Abstract
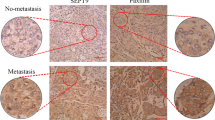
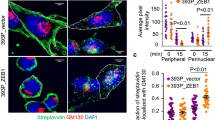
Cell migration driven by the epidermal growth factor receptor (EGFR) propels morphogenesis1 and involves reorganization of the actin cytoskeleton2. Although de novo transcription precedes migration3,4, transcript identity remains largely unknown. Through their actin-binding domains, tensins link the cytoskeleton to integrin-based adhesion sites5. Here we report that EGF downregulates tensin-3 expression, and concomitantly upregulates cten, a tensin family member that lacks the actin-binding domain6. Knockdown of cten or tensin-3, respectively, impairs or enhances mammary cell migration. Furthermore, cten displaces tensin-3 from the cytoplasmic tail of integrin β1, thereby instigating actin fibre disassembly. In invasive breast cancer, cten expression correlates not only with high EGFR and HER2, but also with metastasis to lymph nodes. Moreover, treatment of inflammatory breast cancer patients with an EGFR/HER2 dual-specificity kinase inhibitor significantly downregulated cten expression. In conclusion, a transcriptional tensin-3–cten switch may contribute to the metastasis of mammary cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
References
Wells, A., Kassis, J., Solava, J., Turner, T. & Lauffenburger, D. A. Growth factor-induced cell motility in tumor invasion. Acta Oncol. 41, 124–130 (2002).
Ridley, A. J. et al. Cell migration: integrating signals from front to back. Science 302, 1704–1709 (2003).
Bauer, J. et al. In vitro model of angiogenesis using a human endothelium-derived permanent cell line: contributions of induced gene expression, G-proteins, and integrins. J. Cell Physiol. 153, 437–449 (1992).
Gordon, S. R. & Staley, C. A. Role of the cytoskeleton during injury-induced cell migration in corneal endothelium. Cell Motil. Cytoskeleton 16, 47–57 (1990).
Lo, S. H. Tensin. Int. J. Biochem. Cell Biol. 36, 31–34 (2004).
Lo, S. H. & Lo, T. B. Cten, a COOH-terminal tensin-like protein with prostate restricted expression, is down-regulated in prostate cancer. Cancer Res. 62, 4217–4221 (2002).
Yarden, Y. & Sliwkowski, M. X. Untangling the ErbB signalling network. Nature Rev. Mol. Cell Biol. 2, 127–137 (2001).
Ridley, A. J. & Hall, A. The small GTP-binding protein rho regulates the assembly of focal adhesions and actin stress fibers in response to growth factors. Cell 70, 389–399 (1992).
Malliri, A. et al. The transcription factor AP-1 is required for EGF-induced activation of rho-like GTPases, cytoskeletal rearrangements, motility, and in vitro invasion of A431 cells. J. Cell Biol. 143, 1087–1099 (1998).
Manos, E. J. et al. Dolichol-phosphate-mannose-3 (DPM3)/prostin-1 is a novel phospholipase C-gamma regulated gene negatively associated with prostate tumor invasion. Oncogene 20, 2781–2790 (2001).
Amit, I. et al. A module of negative feedback regulators defines growth factor signaling. Nature Genet. 39, 503–512 (2007).
Guo, W. & Giancotti, F. G. Integrin signalling during tumour progression. Nature Rev. Mol. Cell Biol. 5, 816–826 (2004).
Irie, H. Y. et al. Distinct roles of Akt1 and Akt2 in regulating cell migration and epithelial-mesenchymal transition. J. Cell Biol. 171, 1023–1034 (2005).
Geiger, B., Bershadsky, A., Pankov, R. & Yamada, K. M. Transmembrane crosstalk between the extracellular matrix–cytoskeleton crosstalk. Nature Rev. Mol. Cell Biol. 2, 793–805 (2001).
Rodrigue, C. M. et al. The cancer chemopreventive agent resveratrol induces tensin, a cell-matrix adhesion protein with signaling and antitumor activities. Oncogene 24, 3274–3284 (2005).
Kook, S. et al. Caspase-dependent cleavage of tensin induces disruption of actin cytoskeleton during apoptosis. Biochem. Biophys. Res. Commun. 303, 37–45 (2003).
Garcia-Alvarez, B. et al. Structural determinants of Integrin recognition by Talin. Mol. Cell 11, 49–58 (2003).
