkable stability. The synthetic route is rapid, efficient and possible on the gram scale. The molecular structure was confirmed through single-crystal X-ray diffraction. From variable-temperature Raman spectroscopy and magnetic susceptibility measurements a thermally accessible triplet excited state was found. Organic field-effect transistor device data show an ambipolar performance with balanced electron and hole mobilities. Our results demonstrate the rational design and synthesis of an air- and temperature-stable biradical compound.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Tschitschibabin, A. E. Über einige phenylierte Derivate des p,p-Ditolyls. Chem. Ber. 40, 1810–1819 (1907).
Montgomery, L. K., Huffman, J. C., Jurczak, E. A. & Grendze, M. P. The molecular structures of Thiele's and Chichibabin's hydrocarbons. J. Am. Chem. Soc. 108, 6004–6011 (1986).
Doehnert, D. & Koutecky, J. Occupation numbers of natural orbitals as a criterion for biradical character. Different kinds of biradicals. J. Am. Chem. Soc. 102, 1789–1796 (1980).
Yamaguchi, K. et al. Analytical and ab initio studies of effective exchange interactions, polyradical character, unpaired electron density, and information entropy in radical clusters (R)N: allyl radical cluster (N = 2–10) and hydrogen radical cluster (N = 50). Int. J. Quantum Chem. 90, 370–385 (2002).
Abe, M. Diradicals. Chem. Rev. 113, 7011–7088 (2013).
Karafiloglou, P. The double (or dynamic) spin polarization in π diradicals. J. Chem. Educ. 66, 816–817 (1989).
Shimizu, A. et al. Indeno[2,1-b]fluorene: a 20-π-electron hydrocarbon with very low-energy light absorption. Angew. Chem. Int. Ed. 52, 6076–6079 (2013).
Kubo, T. et al. Synthesis, intermolecular interaction, and semiconductive behavior of a delocalized singlet biradical hydrocarbon. Angew. Chem. Int. Ed. 44, 6564–6568 (2005).
Li, Y. et al. Kinetically blocked stable heptazethrene and octazethrene: closed-shell or open-shell in the ground state? J. Am. Chem. Soc. 134, 14913–14922 (2012).
Konishi, A. et al. Synthesis and characterization of teranthene: a singlet biradical polycyclic aromatic hydrocarbon having Kekulé structures. J. Am. Chem. Soc. 132, 11021–11023 (2010).
Sun, Z., Ye, Q., Chi, C. & Wu, J. Low band gap polycyclic hydrocarbons: from closed-shell near infrared dyes and semiconductors to open-shell radicals. Chem. Soc. Rev. 41, 7857–7889 (2012).
Nakano, M. et al. Second hyperpolarizability (γ) of singlet diradical system: dependence of γ on the diradical character. J. Phys. Chem. A 109, 885–891 (2005).
Motomura, S. et al. Size dependences of the diradical character and the second hyperpolarizabilities in dicyclopenta-fused acenes: relationships with their aromaticity/antiaromaticity. Phys. Chem. Chem. Phys. 13, 20575–20583 (2011).
Nakano, M. et al. Relationship between third-order nonlinear optical properties and magnetic interactions in open-shell systems: a new paradigm for nonlinear optics. Phys. Rev. Lett. 99, 033001 (2007).
Nakano, M. & Champagne, B. Theoretical design of open-shell singlet molecular systems for nonlinear optics. J. Phys. Chem. Lett. 6, 3236–3256 (2015).
Kamada, K. et al. Strong two-photon absorption of singlet diradical hydrocarbons. Angew. Chem. Int. Ed. 46, 3544–3546 (2007).
Zeng, Z. et al. Pro-aromatic and anti-aromatic π-conjugated molecules: an irresistible wish to be diradicals. Chem. Soc. Rev. 44, 6578–6596 (2015).
Morita, Y., Suzuki, S., Sato, K. & Takui, T. Synthetic organic spin chemistry for structurally well-defined open-shell graphene fragments. Nature Chem. 3, 197–204 (2011).
Ito, S., Minami, T. & Nakano, M. Diradical character based design for singlet fission of condensed-ring systems with 4nπ electrons. J. Phys. Chem. C 116, 19729–19736 (2012).
Smith, M. B. & Michl, J. Recent advances in singlet fission. Annu. Rev. Phys. Chem. 64, 361–386 (2013).
