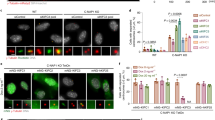
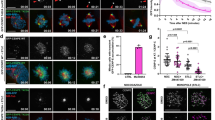
Abstract
Kinetochores attach the replicated chromosomes to the mitotic spindle and orchestrate their transmission to the daughter cells. Kinetochore–spindle binding and chromosome segregation are mediated by the multi-copy KNL1Spc105, MIS12Mtw1 and NDC80Ndc80 complexes that form the so-called KMN network. KMN–spindle attachment is regulated by the Aurora BIpl1 and MPS1Mps1 kinases. It is unclear whether other mechanisms exist that support KMN activity during the cell cycle. Using budding yeast, we show that kinetochore protein Cnn1 localizes to the base of the Ndc80 complex and promotes a functionally competent configuration of the KMN network. Cnn1 regulates KMN activity in a spatiotemporal manner by inhibiting the interaction between its complexes. Cnn1 activity peaks in anaphase and is driven by the Cdc28, Mps1 and Ipl1 kinases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Accession codes
Change history
11 February 2013
In the version of this Article that was originally published, the acknowledgement to M. Winey and reference 48 in the Methods section were omitted in error. These corrections have been made in the PDF and HTML versions of the Article.
References
Lampert, F. & Westermann, S. A blueprint for kinetochores—new insights into the molecular mechanics of cell division. Nat. Rev. Mol. Cell Biol. 12, 407–412 (2011).
Tanaka, K. et al. Molecular mechanisms of kinetochore capture by spindle microtubules. Nature 434, 987–994 (2005).
Tanaka, T. U. Kinetochore-microtubule interactions: steps toward bi-orientation. EMBO J. 29, 4070–4082 (2010).
Cheeseman, I. M., Chappie, J. S., Wilson-Kubalek, E. M. & Desai, A. The conserved KMN network constitutes the core microtubule-binding site of the kinetochore. Cell 127, 983–997 (2006).
Espeut, J. et al. Microtubule binding by KNL-1 contributes to spindle checkpoint silencing at the kinetochore. J. Cell Biol. 196, 469–482 (2012).
Tanaka, T. U. et al. Evidence that the Ipl1-Sli15 (Aurora kinase-INCENP) complex promotes chromosome bi-orientation by altering kinetochore-spindle pole connections. Cell 108, 317–329 (2002).
Lampson, M. A., Renduchitala, K., Khodjakov, A. & Kapoor, T. M. Correcting improper chromosome-spindle attachments during cell division. Nat. Cell Biol. 6, 232–237 (2004).
Maure, J-F., Kitamura, E. & Tanaka, T. U. Mps1 kinase promotes sister-kinetochore bi-orientation by a tension-dependent mechanism. Curr. Biol. 17, 2175–2182 (2007).
Santaguida, S. et al. Dissecting the role of MPS1 in chromosome biorientation and the spindle checkpoint through the small molecule inhibitor reversine. J. Cell Biol. 190, 73–87 (2010).
Maresca, T. J. & Salmon, E. D. Welcome to a new kind of tension: translating kinetochore mechanics into a wait-anaphase signal. J. Cell Sci. 123, 825–835 (2010).
Foley, E. A., Maldonado, M. & Kapoor, T. M. Formation of stable attachments between kinetochores and microtubules depends on the B56-PP2A phosphatase. Nat. Cell Biol. 13, 1265–1271 (2011).
Meadows, J. C. et al. Spindle checkpoint silencing requires association of PP1 to both Spc7 and kinesin-8 motors. Dev. Cell 20, 739–750 (2011).
Rosenberg, J. S., Cross, F. R. & Funabiki, H. KNL1/Spc105 recruits PP1 to silence the spindle assembly checkpoint. Curr. Biol. 21, 942–947 (2011).
De Wulf, P., McAinsh, A. D. & Sorger, P. K. Hierarchical assembly of the budding yeast kinetochore from multiple subcomplexes. Genes Dev. 17, 2902–2921 (2003).
Lechner, J. & Carbon, J. A 240 kd multisubunit protein complex, CBF3, is a major component of the budding yeast centromere. Cell 64, 717–725 (1991).
Coffman, V. C., Wu, P., Parthun, M. R. & Wu, J-Q. CENP-A exceeds microtubule attachment sites in centromere clusters of both budding and fission yeast. J. Cell Biol. 195, 563–572 (2011).
