Abstract
Professional phagocytes (such as macrophages1) and non-professional phagocytes2,3,4,5,6,7,8 (such as epithelial cells) clear billions of apoptotic cells and particles on a daily basis9. Although professional and non-professional macrophages reside in proximity in most tissues, whether they communicate with each other during cell clearance, and how this might affect inflammation, is not known10. Here we show that macrophages, through the release of a soluble growth factor and microvesicles, alter the type of particles engulfed by non-professional phagocytes and influence their inflammatory response. During phagocytosis of apoptotic cells or in response to inflammation-associated cytokines, macrophages released insulin-like growth factor 1 (IGF-1). The binding of IGF-1 to its receptor on non-professional phagocytes redirected their phagocytosis, such that uptake of larger apoptotic cells was reduced whereas engulfment of microvesicles was increased. IGF-1 did not alter engulfment by macrophages. Macrophages also released microvesicles, whose uptake by epithelial cells was enhanced by IGF-1 and led to decreased inflammatory responses by epithelial cells. Consistent with these observations, deletion of IGF-1 receptor in airway epithelial cells led to exacerbated lung inflammation after allergen exposure. These genetic and functional studies reveal that IGF-1- and microvesicle-dependent communication between macrophages and epithelial cells can critically influence the magnitude of tissue inflammation in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Henson, P. M. & Hume, D. A. Apoptotic cell removal in development and tissue homeostasis. Trends Immunol . 27, 244–250 (2006)
Burstyn-Cohen, T. et al. Genetic dissection of TAM receptor-ligand interaction in retinal pigment epithelial cell phagocytosis. Neuron 76, 1123–1132 (2012)
Juncadella, I. J. et al. Apoptotic cell clearance by bronchial epithelial cells critically influences airway inflammation. Nature 493, 547–551 (2013)
Wood, W. et al. Mesenchymal cells engulf and clear apoptotic footplate cells in macrophageless PU.1 null mouse embryos. Development 127, 5245–5252 (2000)
Lu, Z. et al. Phagocytic activity of neuronal progenitors regulates adult neurogenesis. Nat. Cell Biol. 13, 1076–1083 (2011)
Ichimura, T. et al. Kidney injury molecule-1 is a phosphatidylserine receptor that confers a phagocytic phenotype on epithelial cells. J. Clin. Invest. 118, 1657–1668 (2008)
Mesa, K. R. et al. Niche-induced cell death and epithelial phagocytosis regulate hair follicle stem cell pool. Nature 522, 94–97 (2015)
Elliott, M. R. et al. Unexpected requirement for ELMO1 in clearance of apoptotic germ cells in vivo. Nature 467, 333–337 (2010)
Gregory, C. D. & Pound, J. D. Microenvironmental influences of apoptosis in vivo and in vitro. Apoptosis 15, 1029–1049 (2010)
Green, D. R. The end and after: how dying cells impact the living organism. Immunity 35, 441–444 (2011)
Werner, S. & Grose, R. Regulation of wound healing by growth factors and cytokines. Physiol. Rev. 83, 835–870 (2003)
Yakar, S. et al. Circulating levels of IGF-1 directly regulate bone growth and density. J. Clin. Invest. 110, 771–781 (2002)
Smith, T. J. Insulin-like growth factor-I regulation of immune function: a potential therapeutic target in autoimmune diseases? Pharmacol. Rev. 62, 199–236 (2010)
LeRoith, D. Insulin-like growth factor I receptor signaling—overlapping or redundant pathways? Endocrinology 141, 1287–1288 (2000)