Varnavski, O. et al. High yield ultrafast intramolecular singlet exciton fission in a quinoidal bithiophene. J. Phys. Chem. Lett. 6, 1375–1384 (2015).
Thorley, K. J. & Anthony, J. E. The electronic nature and reactivity of the larger acenes. Isr. J. Chem. 13, 642–649 (2014).
Anthony, J. E., Brooks, J. S., Eaton, D. L. & Parkin, S. R. Functionalized pentacene: improved electronic properties from control of solid-state order. J. Am. Chem. Soc. 123, 9482–9483 (2001).
Fudickar, W. & Linker, T. Why triple bonds protect acenes from oxidation and decomposition. J. Am. Chem. Soc. 134, 15071–15082 (2012).
Park, J.-H. et al. Soluble and easily crystallized anthracene derivatives: precursors of solution-processable semiconducting molecules. Org. Lett. 9, 2573–2576 (2007).
Chase, D. T. et al. 6,12-Diarylindeno[1,2-b]fluorenes: syntheses, photophysics, and ambipolar OFETs. J. Am. Chem. Soc. 134, 10349–10352 (2012).
Wood, J. D., Jellison, J. L., Finke, A. D., Wang, L. & Plunkett, K. N. Electron acceptors based on functionalizable cyclopenta[hi]aceanthrylenes and dicyclopenta[de,mn]tetracenes. J. Am. Chem. Soc. 134, 15783–15789 (2012).
Chikamatsu, M. et al. Ambipolar organic field-effect transistors based on a low band gap semiconductor with balance hole and electron mobilities. Appl. Phys. Lett. 91, 043506 (2007).
Koike, H. et al. Stable delocalized singlet biradical hydrocarbon for organic field-effect transistors. Adv. Funct. Mater. 26, 277–283 (2016).
Marshall, J. L. & Haley, M. M. in Organic Redox Systems: Synthesis, Properties and Applications 1st edn (ed. Nishinaga, T.) 311–358 (Wiley, 2016).
Turro, N. J., Ramamurthy, V. & Scaiano, J. C. in Principles of Molecular Photochemistry: An Introduction 1st edn (University Science Books, 2009).
Bleaney, B. & Bowers, K. D. Anomalous paramagnetism of copper acetate. Proc. R. Soc. Lond. A 214, 451–465 (1952).
Bernard, Y. A., Shao, Y. & Krylov, A. I. General formulation of spin-flip time-dependent density functional theory using non-collinear kernels: theory, implementation, and benchmarks. J. Chem. Phys. 136, 204103 (2012).
Casado, J. et al. Raman detection of ‘ambiguous’ conjugated biradicals: rapid thermal singlet-to-triplet intersystem crossing in an extended viologen. Angew. Chem. Int. Ed. 47, 1443–1446 (2008).
Casado, J., Ponce Ortiz, R. & López Navarrete, J. T. Quinoidal oligothiophenes: new properties behind an unconventional electronic structure. Chem. Soc. Rev. 41, 5672–5686 (2012).
Casado, J. et al. Vibrational fingerprint of the structural tuning in push–pull organic chromophores with quinoid or proaromatic spacers. J. Chem. Phys. 126, 074701 (2007).
Albrecht, A. C. On the theory of Raman intensities. J. Chem. Phys. 34, 1476–1484 (1961).
Burdett, J. J. & Bardeen, C. J. Quantum beats in crystalline tetracene delayed fluorescence due to triplet pair coherences produced by direct singlet fission. J. Am. Chem. Soc. 134, 8597–8607 (2012).
Frisch, M. J. et al. Gaussian 09, Revision D.01 (Gaussian, Inc., 2009).
Stephens, P. J., Devlin, F. J., Chabalowski, C. F. & Frisch, M. J. Ab initio calculation of vibrational absorption and circular dichroism spectra using density functional force fields. J. Phys. Chem. 98, 11623–11627 (1994).
Krishnan, R., Binkley, J. S., Seeger, R. & Pople, J. A. Self-consistent molecular orbital methods. XX. A basis set for correlated wave functions. J. Chem. Phys. 72, 650–654 (1980).
Yamaguchi, K. in Self-Consistent Field: Theory and Applications 1st edn (eds Carbó, R. & Klobukowski, M.) 727–828 (Elsevier, 1990).
Herges, R. & Geuenich, D. Delocalization of electrons in molecules. J. Phys. Chem. A 105, 3214–3220 (2001).
