Abstract
BP1 is a member of the homeobox gene superfamily of transcription factors that are essential for early development. Significant mRNA expression and immunohistochemical reactivity of BP1 is present in a majority of breast cancers and in all cases of inflammatory breast cancer. This study attempts to determine whether BP1 expression is detectable in prostate cancer, another hormone dependent solid tumor, and whether this expression correlates with histopathologic and prognostic factors. Paraffin sections from radical prostatectomy cancer specimens and from tissue microarray sections of prostate cancer, obtained from the Prostate Cancer Tissue Registry (NIH), were assayed for BP1 immunoreactivity. Immunoreactivity scoring by two independent pathologists, using a three-tiered system (0, 1+, 2+), was recorded and correlated with Gleason scoring and prostatic specific antigen (PSA) biochemical recurrence. Ki-67 (MIB-1) immunoreactivity was performed to assess proliferation. Kappa and Cochran–Mantel–Haenszel statistical analyses were used to assess interobserver agreement and pathobiologic correlations. Significant BP1 immunoreactivity (2+) was identified in approximately 70% of prostatic adenocarcinomas, whether the analysis was performed on tissue sections (50 cases) or tissue microarray platforms (123 cases). BP1 immunoreactivity was seen in <5% of normal acinar cells. The agreement between two separate observers was very good, with kappa-statistics >0.7. In tissue sections, 12 cases with paired carcinoma and prostatic intraepithelial neoplasia (PIN) showed concordance with strong immunoreactivity. Gleason scores or prostatic specific antigen (PSA) biochemical recurrences were not correlated with strong BP1 immunoreactivity. Tumor proliferation, assayed with Ki-67 (MIB-1) immunoreactivity, was higher in cancer cells that were BP1 immunoreactive, relative to those that were BP1 non-reactive. These findings suggest that BP1 is an important upstream factor in the carcinogenic pathway of prostate cancer and that the expression of BP1 may reflect or directly contribute to tumor progression and/or invasion.
Similar content being viewed by others
Main
Prostate cancer is the most common solid malignancy in men and the third leading cause of cancer deaths in the USA. Prostate cancer shares certain features with breast cancer in being a hormone dependent cancer that arises from an in situ or intraepithelial dysplastic proliferation, and tends to predominate in older rather than younger adults. Similar to breast cancer, the disease has a higher incidence in Americans relative to Japanese and follows a relatively more aggressive course in African Americans than in Caucasian Americans.1
Adenocarcinoma of the acinar glands of the prostate is the most common form of prostate cancer and typically arises in the peripheral zone of the gland with prostatic intraepithelial neoplasia (PIN) as its malignant precursor. Prognostic factors for the survival of men with prostate cancer include Gleason score, a marker of histologic grade and tumor differentiation, tumor size, extent and stage, and serum levels of PSA.2
BP1, a gene cloned in our laboratory, is a member of the homeobox gene superfamily of transcription factors that are essential for early development.3 Our studies in breast cancer have demonstrated that BP1 expression was present in 80% of infiltrating duct carcinomas and relatively absent in matched normal controls.4, 5 We have also shown that BP1 expression was significantly higher in cancers that were estrogen receptor negative relative to estrogen receptor positive breast tumors and in African Americans than in Caucasians.4
BP1 demonstrates significant expression in breast cancers with a relative aggressive phenotype, namely, estrogen receptor negative and inflammatory breast cancers, and consequently, we may propose several analogies that may be relevant to prostate cancers. Invasive cancers are believed to result from a progressive, multi-step process initiated and driven by a sequential accumulation of genetic and biochemical abnormalities.6, 7 BP1 is expressed in precursor lesions, such as ductal carcinoma in situ, in addition to its prominent expression in established invasive breast cancer. By analogy, expression of BP1 in prostate cancer and its precursor lesion, high-grade prostatic intraepithelial carcinoma (PIN) would support the activity of BP1 as an upstream oncogenic factor. Second, as estrogen receptor negative and inflammatory breast cancers have an increased relative aggressive biologic behavior and worse clinical prognosis than usual estrogen receptor positive breast cancers,8, 9 the increased BP1 expression in the more aggressive breast cancer phenotypes may reflect or contribute to the biologic and clinical behavior of these tumors. Similar biologic and clinical behavior may be identified in prostate cancers with BP1 expression, namely that prostate cancers with BP1 expression may demonstrate increased Gleason patterns, higher proliferative rates, or early recurrence.
Based on these findings, this study probes the immunohistochemical expression profile of BP1 as an initial step in understanding the role of BP1 in prostate cancer. The prostate cancer samples have been obtained from prostatectomy specimens and available on tissue microarray platforms. The main goals of this study are to determine whether a defined pattern of BP1 immunoreactivity is detectable in prostate tumor tissues, whether the identification and scoring of immunoreactivity is concordant among pathologists, whether the expression may be present within cancer precursors (PIN), whether morphologically similar tumor cells with and without BP1 expression have different proliferation rates, and whether BP1 expression correlates with pathologic Gleason scoring and tumor recurrence.
Materials and methods
Prostate cancer cases were obtained from the files of the Armed Forces Institute of Pathology. Consecutive sections were made from formalin-fixed, paraffin-embedded normal prostate (n=5) and prostate cancers (n=50) with co-existing normal, and potential regions of prostatic hyperplasia and PIN. The first and last sections from each case were stained with hematoxylin and eosin for morphological classification. Tissue microarrays were obtained from the Cooperative Prostate Cancer Tissue Resource (CPCTR) from the National Cancer Institute of the NIH and were coded according to patient age, Gleason score, and serum PSA levels at the time of prostatic resection or biopsy, and the follow-up results of biochemical recurrence. These individuals ranged from 42 to 73 years, and an average age of 61 years was predominantly Caucasians (56%). Their Gleason scores were mostly 6 and 7, and their pretherapy PSA ranged from 1 to 20 predominantly, with few cases above a value of 20 ng/ml. The treatments were surgery in all the cases and the pathologic stages were T2 (68%) and T3 (32%). Overwhelmingly, the nodal status was N0, followed by NX, and less than 3% N1. Biochemical recurrence was defined as successive incremental values following post-treatment nadir of PSA.