Tosello-Trampont, A. C., Nakada-Tsukui, K. & Ravichandran, K. S. Engulfment of apoptotic cells is negatively regulated by Rho-mediated signaling. J. Biol. Chem. 278, 49911–49919 (2003)
Leverrier, Y. & Ridley, A. J. Requirement for Rho GTPases and PI 3-kinases during apoptotic cell phagocytosis by macrophages. Curr. Biol. 11, 195–199 (2001)
Tosello-Trampont, A. C., Brugnera, E. & Ravichandran, K. S. Evidence for a conserved role for CRKII and Rac in engulfment of apoptotic cells. J. Biol. Chem. 276, 13797–13802 (2001)
Schlam, D. et al. Phosphoinositide 3-kinase enables phagocytosis of large particles by terminating actin assembly through Rac/Cdc42 GTPase-activating proteins. Nat. Commun. 6, 8623 (2015)
Chen, F. et al. An essential role for TH2-type responses in limiting acute tissue damage during experimental helminth infection. Nat. Med. 18, 260–266 (2012)
Ginhoux, F. & Guilliams, M. Tissue-resident macrophage ontogeny and homeostasis. Immunity 44, 439–449 (2016)
Lambrecht, B. N. & Hammad, H. The immunology of asthma. Nat. Immunol. 16, 45–56 (2015)
Marini, M., Vittori, E., Hollemborg, J. & Mattoli, S. Expression of the potent inflammatory cytokines, granulocyte-macrophage-colony-stimulating factor and interleukin-6 and interleukin-8, in bronchial epithelial cells of patients with asthma. J. Allergy Clin. Immunol. 89, 1001–1009 (1992)
Zhou, B. et al. Thymic stromal lymphopoietin as a key initiator of allergic airway inflammation in mice. Nat. Immunol. 6, 1047–1053 (2005)
Gessner, A., Mohrs, K. & Mohrs, M. Mast cells, basophils, and eosinophils acquire constitutive IL-4 and IL-13 transcripts during lineage differentiation that are sufficient for rapid cytokine production. J. Immunol. 174, 1063–1072 (2005)
Sokol, C. L., Barton, G. M., Farr, A. G. & Medzhitov, R. A mechanism for the initiation of allergen-induced T helper type 2 responses. Nat. Immunol. 9, 310–318 (2008)
Bourdonnay, E. et al. Transcellular delivery of vesicular SOCS proteins from macrophages to epithelial cells blunts inflammatory signaling. J. Exp. Med. 212, 729–742 (2015)
Burger, D. et al. Microparticles: biomarkers and beyond. Clin. Sci. (Lond.) 124, 423–441 (2013)
Bissonnette, E. Y. et al. Fibroblast growth factor-2 is a sputum remodeling biomarker of severe asthma. J. Asthma 51, 119–126 (2014)
Zhang, J. et al. Pentraxin 3 (PTX3) expression in allergic asthmatic airways: role in airway smooth muscle migration and chemokine production. PLoS One 7, e34965 (2012)
Chamberland, A., Madore, A. M., Tremblay, K., Laviolette, M. & Laprise, C. A comparison of two sets of microarray experiments to define allergic asthma expression pattern. Exp. Lung Res. 35, 399–410 (2009)
Perl, A. K., Zhang, L. & Whitsett, J. A. Conditional expression of genes in the respiratory epithelium in transgenic mice: cautionary notes and toward building a better mouse trap. Am. J. Respir. Cell Mol. Biol. 40, 1–3 (2009)
Dietrich, P., Dragatsis, I., Xuan, S., Zeitlin, S. & Efstratiadis, A. Conditional mutagenesis in mice with heat shock promoter-driven cre transgenes. Mamm. Genome 11, 196–205 (2000)
Liu, J. L., Yakar, S. & LeRoith, D. Conditional knockout of mouse insulin-like growth factor-1 gene using the Cre/loxP system. Proc. Soc. Exp. Biol. Med. 223, 344–351 (2000)
Liu, J. L. et al. Insulin-like growth factor-I affects perinatal lethality and postnatal development in a gene dosage-dependent manner: manipulation using the Cre/loxP system in transgenic mice. Mol. Endocrinol. 12, 1452–1462 (1998)
Hahn, Y. S. et al. Vγ4+ γδ T cells regulate airway hyperreactivity to methacholine in ovalbumin-sensitized and challenged mice. J. Immunol. 171, 3170–3178 (2003)
