Abstract
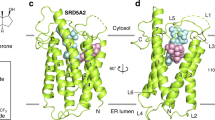
Cytochrome P450 17A1 (also known as CYP17A1 and cytochrome P450c17) catalyses the biosynthesis of androgens in humans1. As prostate cancer cells proliferate in response to androgen steroids2,3, CYP17A1 inhibition is a new strategy to prevent androgen synthesis and treat lethal metastatic castration-resistant prostate cancer4, but drug development has been hampered by lack of information regarding the structure of CYP17A1. Here we report X-ray crystal structures of CYP17A1, which were obtained in the presence of either abiraterone, a first-in-class steroidal inhibitor recently approved by the US Food and Drug Administration for late-stage prostate cancer5, or TOK-001, an inhibitor that is currently undergoing clinical trials4,6. Both of these inhibitors bind the haem iron, forming a 60° angle above the haem plane and packing against the central I helix with the 3β-OH interacting with aspargine 202 in the F helix. Notably, this binding mode differs substantially from those that are predicted by homology models and from steroids in other cytochrome P450 enzymes with known structures, and some features of this binding mode are more similar to steroid receptors. Whereas the overall structure of CYP17A1 provides a rationale for understanding many mutations that are found in patients with steroidogenic diseases, the active site reveals multiple steric and hydrogen bonding features that will facilitate a better understanding of the enzyme’s dual hydroxylase and lyase catalytic capabilities and assist in rational drug design. Specifically, structure-based design is expected to aid development of inhibitors that bind only CYP17A1 and solely inhibit its androgen-generating lyase activity to improve treatment of prostate and other hormone-responsive cancers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Miller, W. L. & Auchus, R. J. The molecular biology, biochemistry, and physiology of human steroidogenesis and its disorders. Endocr. Rev. 32, 81–151 (2011)
Attard, G., Reid, A. H., Olmos, D. & de Bono, J. S. Antitumor activity with CYP17 blockade indicates that castration-resistant prostate cancer frequently remains hormone driven. Cancer Res. 69, 4937–4940 (2009)
Yap, T. A., Carden, C. P., Attard, G. & de Bono, J. S. Targeting CYP17: Established and novel approaches in prostate cancer. Curr. Opin. Pharmacol. 8, 449–457 (2008)
Vasaitis, T. S., Bruno, R. D. & Njar, V. C. CYP17 inhibitors for prostate cancer therapy. J. Steroid Biochem. Mol. Biol. 125, 23–31 (2011)
de Bono, J. S. et al. Abiraterone and increased survival in metastatic prostate cancer. N. Engl. J. Med. 364, 1995–2005 (2011)
Molina, A. & Belidegrun, A. Novel therapeutic strategies for castration resistant prostate cancer: inhibition of persistent androgen production and androgen receptor mediated signaling. J. Urol. 185, 787–794 (2011)
Auchus, R. J., Geller, D. H., Lee, T. C. & Miller, W. L. The regulation of human P450c17 activity: relationship to premature adrenarche, insulin resistance and the polycystic ovary syndrome. Trends Endocrinol. Metab. 9, 47–50 (1998)
Attard, G. et al. Phase I clinical trial of a selective inhibitor of CYP17, abiraterone acetate, confirms that castration-resistant prostate cancer commonly remains hormone driven. J. Clin. Oncol. 26, 4563–4571 (2008)
Brodie, A., Njar, V., Macedo, L. F., Vasitis, T. S. & Sabnis, G. The Coffey Lecture: steroidogenic enzyme inhibitors and hormone dependent cancer. Urol. Oncol. 27, 53–63 (2009)
Imai, T. et al. Expression and purification of functional human 17α-hydroxylase/17,20-lyase (P450c17) in Escherichia coli. Use of this system for study of a novel form of combined 17α-hydroxylase/17,20-lyase deficiency. J. Biol. Chem. 268, 19681–19689 (1993)
Huang, P., Chandra, V. & Rastinejad, F. Structural overview of the nuclear receptor superfamily: insights into physiology and therapeutics. Annu. Rev. Physiol. 72, 247–272 (2010)
