Abstract
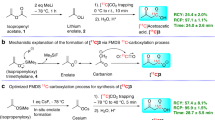
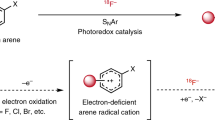
Molecules labelled with the unnatural isotope fluorine-18 are used for positron emission tomography. Currently, this molecular imaging technology is not exploited at its full potential because many 18F-labelled probes are inaccessible or notoriously difficult to produce. Typical challenges associated with 18F radiochemistry are the short half-life of 18F (<2 h), the use of sub-stoichiometric amounts of 18F, relative to the precursor and other reagents, as well as the limited availability of parent 18F sources of suitable reactivity ([18F]F– and [18F]F2). There is a high-priority demand for general methods allowing access to [18F]CF3-substituted molecules for application in pharmaceutical discovery programmes. We report the development of a process for the late-stage [18F]trifluoromethylation of (hetero)arenes from [18F]fluoride using commercially available reagents and (hetero)aryl iodides. This [18F]CuCF3–based protocol benefits from a large substrate scope and is characterized by its operational simplicity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Phelps, M. E. Positron emission tomography provides molecular imaging of biological processes. Proc. Natl Acad. Sci. USA 97, 9226–9233 (2000).
Ametamey, S. M., Honer, M. & Schubiger, P. A. Molecular imaging with PET. Chem. Rev. 108, 1501–1516 (2008).
Matthews, P. M., Rabiner, E. A., Passchier, J. & Gunn, R. N. Positron emission tomography molecular imaging for drug development. Br. J. Clin. Pharmacol. 73, 175–186 (2012).
Wong, D. F., Tauscher, J. & Gründer, G. The role of imaging in proof of concept for CNS drug discovery and development. Neuropsychopharmacol. Rev. 34, 187–203 (2009).
Miller, P. W., Long, N. J., Vilar, R. & Gee, A. D. Synthesis of 11C, 18F, 15O, 13N radiolabels for positron emission tomography. Angew. Chem. Int. Ed. 47, 8998–9033 (2008).
Tredwell, M. & Gouverneur, V. 18F labeling of arenes. Angew. Chem. Int. Ed. 51, 11426–11437 (2012).
Müller, K., Faeh, C. & Diederich, F. Fluorine in pharmaceuticals: looking beyond intuition. Science 317, 1881–1886 (2007).
Purser, S., Moore, P. R., Swallow, S. & Gouverneur, V. Fluorine in medicinal chemistry. Chem. Soc. Rev. 37, 320–330 (2008).
Lee, E. et al. A fluoride-derived electrophilic late-stage fluorination reagent for PET imaging. Science 334, 639–642 (2011).
Lee, E., Hooker, J. M. & Ritter, T. Nickel-mediated oxidative fluorination for PET with aqueous [18F]fluoride. J. Am. Chem. Soc. 134, 17456–17458 (2012).
Gao Z. et al. Metal-free oxidative fluorination of phenols with [18F]fluoride. Angew. Chem. Int. Ed. 51, 6733–6737 (2012).
Kilbourn, M. R., Pavia, M. R. & Gregor, V. E. Synthesis of fluorine-18 labeled GABA uptake inhibitors Appl. Radiat. Isot. 41, 823–828 (1990).
Prabhakaran, J. et al. Synthesis and in vivo evaluation of [18F]-4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl]benzenesulfonamide as a PET imaging probe for COX-2 expression. Bioorg. Med. Chem. 15, 1802–1807 (2007).
Angelini, G., Speranza, M., Shiue, C-Y. & Wolf, A. P. H18F+Sb2O3: a new selective radiofluorinating agent. J. Chem. Soc. Chem. Commun. 924–925 (1986).
Angelini, G., Speranza, M., Wolf, A. P. & Shiue, C-Y. Synthesis of N-(α,α,α-tri[18F]fluoro-m-tolyl)piperazine. A potent serotonin agonist. J. Label. Compd. Radiopharm. 28, 1441–1448 (1990).
Furuya, T., Kamlet, A. S. & Ritter, T. Catalysis for fluorination and trifluoromethylation. Nature 473, 470–477 (2011).
Tomashenko, O. A. & Grushin, V. V. Aromatic trifluoromethylation with metal complexes. Chem. Rev. 111, 4475–4521 (2011).
Wu, X-F., Neumann, H. & Beller, M. Recent developments on the trifluoromethylation of (hetero)arenes. Chem. Asian J. 7, 1744–1754 (2012).
Mizuta, S. et al. Catalytic decarboxylative fluorination for the synthesis of tri- and difluoromethyl arenes. Org. Lett. 15, 2648–2651 (2013).
Teare, H. et al. Radiosynthesis and evaluation of [18F]selectfluor bis(triflate). Angew. Chem. Int. Ed. 49, 6821–6824 (2010).
Bergman, J. & Solin, O. Fluorine-18-labeled fluorine gas for synthesis of tracer molecules. Nucl. Med. Biol. 24, 677–683 (1997).