Yeager, M., Berriman, J. A., Baker, T. S. & Bellamy, A. R. Three-dimensional structure of the rotavirus haemagglutinin VP4 by cryo-electron microscopy and difference map analysis. EMBO J . 13, 1011–1018 (1994)
Casella, J. F., Flanagan, M. D. & Lin, S. Cytochalasin D inhibits actin polymerization and induces depolymerization of actin filaments formed during platelet shape change. Nature 293, 302–305 (1981)
Yarmola, E. G., Somasundaram, T., Boring, T. A., Spector, I. & Bubb, M. R. Actin-latrunculin A structure and function. Differential modulation of actin-binding protein function by latrunculin A. J. Biol. Chem. 275, 28120–28127 (2000)
Hetrick, B., Han, M. S., Helgeson, L. A. & Nolen, B. J. Small molecules CK-666 and CK-869 inhibit actin-related protein 2/3 complex by blocking an activating conformational change. Chem. Biol. 20, 701–712 (2013)
Mulvihill, M. J. et al. Discovery of OSI-906: a selective and orally efficacious dual inhibitor of the IGF-1 receptor and insulin receptor. Future Med. Chem. 1, 1153–1171 (2009)
García-Echeverría, C. et al. In vivo antitumor activity of NVP-AEW541—a novel, potent, and selective inhibitor of the IGF-IR kinase. Cancer Cell 5, 231–239 (2004)
Acknowledgements
The authors thank members of the Ravichandran laboratory for discussions and critical reading of manuscript; S. Arandjelovic, K. Penberthy, and L. Haney for scoring histology slides, and J. Perry for assistance with generation of the heat map, and the UVa Flow Cytometry Core and the UVa Molecular Electron Microscopy Core. This work is supported by grants to K.S.R. from the NIGMS (GM064709), NIMH (MH096484), NHLBI (P01HL120840), and Center for Cell Signalling at the University of Virginia, NHLBI (HL132287 and HL091127) to Y.M.S. and K23 HL12610 to U.E. Additional support was provided via the NIH Training Grants T32 GM008136 (Cell and Molecular Biology) to C.Z.H.; T32 AI007496 (Immunology) to C.Z.H., M.W.B., and I.J.J.; and T32 GM007267 (MSTP) to M.W.B.
Author information
Authors and Affiliations
Contributions
C.Z.H. designed, performed and analysed most of the experiments in this study with input from K.S.R. I.J.J., J.M.K., M.W.B. assisted with in vivo experiments. A.L.K. provided phosphatidylserine liposomes. K.D. performed EM imaging of microvesicles. S.O-G ran the RNA-seq. U.E. performed microvesicle quantification using qNano. S.D.T. analysed the RNA-seq data. Y.M.S. performed airway resistance experiments. K.S.T. assisted in evaluation of lung pathology. C.Z.H. and K.S.R. wrote the manuscript with input from co-authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Reviewer Information Nature thanks L. Hedrick and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Extended data figures and tables
Extended Data Figure 1 IGF-1, but not EGF, VEGF, PDFG-AA/-BB, suppresses phagocytosis of apoptotic cells in non-professional phagocytes.
a, Representative engulfment assay in which LR73 cells were treated with indicated growth factors at increasing concentrations and assessed for engulfment of apoptotic thymocytes (n = 3). b–e, Serum-starved LR73 cells were stimulated with 100 ng ml−1 of specified growth factors for indicated time and the phosphorylation of Erk1/2 was determined by immunoblotting (n = 2). f, g, Representative engulfment assay in which the uptake of apoptotic thymocytes by 16HBE14o- human airway epithelial cells (f) or SVEC-40 endothelial cells (g) is dampened by IGF-1 treatment (n = 3). Error bars represent s.d., P value of <0.001 (***).
