Abstract
Junctional epidermolysis bullosa (JEB) is a severe and often lethal genetic disease caused by mutations in genes encoding the basement membrane component laminin-332. Surviving patients with JEB develop chronic wounds to the skin and mucosa, which impair their quality of life and lead to skin cancer. Here we show that autologous transgenic keratinocyte cultures regenerated an entire, fully functional epidermis on a seven-year-old child suffering from a devastating, life-threatening form of JEB. The proviral integration pattern was maintained in vivo and epidermal renewal did not cause any clonal selection. Clonal tracing showed that the human epidermis is sustained not by equipotent progenitors, but by a limited number of long-lived stem cells, detected as holoclones, that can extensively self-renew in vitro and in vivo and produce progenitors that replenish terminally differentiated keratinocytes. This study provides a blueprint that can be applied to other stem cell-mediated combined ex vivo cell and gene therapies.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Fine, J. D. et al. Inherited epidermolysis bullosa: updated recommendations on diagnosis and classification. J. Am. Acad. Dermatol. 70, 1103–1126 (2014)
Fine, J. D., Johnson, L. B., Weiner, M. & Suchindran, C. Cause-specific risks of childhood death in inherited epidermolysis bullosa. J. Pediatr. 152, 276–280 (2008)
Barrandon, Y. & Green, H. Three clonal types of keratinocyte with different capacities for multiplication. Proc. Natl Acad. Sci. USA 84, 2302–2306 (1987)
Pellegrini, G. et al. Location and clonal analysis of stem cells and their differentiated progeny in the human ocular surface. J. Cell Biol. 145, 769–782 (1999)
Gallico, G. G. III, O’Connor, N. E., Compton, C. C., Kehinde, O. & Green, H. Permanent coverage of large burn wounds with autologous cultured human epithelium. N. Engl. J. Med. 311, 448–451 (1984)
Pellegrini, G. et al. The control of epidermal stem cells (holoclones) in the treatment of massive full-thickness burns with autologous keratinocytes cultured on fibrin. Transplantation 68, 868–879 (1999)
Pellegrini, G. et al. Long-term restoration of damaged corneal surfaces with autologous cultivated corneal epithelium. Lancet 349, 990–993 (1997)
Rama, P. et al. Limbal stem-cell therapy and long-term corneal regeneration. N. Engl. J. Med. 363, 147–155 (2010)
Ronfard, V., Rives, J. M., Neveux, Y., Carsin, H. & Barrandon, Y. Long-term regeneration of human epidermis on third degree burns transplanted with autologous cultured epithelium grown on a fibrin matrix. Transplantation 70, 1588–1598 (2000)
Bauer, J. W. et al. Closure of a large chronic wound through transplantation of gene-corrected epidermal stem cells. J. Invest. Dermatol. 137, 778–781 (2017)
De Rosa, L. et al. Long-term stability and safety of transgenic cultured epidermal stem cells in gene therapy of junctional epidermolysis bullosa. Stem Cell Reports 2, 1–8 (2013)
Mavilio, F. et al. Correction of junctional epidermolysis bullosa by transplantation of genetically modified epidermal stem cells. Nat. Med. 12, 1397–1402 (2006)
Markowitz, D., Goff, S. & Bank, A. Construction and use of a safe and efficient amphotropic packaging cell line. Virology 167, 400–406 (1988)
De Luca, M., Pellegrini, G. & Green, H. Regeneration of squamous epithelia from stem cells of cultured grafts. Regen. Med. 1, 45–57 (2006)
Chapman, D. G. & Robbins, H. Minimum variance estimation without regularity assumptions. Ann. Math. Stat. 22, 581–586 (1951)