Lawrimore, J., Bloom, K. S. & Salmon, E. D. Point centromeres contain more than a single centromere-specific Cse4 (CENP-A) nucleosome. J. Cell Biol. 195, 573–582 (2011).
Ubersax, J. A. et al. Targets of the cyclin dependent kinase Cdk1. Nature 425, 859–864 (2003).
Loog, M. & Morgan, D. O. Cyclin specificity in the phosphorylation of cyclin-dependent kinase substrates. Nature 434, 104–108 (2005).
Breitkreutz, A. et al. A global protein kinase and phosphatase interaction network in yeast. Science 328, 1043–1046 (2010).
Cheeseman, I. M. et al. Phospho-regulation of kinetochore-microtubule attachments by the aurora kinase Ipl1p. Cell 111, 163–172 (2002).
De Wulf, P., Montani, F. & Visintin, R. Protein phosphatases take the mitotic stage. Curr. Opin. Cell Biol. 21, 806–815 (2009).
Wei, R. R., Sorger, P. K. & Harrison, S. C. Molecular organization of the Ndc80 complex, an essential kinetochore component. Proc. Natl Acad. Sci. USA 102, 5363–5367 (2005).
Lingelbach, L. B. & Kaplan, K. B. The interaction between Sgt1p and Skp1p is regulated by HSP90 chaperones and is required for proper CBF3 assembly. Mol. Cell. Biol. 24, 8938–8950 (2004).
Blake-Hodek, K. A., Cassimeris, L. & Huffaker, T. C. Regulation of microtubule dynamics by Bim1 and Bik1, the budding yeast members of the EB1 and CLIP-170 families of plus-end tracking proteins. Mol. Biol. Cell 21, 2013–2023 (2010).
Wong, J. et al. A protein interaction map of the mitotic spindle. Mol. Biol. Cell 18, 3800–3809 (2007).
Schuyler, S. C. & Pellman, D. Analysis of the size and shape of protein complexes from yeast. Meth. Enzymol. 351, 150–168 (2002).
Gillett, E. S., Espelin, C. W. & Sorger, P. K. Spindle checkpoint proteins and chromosome-microtubule attachment in budding yeast. J. Cell Biol. 164, 535–546 (2004).
Pagliuca, C., Draviam, V. M., Marco, E., Sorger, P. K. & De Wulf, P. Roles for the conserved Spc105p-Kre28p complex in kinetochore-microtubule binding and the spindle assembly checkpoint. PLoS ONE 4, e7640 (2009).
Krenn, V. et al. Structural analysis reveals features of the spindle checkpoint kinase Bub1-kinetochore subunit Knl1 interaction. J. Cell Biol. 196, 451–467 (2012).
Kitamura, E., Tanaka, K., Kitamura, Y. & Tanaka, T. U. Kinetochore microtubule interaction during S phase in Saccharomyces cerevisiae. Genes Dev. 21, 3319–3330 (2007).
Koshland, D. & Hieter, P. Visual assay for chromosome ploidy. Meth. Enzymol. 155, 351–372 (1987).
Tong, A. H. & Boone, C. Synthetic genetic array analysis in Saccharomyces cerevisiae. Meth. Mol. Biol. 313, 171–192 (2006).
Tien, J. F. et al. Cooperation of the Dam1 and Ndc80 kinetochore complexes enhances microtubule coupling and is regulated by aurora B. J. Cell Biol. 189, 713–723 (2010).
Lampert, F., Hornung, P. & Westermann, S. The Dam1 complex confers microtubule plus end-tracking activity to the Ndc80 kinetochore complex. J. Cell Biol. 189, 641–649 (2010).
Maskell, D. P., Hu, X-W. & Singleton, M. R. Molecular architecture and assembly of the yeast kinetochore MIND complex. J. Cell Biol. 190, 823–834 (2010).
Schleiffer, A. et al. CENP-T proteins are conserved centromere receptors of the Ndc80 complex. Nat. Cell Biol.http://dx.doi.org/10.1038/ncb2493 (2012).
Perpelescu, M. & Fukagawa, T. The ABCs of CENPs. Chromosoma 120, 425–446 (2011).
Nishino, T. et al. CENP-T-W-S-X forms a unique centromeric chromatin structure with a histone-like fold. Cell 148, 487–501 (2012).
Gascoigne, K. E. et al. Induced ectopic kinetochore assembly bypasses the requirement for CENP-A nucleosomes. Cell 145, 410–422 (2011).
Przewloka, M. R. et al. CENP-C is a structural platform for kinetochore assembly. Curr. Biol 21, 399–405 (2011).
