Abstract
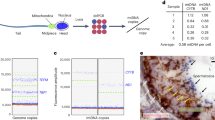
Mutations in mitochondrial DNA (mtDNA) are a common cause of genetic disease. Pathogenic mutations in mtDNA are detected in approximately 1 in 250 live births1,2,3 and at least 1 in 10,000 adults in the UK are affected by mtDNA disease4. Treatment options for patients with mtDNA disease are extremely limited and are predominantly supportive in nature. Mitochondrial DNA is transmitted maternally and it has been proposed that nuclear transfer techniques may be an approach for the prevention of transmission of human mtDNA disease5,6. Here we show that transfer of pronuclei between abnormally fertilized human zygotes results in minimal carry-over of donor zygote mtDNA and is compatible with onward development to the blastocyst stage in vitro. By optimizing the procedure we found the average level of carry-over after transfer of two pronuclei is less than 2.0%, with many of the embryos containing no detectable donor mtDNA. We believe that pronuclear transfer between zygotes, as well as the recently described metaphase II spindle transfer, has the potential to prevent the transmission of mtDNA disease in humans.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Elliott, H. R., Samuels, D. C., Eden, J. A., Relton, C. L. & Chinnery, P. F. Pathogenic mitochondrial DNA mutations are common in the general population. Am. J. Hum. Genet. 83, 254–260 (2008)
Vandebona, H. et al. Prevalence of mitochondrial 1555A→G mutation in adults of European descent. N. Engl. J. Med. 360, 642–644 (2009)
Bitner-Glindzicz, M. et al. Prevalence of mitochondrial 1555A→G mutation in European children. N. Engl. J. Med. 360, 640–642 (2009)
Schaefer, A. M. et al. Prevalence of mitochondrial DNA disease in adults. Ann. Neurol. 63, 35–39 (2008)
Sato, A. et al. Gene therapy for progeny of mito-mice carrying pathogenic mtDNA by nuclear transplantation. Proc. Natl Acad. Sci. USA 102, 16765–16770 (2005)
Brown, D. T. et al. Transmission of mitochondrial DNA disorders: possibilities for the future. Lancet 368, 87–89 (2006)
Wallace, D. C. Why do we still have a maternally inherited mitochondrial DNA? Insights from evolutionary medicine. Annu. Rev. Biochem. 76, 781–821 (2007)
DiMauro, S. & Schon, E. A. Mitochondrial disorders in the nervous system. Annu. Rev. Neurosci. 31, 91–123 (2008)
Man, P. Y. et al. The epidemiology of Leber hereditary optic neuropathy in the north east of England. Am. J. Hum. Genet. 72, 333–339 (2003)
Taylor, R. W. & Turnbull, D. M. Mitochondrial DNA mutations in human disease. Nature Rev. Genet. 6, 389–402 (2005)
Steffann, J. et al. Analysis of mtDNA variant segregation during early human embryonic development: a tool for successful NARP preimplantation diagnosis. J. Med. Genet. 43, 244–247 (2006)
Piko, L. & Taylor, K. D. Amounts of mitochondrial DNA and abundance of some mitochondrial gene transcripts in early mouse embryos. Dev. Biol. 123, 364–374 (1987)
Barritt, J. A., Kokot, M., Cohen, J., Steuerwald, N. & Brenner, C. A. Quantification of human ooplasmic mitochondria. Reprod. Biomed. Online 4, 243–247 (2002)
Lin, D. P. et al. Comparison of mitochondrial DNA contents in human embryos with good or poor morphology at the 8-cell stage. Fertil. Steril. 81, 73–79 (2004)
Tajima, H. et al. The development of novel quantification assay for mitochondrial DNA heteroplasmy aimed at preimplantation genetic diagnosis of Leigh encephalopathy. J. Assist. Reprod. Genet. 24, 227–232 (2007)
Tachibana, M. et al. Mitochondrial gene replacement in primate offspring and embryonic stem cells. Nature 461, 367–372 (2009)
Battaglia, D. E., Goodwin, P., Klein, N. A. & Soules, M. R. Fertilization and early embryology: influence of maternal age on meiotic spindle assembly oocytes from naturally cycling women. Hum. Reprod. 11, 2217–2222 (1996)
Almeida, P. A. & Bolton, V. N. The effect of temperature fluctuations on the cytoskeletal organisation and chromosomal constitution of the human oocyte. Zygote 3, 357–365 (1995)
Meirelles, F. V. & Smith, L. C. Mitochondrial genotype segregation in a mouse heteroplasmic lineage produced by embryonic karyoplast transplantation. Genetics 145, 445–451 (1997)