Hanahan, D. & Weinberg, R. A. Hallmarks of cancer: the next generation. Cell 144, 646–674 (2011)
Aiuti, A. et al. Gene therapy for immunodeficiency due to adenosine deaminase deficiency. N. Engl. J. Med. 360, 447–458 (2009)
Biasco, L. et al. Integration profile of retroviral vector in gene therapy treated patients is cell-specific according to gene expression and chromatin conformation of target cell. EMBO Mol. Med. 3, 89–101 (2011)
Cavazza, A. et al. Self-inactivating MLV vectors have a reduced genotoxic profile in human epidermal keratinocytes. Gene Ther. 20, 949–957 (2013)
Hacein-Bey-Abina, S. et al. Insertional oncogenesis in 4 patients after retrovirus-mediated gene therapy of SCID-X1. J. Clin. Invest. 118, 3132–3142 (2008)
Hacein-Bey-Abina, S. et al. A serious adverse event after successful gene therapy for X-linked severe combined immunodeficiency. N. Engl. J. Med. 348, 255–256 (2003)
Howe, S. J. et al. Insertional mutagenesis combined with acquired somatic mutations causes leukemogenesis following gene therapy of SCID-X1 patients. J. Clin. Invest. 118, 3143–3150 (2008)
Siprashvili, Z. et al. Safety and wound outcomes following genetically corrected autologous epidermal grafts in patients with recessive dystrophic epidermolysis bullosa. J. Am. Med. Assoc. 316, 1808–1817 (2016)
Hsu, Y. C., Li, L. & Fuchs, E. Transit-amplifying cells orchestrate stem cell activity and tissue regeneration. Cell 157, 935–949 (2014)
Clayton, E. et al. A single type of progenitor cell maintains normal epidermis. Nature 446, 185–189 (2007)
Mascré, G. et al. Distinct contribution of stem and progenitor cells to epidermal maintenance. Nature 489, 257–262 (2012)
Pellegrini, G. et al. Biological parameters determining the clinical outcome of autologous cultures of limbal stem cells. Regen. Med. 8, 553–567 (2013)
Biasco, L. et al. In vivo tracking of human hematopoiesis reveals patterns of clonal dynamics during early and steady-state reconstitution phases. Cell Stem Cell 19, 107–119 (2016)
Schwieger-Briel, A. et al. Instrument for scoring clinical outcome of research for epidermolysis bullosa: a consensus-generated clinical research tool. Pediatr. Dermatol. 32, 41–52 (2015)
Herndon, D. N. et al. Long-term propranolol use in severely burned pediatric patients: a randomized controlled study. Ann. Surg. 256, 402–411 (2012)
Goldschneider, K. R. et al. Pain care for patients with epidermolysis bullosa: best care practice guidelines. BMC Med. 12, 178 (2014)
Rodriguez, N. A., Jeschke, M. G., Williams, F. N., Kamolz, L. P. & Herndon, D. N. Nutrition in burns: Galveston contributions. JPEN J. Parenter. Enteral Nutr. 35, 704–714 (2011)
Dellambra, E. et al. Corrective transduction of human epidermal stem cells in laminin-5-dependent junctional epidermolysis bullosa. Hum. Gene Ther. 9, 1359–1370 (1998)
Mathor, M. B. et al. Clonal analysis of stably transduced human epidermal stem cells in culture. Proc. Natl Acad. Sci. USA 93, 10371–10376 (1996)
Guerra, L. et al. Treatment of “stable” vitiligo by timed surgery and transplantation of cultured epidermal autografts. Arch. Dermatol. 136, 1380–1389 (2000)
Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet.journal 17, 10–12 (2011)
Li, H. Aligning sequence reads, clone sequences and assembly contigs with BWA-MEM. Preprint at https://arxiv.org/abs/1303.3997 (2013)
Quinlan, A. R. BEDTools: the Swiss-army tool for genome feature analysis. Curr. Protoc. Bioinformatics 47, 11–34 (2014)
Chapman, D. G. Some Properties of the Hypergeometric Distribution with Applications to Zoological Sample Censuses (Univ. California Press, 1951)