Pereira de Jésus-Tran, K. et al. Comparison of crystal structures of human androgen receptor ligand-binding domain complexed with various agonists reveals molecular determinants responsible for binding affinity. Protein Sci. 15, 987–999 (2006)
Vasaitis, T. et al. Androgen receptor inactivation contributes to antitumor efficacy of 17α-hydroxylase/17,20-lyase inhibitor 3β-hydroxy-17-(1H-benzimidazole-1-yl)androsta-5,16-diene in prostate cancer. Mol. Cancer Ther. 7, 2348–2357 (2008)
Ghosh, D., Griswold, J., Erman, M. & Pangborn, W. Structural basis for androgen specificity and oestrogen synthesis in human aromatase. Nature 457, 219–223 (2009)
Mast, N. et al. Structural basis for three-step sequential catalysis by the cholesterol side chain cleavage enzyme CYP11A1. J. Biol. Chem. 286, 5607–5613 (2011)
Mast, N. et al. Crystal structures of substrate-bound and substrate-free cytochrome P450 46A1, the principal cholesterol hydroxylase in the brain. Proc. Natl Acad. Sci. USA 105, 9546–9551 (2008)
Dhir, V. et al. Steroid 17α-hydroxylase deficiency: Functional characterization of four mutations (A174E, V178D, R440C, L465P) in the CYP17A1 gene. J. Clin. Endocrinol. Metab. 94, 3058–3064 (2009)
Rosa, S. et al. Clinical, genetic and functional characteristics of three novel CYP17A1 mutations causing combined 17α-hydroxylase/17,20-lyase deficiency. Horm. Res. Paediatr. 73, 198–204 (2010)
Katsumata, N., Ogawa, E., Fujiwara, I. & Fujikura, K. Novel CYP17A1 mutation in a Japanese patient with combined 17α-hydroxylase/17,20-lyase deficiency. Metabolism 59, 275–278 (2010)
Ergun-Longmire, B. et al. Two novel mutations found in a patient with 17α-hydroxylase enzyme deficiency. J. Clin. Endocrinol. Metab. 91, 4179–4182 (2006)
Sahakitrungruang, T., Tee, M. K., Speiser, P. W. & Miller, W. L. Novel P450c17 mutation H373D causing combined 17α-hydroxylase/17,20-lyase deficiency. J. Clin. Endocrinol. Metab. 94, 3089–3092 (2009)
Biason-Lauber, A. et al. 17α-hydroxylase/17,20-lyase deficiency as a model to study enzymatic activity regulation: role of phosphorylation. J. Clin. Endocrinol. Metab. 85, 1226–1231 (2000)
Lee-Robichaud, P. et al. The cationic charges on Arg347, Arg358 and Arg449 of human cytochrome P450c17 (CYP17) are essential for the enzyme's cytochrome b5-dependent acyl-carbon cleavage activities. J. Steroid Biochem. Mol. Biol. 92, 119–130 (2004)
Gupta, M. K., Geller, D. H. & Auchus, R. J. Pitfalls in characterizing P450c17 mutations associated with isolated 17,20-lyase deficiency. J. Clin. Endocrinol. Metab. 86, 4416–4423 (2001)
Tiosano, D. et al. Metabolic evidence for impaired 17α-hydroxylase activity in a kindred bearing the E305G mutation for isolate 17,20-lyase activity. Eur. J. Endocrinol. 158, 385–392 (2008)
Auchus, R. J., Lee, T. C. & Miller, W. L. Cytochrome b5 augments the 17,20-lyase activity of human P450c17 without direct electron transfer. J. Biol. Chem. 273, 3158–3165 (1998)
Swart, A. C., Storbeck, K. H. & Swart, P. A single amino acid residue, Ala 105, confers 16α-hydroxylase activity to human cytochrome P450 17α-hydroxylase/17,20 lyase. J. Steroid Biochem. Mol. Biol. 119, 112–120 (2010)
Haider, S. M., Patel, J. S., Poojari, C. S. & Neidle, S. Molecular modeling on inhibitor complexes and active-site dynamics of cytochrome P450 C17, a target for prostate cancer therapy. J. Mol. Biol. 400, 1078–1098 (2010)
Jagusch, C. et al. Synthesis, biological evaluation and molecular modelling studies of methyleneimidazole substituted biaryls as inhibitors of human 17α-hydroxylase-17,20-lyase (CYP17). Part I: heterocyclic modifications of the core structure. Bioorg. Med. Chem. 16, 1992–2010 (2008)
Pechurskaya, T. A., Lukashevich, O. P., Gilep, A. A. & Usanov, S. A. Engineering, expression, and purification of “soluble” human cytochrome P45017α and its functional characterization. Biochemistry 73, 806–811 (2008)
Leslie, A. G. W. MOSFLM 6.0 (Cambridge, 1998)
Collaborative Computational Project, 4. The CCP4 suite: programs for protein crystallography. Acta Crystallogr. D 50, 760–763 (1994)
Long, F., Vagin, A. A., Young, P. & Murshudov, G. N. BALBES: a molecular replacement pipeline. Acta Crystallogr. D 64, 125–132 (2008)