Feenan, K. & Herbert, M. Can ‘abnormally’ fertilized zygotes give rise to viable embryos? Hum. Fertil. (Camb.) 9, 157–169 (2006)
Chinnery, P., Majamaa, K., Turnbull, D. & Thorburn, D. Treatment for mitochondrial disorders. Cochrane Database Syst. Rev. CD004426 (2006)
McFarland, R. et al. Multiple neonatal deaths due to a homoplasmic mitochondrial DNA mutation. Nature Genet. 30, 145–146 (2002)
Rahman, S., Poulton, J., Marchington, D. & Suomalainen, A. Decrease of 3243 A→G mtDNA mutation from blood in MELAS syndrome: a longitudinal study. Am. J. Hum. Genet. 68, 238–240 (2001)
Reik, W. et al. Adult phenotype in the mouse can be affected by epigenetic events in the early embryo. Development 119, 933–942 (1993)
Taylor, R. W., Taylor, G. A., Durham, S. E. & Turnbull, D. M. The determination of complete human mitochondrial DNA sequences in single cells: implications for the study of somatic mitochondrial DNA point mutations. Nucleic Acids Res. 29, e74 (2001)
Andrews, R. M. et al. Reanalysis and revision of the Cambridge reference sequence for human mitochondrial DNA. Nature Genet. 23, 147 (1999)
Taylor, R. W. et al. A homoplasmic mitochondrial transfer ribonucleic acid mutation as a cause of maternally inherited hypertrophic cardiomyopathy. J. Am. Coll. Cardiol. 41, 1786–1796 (2003)
McFarland, R. et al. Familial myopathy: new insights into the T14709C mitochondrial tRNA mutation. Ann. Neurol. 55, 478–484 (2004)
He, L. et al. Detection and quantification of mitochondrial DNA deletions in individual cells by real-time PCR. Nucleic Acids Res. 30, e68 (2002)
Krishnan, K. J., Bender, A., Taylor, R. W. & Turnbull, D. M. A multiplex real-time PCR method to detect and quantify mitochondrial DNA deletions in individual cells. Anal. Biochem. 370, 127–129 (2007)
Acknowledgements
We thank M. Nesbitt, L. Burgess and S. Byerley for help with embryo donation and collection, and V. Wilson and S. Abbs for help with the nuclear genotyping. We thank the patients and staff at Newcastle Fertility Centre, and J. Lawford-Davies, K. Stern, Sir John Burn and Lord Walton of Detchant for helping us to obtain a Human Fertilisation and Embryology Authority research licence and guidance with the legislation. This work was funded by the Muscular Dystrophy Campaign, the Wellcome Trust (074454/Z/04/Z), the Medical Research Council (G0601157, G0601943), One North East, the UK National Institute for Health Research Biomedical Research Centre for Ageing and Age-related Disease and the Newcastle University Centre for Brain Ageing and Vitality supported by the Biotechnology and Biological Sciences Research Council, the Engineering and Physical Sciences Research Council, the Economic and Social Research Council and the Medical Research Council (G0700718). P.F.C. is a Wellcome Trust Senior Fellow in Clinical Science.
Author information
Authors and Affiliations
Contributions
M.H., A.P.M., R.N.L. and D.M.T. conceived the project and designed the experiments. L.C., H.A.T., S.J.H., G.D.G., J.L.M., L.M.C., P.F.C. and R.W.T. performed experiments and analysed data. L.C., M.H., H.A.T. and D.M.T. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figure 1 with legend and Supplementary Tables 1-2. (PDF 158 kb)
Rights and permissions
About this article
Cite this article
Craven, L., Tuppen, H., Greggains, G. et al. Pronuclear transfer in human embryos to prevent transmission of mitochondrial DNA disease. Nature 465, 82–85 (2010). https://doi.org/10.1038/nature08958
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature08958
This article is cited by
-
Novel economical, accurate, sensitive, single-cell analytical method for mitochondrial DNA quantification in mtDNA mutation carriers
Journal of Assisted Reproduction and Genetics (2023)
-
Assessment of ForenSeq mtDNA Whole Genome Kit for forensic application
International Journal of Legal Medicine (2023)
-
Mitochondrial Replacement Therapy: An Islamic Perspective
Journal of Bioethical Inquiry (2023)
-
Mesenchymal stem cells (MSCs) in Leber’s hereditary optic neuropathy (LHON): a potential therapeutic approach for future
International Ophthalmology (2022)
-
Mitochondria: emerging therapeutic strategies for oocyte rescue
Reproductive Sciences (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.