Yu, G., Wang, L. G. & He, Q. Y. ChIPseeker: an R/Bioconductor package for ChIP peak annotation, comparison and visualization. Bioinformatics 31, 2382–2383 (2015)
Yu, G., Wang, L. G., Han, Y. & He, Q. Y. clusterProfiler: an R package for comparing biological themes among gene clusters. OMICS 16, 284–287 (2012)
Cavazza, A. et al. Dynamic transcriptional and epigenetic regulation of human epidermal keratinocyte differentiation. Stem Cell Reports 6, 618–632 (2016)
Akagi, K., et al. RTCGD: retroviral tagged cancer gene database. Nucleic Acids Res. 32, 523–527 (2004)
Acknowledgements
Holostem Terapie Avanzate s.r.l. met all costs of GMP production and procedures of transgenic epidermal grafts. This work was partially supported by the Italian Ministry of Education, University and Research (MIUR), no. CTN01_00177_888744; Regione Emilia-Romagna, Asse 1 POR-FESR 2007-13; Fondazione Cassa di Risparmio di Modena; DEBRA Südtirol - Alto Adige; DEBRA Austria; European Research Council (ERC) under the European Union’s Horizon 2020 Research and Innovation Program (grant agreement no. 670126-DENOVOSTEM); ERC under the European Union’s Seventh Framework Programme (grant agreement no. 294780-NOVABREED); and Epigenetics Flagship project CNR-MIUR grants. We thank H. Green for continuous support; O. Goertz for his contribution to the surgical procedures; the Department of Anaesthesiology, in particular P. Zahn and T. Maecken, and the entire OR staff, in particular S. Taszarski and V. Stroh, for their dedicated perioperative care; the nurses of ward PÄD1 for continuous and devoted assistance; A. Neumayer and J. Frank for technical assistance in defining clone integrations; B. Mussnig for performing indirect immunofluorescence; M. C. Latella for determining the average number of integrations in pre- and post-graft cultures; M. Forcato for feedback on the bioinformatics analyses; and G. De Santis for control skin specimens.
Author information
Authors and Affiliations
Contributions
T.H., T.R., N.T., J.W.B. and G.P. defined strategic procedures and performed transplantation of the transgenic grafts, surgical and medical procedures and clinical follow-up; L.D.R. performed immunofluorescence data and imaging analysis, analysed the data and assembled all input data, prepared the figures and edited the manuscript; D.S., I.J. and M.M. performed integration profiles of transgenic epidermis; R.C., J.R., A.K. and D.K. performed clonal tracing in epidermal cells; O.R. and S.Bi. conducted all bioinformatics analyses; A.S.S. and E.E. performed in situ hybridization; S.C. and S.Bo. performed all culture procedures and preparation of genetically modified epidermal graft; F.J., T.L., M.L., M.F. and M.K. carried out the follow-up on the patient; D.Q. performed electron microscopy analysis; M.D.L. coordinated the study, defined strategic procedures, administered the experiments and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
G.P. and M.D.L. are co-founders and members of the Board of Directors of Holostem Terapie Avanzate (HTA), s.r.l, Modena, Italy; Chiesi Farmaceutici S.p.A. (a co-founder of HTA), holds an Orphan Drug Medicinal Product designation (EU/3/15/1465) for the transgenic cultures used in this paper.
Additional information
Reviewer Information Nature thanks A. Aiuti, C. Blanpain, D. Strech and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
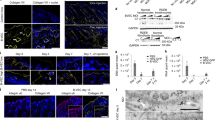
Extended Data Figure 1 Schematic representation of combined ex vivo cell and gene therapy.