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D 60, 2126–2132 (2004)
Murshudov, G. N., Vagin, A. A. & Dodson, E. J. Refinement of macromolecular structures by the maximum-likelihood method. Acta Crystallogr. D 53, 240–255 (1997)
Hooft, R. W., Vriend, G., Sander, C. & Abola, E. E. Errors in protein structures. Nature 381, 272 (1996)
Laskowski, R. A., MacArthur, M. W., Moss, D. S. & Thornton, J. M. PROCHECK: a program to check the stereochemical quality of protein structures. J. Appl. Crystallogr. 26, 283–291 (1993)
DeVore, N. M. et al. Key residues controlling binding of diverse ligands to human cytochrome P450 2A enzymes. Drug Metab. Dispos. 37, 1319–1327 (2009)
DeLano, W. L. The PyMol Molecular Graphics System (DeLano Scientific, 2002)
Kleywegt, G. J. & Jones, T. A. Detection, delineation, measurement and display of cavities in macromolecular structures. Acta Crystallogr. D 50, 178–185 (1994)
Jain, A. N. Surflex: fully automatic flexible molecular docking using a molecular similarity-based search engine. J. Med. Chem. 46, 499–511 (2003)
Hutschenreuter, T. U., Ehmer, P. B. & Hartmann, R. W. Synthesis of hydroxy derivatives of highly potent non-steroidal CYP 17 inhibitors as potential metabolites and evaluation of their activity by a non cellular assay using recombinant human enzyme. J. Enzyme Inhib. Med. Chem. 19, 17–32 (2004)
Shen, A. L., Porter, T. D., Wilson, T. E. & Kasper, C. B. Structural analysis of the FMN binding domain of NADPH-cytochrome P-450 oxidoreductase by site-directed mutagenesis. J. Biol. Chem. 264, 7584–7589 (1989)
Acknowledgements
X-ray data were collected at the Stanford Synchrotron Radiation Lightsource (SSRL). The SSRL Structural Molecular Biology Program is supported by the US Department of Energy Office of Biological and Environmental Research and by the US National Institutes of Health (NIH), National Center for Research Resources, Biomedical Technology Program and the National Institute of General Medical Sciences. We thank C.-J. Liu and the University of Kansas (KU) Center of Biomedical Research Excellence (COBRE) Center for Cancer Experimental Therapeutics for synthesizing abiraterone (NIH RR030926), M. R. Waterman for the full-length CYP17A1 construct, J. Wang for assistance with the Fe(iv) = O construct used in docking and A. Skinner and J. Aubé for manuscript suggestions. This research was funded by the NIH through the KU COBRE Center for Protein Structure and Function (NIH RR17708) and GM076343.
Author information
Authors and Affiliations
Contributions
Author Contributions N.M.D. engineered, expressed, characterized, purified and crystallized CYP17A1 under the direction of E.E.S. N.M.D. and E.E.S. jointly performed X-ray diffraction experiments, solved and refined the structures, and wrote the manuscript. N.M.D. performed the docking studies of CYP17A1.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-6 with legends, Supplementary Tables 1-2 and additional references. (PDF 2353 kb)
Rights and permissions
About this article
Cite this article
DeVore, N., Scott, E. Structures of cytochrome P450 17A1 with prostate cancer drugs abiraterone and TOK-001. Nature 482, 116–119 (2012). https://doi.org/10.1038/nature10743
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature10743
This article is cited by
-
Steroidogenic cytochrome P450 enzymes as drug target
Toxicological Research (2024)
-
Enhancing the biotransformation efficiency of human CYP17A1 in Pichia pastoris by co-expressing CPR and glucose-6-phosphate dehydrogenase simultaneously
Systems Microbiology and Biomanufacturing (2024)
-
Development of abiraterone acetate tablets with enhanced oral bioavailability
Journal of Pharmaceutical Investigation (2024)
-
In-silico activity prediction and docking studies of some flavonol derivatives as anti-prostate cancer agents based on Monte Carlo optimization
BMC Chemistry (2023)
-
Selective steroidogenic cytochrome P450 haem iron ligation by steroid-derived isonitriles
Communications Chemistry (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.