Screpanti, E. et al. Direct binding of Cenp-C to the Mis12 complex joins the inner and outer kinetochore. Curr. Biol. 21, 391–398 (2011).
Liu, S-T., Rattner, J. B., Jablonski, S. A. & Yen, T. J. Mapping the assembly pathways that specify formation of the trilaminar kinetochore plates in human cells. J. Cell Biol. 175, 41–53 (2006).
Ohkuni, K., Abdulle, R., Tong, A. H., Boone, C. & Kitagawa, K. Ybp2 associates with the central kinetochore of Saccharomyces cerevisiae and mediates proper mitotic progression. PLoS ONE 3, e1617 (2008).
Bermejo, R., Katou, Y. M., Shirahige, K. & Foiani, M. ChIP-on-chip analysis of DNA topoisomerases. Meth. Mol. Biol. 582, 103–118 (2009).
Akiyoshi, B. et al. Tension directly stabilizes reconstituted kinetochore-microtubule attachments. Nature 468, 576–579 (2010).
Cho, R. J. et al. Parallel analysis of genetic selections using whole genome oligonucleotide arrays. Proc. Natl. Acad. Sci. USA 95, 3752–3757 (1998).
Holinger, E. P. et al. Budding yeast centrosome duplication requires stabilization of Spc29 via Mps1-mediated phosphorylation. J. Biol. Chem. 284, 12949–12955 (2009).
Cox, J. & Mann, M. MaxQuant enables high peptide identification rates, individualized p.p.b.-range mass accuracies and proteome-wide protein quantification. Nat. Biotechnol. 26, 1367–1372 (2008).
Acknowledgements
The authors thank A. Amon (MIT, USA), S. Biggins (University of Washington, USA), D. Koshland (University of California, Berkeley, USA), M. Singleton (Cancer Research UK), F. Solomon (MIT, USA), P. K. Sorger (Harvard Medical School, USA) and M. Winey (University of Colorado, USA) for strains or reagents. We also thank M. Foiani, S. Ghandi, D. Lamont, J. Lechner, C. Lucca, J. Ortiz, S. ten Have and R. Visintin for technical support or discussions. We thank A. Schleiffer and S. Westermann for communicating results before publication. P.D.W. acknowledges financial support from the Italian Association for Cancer Research (grant 8840). T.R.H. recognizes support from the N.I.H. (grant GM087461) and the American Cancer Society (grant IRG 58-006-50). T.U.T. acknowledges a Cancer Research U.K. senior fellowship and Wellcome Trust program grant. L.J.B. acknowledges a doctoral fellowship from the European School of Molecular Medicine.
Author information
Authors and Affiliations
Contributions
L.J.B. and C.P. performed all the genetic, molecular biological, biochemical and cell biological experiments. N.K. performed the time-lapse CEN-reactivation and Cnn1–3GFP imaging experiments, R.A.G. performed the Y2H screens, Y.O. performed the in vitro kinase experiments, K.S. performed the in vitro Cnn1–KMN binding experiments, C.A. helped with the genetic interaction screens and the ChIP–chip experiment, C.G. and M.D.M. performed the cell-cycle experiments, PhosTag western hybridization and Cnn1–Cnn1 co-immunoprecipitation experiments. A.O. performed the FRAP experiments and helped with the quantification of Cnn1–3GFP and Ndc80–3GFP at kinetochores, R.B. helped with the ChIP–chip experiment and converted the microarray data into graphic format, T.R.H. supervised the Y2H screens and the in vitro Cnn1–KMN binding experiments. T.U.T. supervised the live-cell imaging experiments. P.D.W. supervised the project, helped with yeast imaging and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 800 kb)
Rights and permissions
About this article
Cite this article
Bock, L., Pagliuca, C., Kobayashi, N. et al. Cnn1 inhibits the interactions between the KMN complexes of the yeast kinetochore. Nat Cell Biol 14, 614–624 (2012). https://doi.org/10.1038/ncb2495
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb2495
This article is cited by
-
Bridgin connects the outer kinetochore to centromeric chromatin
Nature Communications (2021)
-
Cell-cycle phospho-regulation of the kinetochore
Current Genetics (2021)
-
Multiple phosphorylations control recruitment of the KMN network onto kinetochores
Nature Cell Biology (2018)
-
Regulation of kinetochore configuration during mitosis
Current Genetics (2018)
-
Kinetochore assembly and function through the cell cycle
Chromosoma (2016)