The scheme shows the entire procedure, from skin biopsy to transplantation and follow-up. The total number of keratinocytes, corresponding clonogenic fraction and days of cultivation are shown for each passage. All analyses performed at each follow-up are indicated. Immunofluorescence (IF), in situ hybridization (ISH) and transmission electron microscopy (TEM) were performed on randomly taken 0.2–0.4-mm2 punch biopsies. Genome-wide analysis (NGS) was performed on pre-graft cultures (PGc) and on primary cultures initiated from approximately 0.5-cm2 biopsies taken from the left leg (4Mc and 8Mc2) and the left arm (8Mc1). Clonal analysis and tracing were performed on PGc, 4Mc and 8Mc1.
Extended Data Figure 2 Regeneration of the epidermis by transduced keratinocyte cultures.
a, Preparation of a dermal wound bed at the time of transplantation. b, Transplantation onto the left arm of plastic-cultured epidermal grafts, mounted on a non-adhering gauze (asterisks). c, The engrafted epidermis (asterisks) is evident upon removal of the gauze (arrows) ten days after grafting. d, Regenerated epidermis on the left arm after 1 month. e, f, Transplantation (e) and engraftment (f) of both plastic-cultured (asterisk) and fibrin-cultured (arrow and inset in e) grafts on the left leg. f, Inset, complete epidermal regeneration is evident after 1 month. g, The back of the patient was covered with fibrin-cultured grafts (inset). h, Complete epidermal regeneration was observed after 1 month, with the exception of areas marked with asterisks. Islands of epidermis were observed inside those denuded areas (arrows). i, Within 4 months, the regenerated epidermis surrounding the open lesions and the epidermal islands detected within those open lesions had spread and covered the denuded areas.
Extended Data Figure 3 Restoration of a normal dermis–epidermis junction.
a, Haematoxylin and eosin staining of skin sections (7-μm thick) prepared from normal skin and from the patient at admission and at 4, 8 and 21 month follow-ups. Black arrows show ruptures at the epidermis–dermis junction. Scale bar, 20 μm. b, Sections (7-μm thick) from normal skin, the patient’s skin at admission and the patient’s skin 21 months after transplantation were immunostained using antibodies against laminin 332-α3, laminin 332-γ2, α6 integrin and β4 integrin. c, Adhesion of cohesive cultured epidermal sheets. Top, spontaneous detachment (arrow) of confluent culture of laminin 332-β3 null keratinocytes from the patient. Bottom, culture of patient’s genetically corrected keratinocytes remained firmly attached to the substrate. As with normal control cells, detachment would have required prolonged enzymatic treatment. d, The absence of a humoral immune response to the transgene product was verified by indirect immunofluorescence performed on monkey oesophagus and normal human split skin (NH-SS) sections, using the patient’s plasma taken 21 months after transplantation. An anti-human laminin-332 antibody (anti-GB3) was used as a positive control (C+). A healthy donor’s plasma was used as negative control (C−). Arrows denote the expected localization of the laminin-332 labelling. Scale bar, 100 μm.
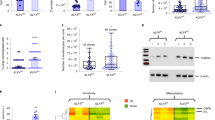
Extended Data Figure 4 Schematic model of epidermal stem and progenitor cell function.
Clonogenic progenitors (blue cells) contained in the original skin biopsy and in 8,472 cm2 of transgenic epidermis are indicated. Stem cells, detected as holoclones (pink cells), were identified by clonal analysis (Methods and Extended Data Fig. 5). The number of holoclones contained in the primary culture has been estimated. The schematic model posits the existence of specific long-lived stem cells generating pools of short-lived progenitors (Hypothesis 1) or a population of equipotent epidermal progenitors (Hypothesis 2). The number of integrations predicted by the Chapman–Wilson capture–re-capture model and formally detected by NGS analysis in 4Mc, 8Mc1 and 8Mc2 (right) is consistent with the number of transplanted holoclones and therefore supports Hypothesis 1.
Extended Data Figure 5 Clonal analysis scheme.