MacNeil, J. G. Jr & Burton, D. J. Generation of trifluoromethylcopper from chlorodifluoroacetate. J. Fluor. Chem. 55, 225–227 (1991).
Su, D-B., Duan, J-X., Yu, A-J. & Chen, Q-Y. Synthesis of functionalized long-chain perfluoroalkanes from methyl halofluoroacetates: a process of difluorocarbene insertion into copper–carbon bonds. J. Fluor. Chem. 65, 11–14 (1993).
Duan, J-X., Su, D-B. & Chen, Q-Y. Trifluoromethylation of organic halides with methyl halodifluoroacetates—a process via difluorocarbene and trifluoromethide intermediates. J. Fluor. Chem. 61, 279–284 (1993).
Duan, J-X., Su, D-B., Wu, J-P. & Chen, Q-Y. Synthesis of trifluoromethyl aryl derivatives via difluorocarbene precursors and nitro-substituted aryl chlorides. J. Fluor. Chem. 66, 167–169 (1994).
Oishi, M., Kondo, H. & Amii, H. Aromatic trifluoromethylation catalytic in copper. Chem. Commun. 1909–1911 (2009).
Morimoto, H., Tsubogo, T., Litvinas, N. D. & Hartwig, J. F. A broadly applicable copper reagent for trifluoromethylations and perfluoroalkylations of aryl iodides and bromides. Angew. Chem. Int. Ed. 50, 3793–3798 (2011).
Chu, L. & Qing, F-L. Copper-catalyzed direct C–H oxidative trifluoromethylation of heteroarenes. J. Am. Chem. Soc. 134, 1298–1304 (2012).
Naimi, E., Duan, W., Wiebe, L. I. & Knaus, E. E. Synthesis of unnatural 7-substituted-1-(2-deoxy-β-D-ribofuranosyl)isocarbostyrils: ‘thymine replacement’ analogs of deoxythymidine for evaluation as antiviral and anticancer agents. Nucleosides Nucleotides Nucleic Acids 20, 1533–1553 (2001).
Nagib, D. A. & MacMillan, D. W. C. Trifluoromethylation of arenes and heteroarenes by means of photoredox catalysis. Nature 480, 224–228 (2011).
Ji, Y. et al. Innate C–H trifluoromethylation of heterocycles. Proc. Natl Acad. Sci. USA 108, 14411–14415 (2011).
Hammadi, A. & Crouzel, C. Synthesis of [18F]-(S)-fluoxetine: a selective serotonine uptake inhibitor. J. Label. Compd. Radiopharm. 33, 703–710 (1993).
Jacobson, O. et al. Prostate cancer PET bioprobes: synthesis of [18F]-radiolabeled hydroxyflutamide derivatives. Bioorg. Med. Chem. 13, 6195–6205 (2005).
Lapi, S. E. & Welch, M. J. A historical perspective on the specific activity of radiopharmaceuticals: what have we learned in the 35 years of the ISRC? Nucl. Med. Biol. 40, 314–320 (2013).
Anderson, C. J. & Ferdani, R. Copper-64 radiopharmaceuticals for PET imaging of cancer: advances in preclinical and clinical research. Cancer Biother. Radiopharm. 24, 379–393 (2009).
Zeng, D., Zeglis, B. M., Lewis, J. S. & Anderson, C. J. The growing impact of bioorthogonal click chemistry on the development of radiopharmaceuticals. J. Nucl Med. 54, 829–832 (2013).
Acknowledgements
Financial support was provided by Imanova, GlaxoSmithKline, the European Union (grant PIIF-GA-20100274903 to S.M.), the Engineering and Physical Sciences Research Council (M.T.) and the Cancer Research UK (M.T.). The authors thank P. Holdship (Department of Earth Sciences, University of Oxford) for the ICP-MS measurements. V.G. is a recipient of a Royal Society Wolfson Research Merit Award.
Author information
Authors and Affiliations
Contributions
M.H., M.T. and S.M. performed and analysed experiments. All authors contributed to the design of experiments to develop this reaction and probe its utility. V.G. and J.P. prepared the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 2866 kb)
Rights and permissions
About this article
Cite this article
Huiban, M., Tredwell, M., Mizuta, S. et al. A broadly applicable [18F]trifluoromethylation of aryl and heteroaryl iodides for PET imaging. Nature Chem 5, 941–944 (2013). https://doi.org/10.1038/nchem.1756
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.1756
This article is cited by
-
Radiochemistry for positron emission tomography
Nature Communications (2023)
-
Highlight selection of radiochemistry and radiopharmacy developments by editorial board
EJNMMI Radiopharmacy and Chemistry (2022)
-
[18F]Difluorocarbene for positron emission tomography
Nature (2022)
-
Closing the gap between 19F and 18F chemistry
EJNMMI Radiopharmacy and Chemistry (2021)
-
Contemporary synthetic strategies in organofluorine chemistry
Nature Reviews Methods Primers (2021)