Extended Data Figure 2 Insulin and IGF-II also decrease apoptotic cell engulfment, similar to IGF-1 that is reversed by treatment with NVP-AEW541.
a, Left, engulfment of apoptotic thymocytes by LR73 cells treated with various doses of NVP-AEW541, a small molecule inhibitor of IGF-1R (n = 3). Right, representative immunoblot of LR73 cells stimulated with IGF-1 and treated with increasing doses of NVP-AEW541 (n = 2). b, c, Engulfment of apoptotic thymocytes by LR73 cells treated with the indicated concentrations of human insulin (b) and human IGF-II (c) (n = 2). Error bars represent s.d.
Extended Data Figure 3 Blocking canonical signalling intermediates downstream of IGF-1 receptor signalling, Rho-kinase- or Arp2/3-mediated functions, does not reverse the IGF-1 mediated engulfment modulation.
a–f, Engulfment of apoptotic thymocytes by LR73 cells treated with U2016 (Erk1/2 inhibitor) (a), MK-2206 (Akt1/2/3 inhibitor) (b), Rapamycin (mTOR inhibitor) (c), or Wortmannin (PI 3-Kinase inhibitor) (d), Rho-kinase inhibitors Y27632 (e) or GSK269962 (f) in the presence or absence of IGF-1 (n = 2–3). Initially, it appeared that inhibition of Rho-kinase was able to partially rescue IGF-1 induced engulfment suppression. However, as Rho-kinase inhibition basally increases phagocytosis of apoptotic cells (consistent with what has been previously reported), we normalized the change in phagocytosis for each inhibitor concentration to the appropriate control (right panel). After normalizing, we observed that Rho-kinase inhibition did not increase apoptotic cell uptake in LR73 cells in the presence of IGF-1 more than the increase observed basally owing to Rho-kinase inhibition. Thus, inhibition of Rho-kinase does not appear to rescue IGF-1-induced engulfment suppression. g, LR73 cells were treated with CK-666 and then assessed for uptake of liposomes in the presence of IGF-1. Data represented as mean ± s.d.
Extended Data Figure 4 Macrophages express IGF-1R and phosphorylate IGF-1R upon IGF-1 stimulation, but engulf apoptotic cells at normal capacity when exposed to IGF-1 or insulin.
a, b, J774 cells (a) or LR73 cells (b) treated with 100 ng ml−1 mouse IGF-1 were assessed for their ability to engulf apoptotic thymocytes, or serum-starved for 6 h and stimulated with 100 ng ml−1 mouse IGF-1 and assessed for phosphorylation of IGF-1R by western blot. c, Flow cytometry histograms of IGF-1R expression on J774 cells (left), bone-marrow-derived macrophages (middle), and peritoneal macrophages (right) (n = 3–4). d, e, IC-21 cells treated with indicated concentrations of mouse IGF-1 (d) or human insulin (e) were assessed for their ability to engulf apoptotic thymocytes (n = 2–3). Error bars represent s.d.
Extended Data Figure 5 Production of IGF-1 by peritoneal macrophages after apoptotic cell or IL-4 stimulation correlates with new transcription.
a, Peritoneal macrophages were either untreated, stimulated with rIL-4 or apoptotic Jurkat cells and Igf1 mRNA (top panels) and IGF-1 protein in the supernatant (bottom panels) were assessed in a time course (n.d., not detected) (n = 3). Data represented as mean ± s.d. b, c, Lung sections from wild-type mice were stained with antibodies against alveolar macrophages (Mac-2), airway epithelial cells (CC-10), and IGF-1R. d, Alveolar macrophages isolated from LysM-Cre/Igf1fl/fl and littermate controls were assessed for Igf1 mRNA expression (n = 2 per group, data represented as mean ± s.e.m.).
Extended Data Figure 6 CCSP-Cre/Igf1rfl/fl mice exposed to HDM have greater airway resistance and show a trend towards greater immune cell infiltration in the lungs and more apoptotic cells.
a, Total cell counts of lung CD3+CD4+ T cells (left), CD3+CD4+CD44+ T cells (middle), and CD3+CD4+CD69+ T cells (right panel) in the lungs of CCSP-Cre/Igf1r+/+ and Igf1rfl/fl mice given the full HDM course. b, Airway hyper-responsiveness to methacholine (another measure of allergen sensitivity) in the CCSP-Cre/Igf1rfl/fl mice compared to control CCSP-Cre/Igf1r+/+ mice treated with HDM (n = 6–8 mice per group). c, Representative histology images of cleaved caspase (CC3) staining in lung sections of mice given the full HDM course. Black arrowheads indicate positive staining. Average CC3-positive cells per mouse are quantified on the right (n = 3 per group). Data represented as mean ± s.e.m. P value of <0.01 (**).