Keith, T. A. & Bader, R. F. W. Calculation of magnetic response properties using a continuous set of gauge transformations. Chem. Phys. Lett. 210, 223–231 (1993).
Becke, A. D. Density-functional exchange-energy approximation with correct asymptotic behavior. Phys. Rev. A 38, 3098–3100 (1988).
Lee, C., Yang, W. & Parr, R. G. Development of the Colle–Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 37, 785–789 (1988).
Iikura, H., Tsuneda, T., Yanai, T. & Hirao, K. A long-range correction scheme for generalized-gradient-approximation exchange functionals. J. Chem. Phys. 115, 3540–3544 (2001).
Gershoni-Poranna, R. & Stanger, A. The NICS-XY-scan: identification of local and global ring currents in multi-ring systems. Chem. Eur. J. 20, 5673–5688 (2014).
Wolinski, K., Hinton, J. F. & Pulay, P. Efficient implementation of the gauge-independent atomic orbital method for NMR chemical shift calculations. J. Am. Chem. Soc. 112, 8251–8260 (1990).
Stanger, A. Obtaining relative induced ring currents quantitatively from NICS. J. Org. Chem. 75, 2281–2288 (2010).
Acknowledgements
This work was supported by the US National Science Foundation (CHE-1301485), by the Spanish Government, MINECO (CTQ2012-33733 and CTQ2011-26507), by Junta de Andalucía (P09-FQM-4708) and Generalitat Valenciana (PrometeoII/2014/076), by a Japan Society for the Promotion of Science (JSPS) Research Fellowship for Young Scientists (No. 15J04949), Grant-in-Aid for Scientific Research (A) (No. 25248007) from the JSPS, a Grant-in-Aid for Scientific Research on Innovative Areas ‘Stimuli-Responsive Chemical Species’ (No. A24109002a), ‘π-System Figuration’ (15H00999), ‘Photosynergetics’ (A26107004a), the Strategic Programs for Innovative Research, Ministry of Education, Culture, Sports, Science & Technology, Japan, the Computational Materials Science Initiative, Japan, the Swedish Research Council (project grant 621-2011-4177) and the Swedish National Infrastructure for Computation (NSC, Linköping). K.J. and H.O. thank R. Herges for providing the AICD 2.0.0 program. The authors acknowledge the Biomolecular Mass Spectrometry Core of the Environmental Health Sciences Core Center at Oregon State University (NIH P30ES000210). We thank T. Kubo (Osaka) for insightful discussions.
Author information
Authors and Affiliations
Contributions
G.E.R. conceived the project, and designed and carried out the experiments, analysed the data and wrote the manuscript. M.M.H. played a critical role in the discussion of the experimental design, project direction, experiments and results, and in the preparation of the manuscript. J.L.M. acquired and analysed the CV data. I.A.M., G.L.E. and R.P.O. obtained the OFET data. L.N.Z. acquired and analysed the X-ray crystallographic data. J.L.Z. performed the Raman spectroscopic measurements. C.G.G. performed the SQUID experiments. J.C. interpreted the magnetic and spectroscopic data and co-wrote the paper. K.F. and M.N. performed the calculation and discussed the geometry optimization, the open-shell character and the S–T gaps. K.J. performed the ACID and NICS-XY density functional theory calculations, and, together with H.O., analysed the data from these computations. All the authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 5799 kb)
Supplementary information
Crystallographic data for compound DIAn. (CIF 1014 kb)
Rights and permissions
About this article
Cite this article
Rudebusch, G., Zafra, J., Jorner, K. et al. Diindeno-fusion of an anthracene as a design strategy for stable organic biradicals. Nature Chem 8, 753–759 (2016). https://doi.org/10.1038/nchem.2518
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.2518
This article is cited by
-
A highly efficient open-shell singlet luminescent diradical with strong magnetoluminescence properties
Light: Science & Applications (2023)
-
Understanding the Stability of π-Conjugated Diradicaloid Organic Molecules
Journal of Electronic Materials (2023)
-
Progress of Indeno-type Organic Diradicaloids
Chemical Research in Chinese Universities (2023)
-
Achieving large thermal hysteresis in an anthracene-based manganese(II) complex via photo-induced electron transfer
Nature Communications (2022)
-
Design of an open-shell nitrogen-centered diradicaloid with tunable stimuli-responsive electronic properties
Communications Chemistry (2022)