Wu, M. & Merajver, S. D. Molecular biology of inflammatory breast cancer: applications to diagnosis, prognosis, and therapy. Breast Dis. 22, 25–34 (2005).
Allred, D. C. et al. HER-2/neu in node-negative breast cancer: prognostic significance of overexpression influenced by the presence of in situ carcinoma. J. Clin. Oncol. 10, 599–605 (1992).
Xia, W. et al. Anti-tumor activity of GW572016: a dual tyrosine kinase inhibitor blocks EGF activation of EGFR/erbB2 and downstream Erk1/2 and AKT pathways. Oncogene 21, 6255–6263 (2002).
Spector, N. L. et al. Study of the biologic effects of lapatinib, a reversible inhibitor of ErbB1 and ErbB2 tyrosine kinases, on tumor growth and survival pathways in patients with advanced malignancies. J. Clin. Oncol. 23, 2502–2512 (2005).
Thiery, J. P. Epithelial–mesenchymal transitions in tumour progression. Nature Rev. Cancer 2, 442–454 (2002).
Zamir, E. et al. Dynamics and segregation of cell–matrix adhesions in cultured fibroblasts. Nature Cell Biol. 2, 191–196 (2000).
Albiges-Rizo, C., Frachet, P. & Block, M. R. Downregulation of talin alters cell adhesion and the processing of the α5β1 integrin. J. Cell Sci. 108, 3317–3329 (1995).
Hazan, R. B., Qiao, R., Keren, R., Badano, I. & Suyama, K. Cadherin switch in tumor progression. Ann. NY Acad. Sci. 1014, 155–163 (2004).
Friedl, P. & Wolf, K. Tumour-cell invasion and migration: diversity and escape mechanisms. Nature Rev. Cancer 3, 362–374 (2003).
Sasaki, H., Yukiue, H., Kobayashi, Y., Fukai, I. & Fujii, Y. Cten mRNA expression is correlated with tumor progression in thymoma. Tumour Biol. 24, 271–274 (2003).
Sasaki, H. et al. Cten mRNA expression was correlated with tumor progression in lung cancers. Lung Cancer 40, 151–155 (2003).
Cui, Y., Liao, Y. C. & Lo, S. H. Epidermal growth factor modulates tyrosine phosphorylation of a novel tensin family member, tensin3. Mol. Cancer Res. 2, 225–232 (2004).
Calderwood, D. A. et al. Integrinβ cytoplasmic domain interactions with phosphotyrosine-binding domains: a structural prototype for diversity in integrin signaling. Proc. Natl Acad. Sci. USA 100, 2272–2277 (2003).
Acknowledgements
We thank Kenneth Yamada, Shin Lin and Mark Ginsberg for plasmids; Dalia Seger and Hadassa Degani for guidance with xenografts; Miriam Eisenstein for structure prediction; and Benjamin Geiger and Alexander Bershadsky for critical comments. Our laboratory is supported by research grants from the Israel Cancer Research Fund, the German Israel Foundation, the Prostate Cancer Foundation, the European Commission and the National Cancer Institute (grants CA72981 and CA102537). Y.Y. is the incumbent of the Harold and Zelda Goldenberg Professorial Chair. T.S. was supported in part by the Ridgefield Foundation, the Israel Science Fund and the European Commission (EC FP6). S.C. is the recipient of a grant from Fundação para Ciência e Tecnologia (FCT), Lisboa, Portugal.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figure S1, S2, S3, S4 and Table S1 (PDF 1308 kb)
Rights and permissions
About this article
Cite this article
Katz, M., Amit, I., Citri, A. et al. A reciprocal tensin-3–cten switch mediates EGF-driven mammary cell migration. Nat Cell Biol 9, 961–969 (2007). https://doi.org/10.1038/ncb1622
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1622
This article is cited by
-
STR mutations on chromosome 15q cause thyrotropin resistance by activating a primate-specific enhancer of MIR7-2/MIR1179
Nature Genetics (2024)
-
Nuclear-localized CTEN is a novel transcriptional regulator and promotes cancer cell migration through its downstream target CDC27
Journal of Physiology and Biochemistry (2023)
-
Metallic nanoparticles reduce the migration of human fibroblasts in vitro
Nanoscale Research Letters (2017)
-
Curcumin inhibits growth of human breast cancer cells through demethylation of DLC1 promoter
Molecular and Cellular Biochemistry (2017)
-
ΔNp63α expression induces loss of cell adhesion in triple-negative breast cancer cells
BMC Cancer (2016)