Extended Data Figure 7 Schematic IGF-1R deletion during the sensitization versus challenge phases of HDM administration, and the response of CCSP-Cre/Igf1r+/+ and Igf1rfl/fl mice in the second regime (the challenge phase).
a, Schematic describing the different time courses for Igf1r deletion from Club cells (induced via administration of doxycycline) and for the allergen HDM exposure. b, Total cell counts of various populations in the BAL fluid of CCSP-Cre/Igf1r+/+ and CCSP-Cre/Igf1rfl/fl mice given HDM according to the second regimen. c, Total cell counts of CD3+CD4+ T cells of draining lymph nodes of CCSP-Cre/Igf1r+/+ and CCSP-Cre/Igf1rfl/fl mice given HDM according to the second regimen. Data represented as mean ± s.e.m. NS, notsignificant. P value of <0.05 (*).
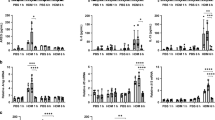
Extended Data Figure 8 Alveolar-macrophage-derived microvesicles suppress gene expression in lung epithelial cells exposed to HDM extract.
a, Microvesicles (MV) were collected from either control or IL-4-treated MH-S alveolar macrophages and then counted using qNano (n = 3). b, Supernatants from IL-4-treated MH-S macrophages were assessed for IGF-1 secretion. c, BEAS-2B cells were treated with HDM either in the presence or absence of alveolar-macrophage-derived microvesicles for 3 h and then assessed for expression of FGF2, KLF4, IFIT2, and PTX3 (n = 6). Data represented as mean ± s.e.m. P value of <0.01 (**), or <0.001 (***).
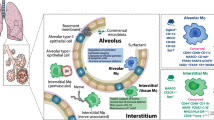
Extended Data Figure 9 Model for alveolar macrophage regulation of airway epithelial cells, with respect to particle uptake and the response to allergens, through IGF-1 and microvesicles.
Exposure of airways to allergens, such as HDM, can cause apoptotic cell death as well as IL-4 and IL-13 production, from mast cells and type-2 innate lymphoid cells (ILC2s). These cytokines, along with apoptotic cells, trigger alveolar macrophages to produce IGF-1. The released IGF-1 (a) then acts on the airway epithelium to elicit two actions: first, to decrease the uptake of apoptotic cells, and second, to enhance the uptake of macrophage-derived microvesicles. These microvesicles (b) dampen inflammatory cytokine production through airway epithelial cells.
Supplementary information
Supplementary Information
This file contains the raw data for Figures 1b,g,n Extended Data Figures 1b, c-e, 2a and 4a,b. (PDF 7269 kb)
Rights and permissions
About this article
Cite this article
Han, C., Juncadella, I., Kinchen, J. et al. Macrophages redirect phagocytosis by non-professional phagocytes and influence inflammation. Nature 539, 570–574 (2016). https://doi.org/10.1038/nature20141
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature20141
This article is cited by
-
Advances in biological functions and applications of apoptotic vesicles
Cell Communication and Signaling (2023)
-
Macrophage-based therapy for intervertebral disc herniation: preclinical proof-of-concept
npj Regenerative Medicine (2023)
-
Efferocytosis requires periphagosomal Ca2+-signaling and TRPM7-mediated electrical activity
Nature Communications (2022)
-
Understanding the Phagocytosis of Particles: the Key for Rational Design of Vaccines and Therapeutics
Pharmaceutical Research (2022)
-
Determining the effector response to cell death
Nature Reviews Immunology (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.