Sub-confluent cultures were trypsinized, serially diluted and inoculated (0.5 cells per well) onto 96-multiwell plates containing irradiated 3T3-J2 cells. After 7 days of cultivation, single clones were identified under an inverted microscope (scale bar, 100 μm), trypsinized, transferred to two dishes and cultivated. One dish (one-quarter of the clone) was fixed 12 days later and stained with rhodamine B for the classification of clonal type. The clonal type was determined by the percentage of aborted colonies formed by the progeny of the founding cell. The clone was scored as a holoclone when 0–5% of colonies were terminal. When 95–100% of colonies were terminal (or when no colonies formed), the clone was classified as a paraclone. When the amount of terminal colonies was between 5% and 95%, the clone was classified as a meroclone. The second dish (three-quarters of the clone) was used for integration analysis after 7 days of cultivation.
Extended Data Figure 6 Determination of provirus copy number.
Quantitative PCR was performed on genomic DNA from pre-graft cultures (PGc), primary cultures generated at 4 months (4Mc) and 8 months (8Mc1, 8Mc2) follow-up and selected holoclones (PRE.G_H1, PRE.G_H10, FU4m_H1-11, PRE.G_H7). The experiment was performed in duplicate and the plot is presented as two individual data points for each sample.
Extended Data Figure 7 Schematic model of holoclone tracing in the regenerated patient’s epidermis.
Transgenic epidermal cultures (PGc) contain a mixed population of clonogenic basal stem cells (blue) and transient amplifying progenitors (grey). Upon engraftment and initial epidermal regeneration, both stem and transient amplifying cells can proliferate and eventually generate suprabasal terminally differentiated cells. Upon epidermal renewal (after 4 and 8 months), the short-lived transient amplifying progenitors (grey) are progressively lost. The long-lived stem cells then generate new pools of transient amplifying progenitors (now blue basal cells), which will produce terminally differentiated cells (suprabasal blue cells).
Extended Data Figure 8 Clinical data.
During his time in hospital, the patient’s inflammatory and nutritional status was documented by blood concentration of C-reactive protein (CRP; upper panel) and albumin (lower panel). The times of biopsy sampling (B) and epidermal culture transplantation are shown with arrows. The linear regressions visualize the trend of pre-graft (dotted) and post-graft (black line) progressions. The red line within the CRP time course demonstrates the CRP limit, which is considered a criterion for severe inflammation. These data demonstrate the critical situation of the patient at admission and before transplantation and the improvement of his general status upon epidermal regeneration.
Supplementary information
Supplementary Information
This file contains Supplementary Tables 1-3 and Supplementary Data. (PDF 70 kb)
Supplementary Table 4
A list of independent integrations identified by NGS analysis. The libraries of integrations were obtained using two independent LTR-primers (3pIN, 3pOUT). The file contains the list of independent integrations found in PGc, 4Mc, 8Mc1 and 8Mc2 and merged data (all_integrations) showing integrations retrieved across samples. (XLSX 1599 kb)
Mechanical stress
Removal of adherent dressing from sites of follow-up biopsies shows stability of the regenerated epidermis. (MOV 9088 kb)
Rights and permissions
About this article
Cite this article
Hirsch, T., Rothoeft, T., Teig, N. et al. Regeneration of the entire human epidermis using transgenic stem cells. Nature 551, 327–332 (2017). https://doi.org/10.1038/nature24487
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature24487
This article is cited by
-
Malignant features of minipig melanomas prior to spontaneous regression
Scientific Reports (2024)
-
Gene-edited cells: novel allogeneic gene/cell therapy for epidermolysis bullosa
Journal of Applied Genetics (2024)
-
Bioengineered skin organoids: from development to applications
Military Medical Research (2023)
-
Gene editing with ‘pencil’ rather than ‘scissors’ in human pluripotent stem cells
Stem Cell Research & Therapy (2023)
-
Bioengineering human intestinal mucosal grafts using patient-derived organoids, fibroblasts and scaffolds
Nature Protocols (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.