A polyclonal antibody against the BP1 peptide was made from rabbit, as previously described.3 Monoclonal antibodies against human Ki-67 antigen (clone MIB-1), a biotinylated secondary antibody, an ABC detection kit (avidin-proxidase or Avidin-alkaline-phosphatase conjugated), a chromogen diaminobenzidine (DAB) kit, and normal serum were purchased from Vector (Burlingame, CA). The AP-red chromogen kit was obtained from Zymed (San Francisco, CA). A background block solution was from Cell Marque (Hot Springs, AR). Other immunoassaying related reagents, including the antigen retrieval solution, Tris-buffered saline (TBS), and hematoxylin and eosin were purchased from Biocare-Midical, LLC (Walnut, CA).
The sensitivity and specificity of the BP1 immunostaining was assured by purified BP1 protein pre-absorption, by antibody titration, and by antibody substitution with PBS. Optimization of immunoreactivity was obtained by pretreatment at 80°C for 2 h, an overnight incubation of the primary antibody at 4°C, and detection with avidin-alkaline phosphatase complex. This approach was used for immunoassaying of tissue section slides and tissue microarray platforms.
To monitor BP1 expression associated with cell proliferation, sections from morphologically similar prostatic cancers with (n=5) and without (n=5) BP1 expression were double immunostained for BP1 and a proliferation marker, MIB-1. The cell proliferation among 1000 BP1 immunoreactive and non-reactive tumor cells were enumerated and the rates among cases in each category were averaged and statistically compared using Student's t-test.
Normal and hyperplastic glands, high-grade PIN, and prostatic carcinoma were examined for BP1 expression. The immunoreactivity was scored as zero (0) for cases showing no immunoreactivity, low (1+) for weak or sparse immunoreactivity, and strong (2+) for cases of significant or intense immunoreactivity. Positive BP1 expression was assessed by identifying strong immunoreactivity (2+) in greater than 5% of acinar or ductal elements. Tissue microarray sections were similarly scored. Paired evaluations by two separate pathologists for the tissue sections (YGM and AMS) and for the microarray platforms (MKR and AMS) were performed and their results were calculated by kappa-statistics to evaluate diagnostic concordance. In cases containing multiple histological components, the BP1 expression status in each component was separately evaluated and recorded. The frequency of BP1 expression among normal, PIN, and neoplastic cells was statistically compared with the Student's t-test. Kappa and Cochran–Mantel–Haenszel statistical analyses were used to assess interobserver agreement and pathobiologic correlations.
Results
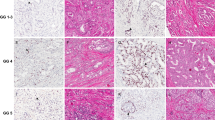
Samples from 50 prostate cancer cases were sectioned and immunostained with anti-BP1 antibody. Distinct BP1 immunoreactivity was identified predominantly in malignant acinar cells, PIN, and less commonly in benign acinar elements and rarely in endothelial cells (Figure 1). No distinct BP1 immunostaining was detected in fibroblasts, adipocytes, though intermediate (1+) immunostaining was occasionally present in smooth muscle cells. BP1 immunoreactivity was predominantly present in the cytoplasm, and minimally seen in the nuclei. Of the five normal controls containing only benign prostatic tissue, three were completely devoid of BP1 immunostaining in the glandular epithelium, and two contained BP1 immunoreactive cell clusters, which accounted for less than 5% of the total cell population, so did not meet the criteria for BP1 positivity. Within benign acinar glands, BP1 immunoreactivity was also occasionally identified in basal cells.
Evaluating the 50 cases containing co-existing normal prostatic tissue, benign acinar glands adjacent to carcinoma, PIN, and prostatic acinar adenocarcinoma demonstrated that the prevalence of BP1 positive elements increased significantly in association with PIN and carcinoma (P<0.001) (Table 1). Of the 50 tissue sections containing normal prostatic tissue adjacent to prostatic carcinoma, 10 areas of benign acinar glands (20%) adjacent to prostatic carcinoma were immunoreactive for BP1. Among the 50 tissue sections of carcinoma, 12 sections had identifiable areas of high-grade PIN, which were significantly BP1 immunoreactive (2+), in conformity with the staining intensity identified in the prostatic adenocarcinoma. BP1 immunoreactive adenocarcinoma of significant intensity was demonstrated in 37 of 50 cases. The majority of cancer cases were Gleason scores of 6, of which 72% were immunoreactive for BP1 expression. Though several cases of adenocarcinoma had Gleason scores of 8 and demonstrated BP1 immunoreactivity, the numbers are too small to generate statistical conclusions (Table 3). Interobserver concordance of the BP1 immunohistochemical scoring revealed a kappa-statistic above 0.65, for concordant 2+ immunostaining, indicative of good agreement.
In addition to the substantial increase in the percentage of BP1 positive cells in neoplastic relative to benign acinar glands, the intensity of BP1 immunostaining became more significant. In normal and acinar glands adjacent to carcinoma, BP1 positivity was generally distributed as clusters with a defined boundary with their adjacent BP1 negative acini and the intensity of staining was usually weak (1+) and less commonly significant (2+). In the malignant glands, BP1 immunoreactivity tended to be more uniform and intense (2+) (Figure 2). When the prostatic adenocarcinoma was doubly stained for BP1 and a proliferative marker (MIB-1), BP1 positive cells relative to non-reactive tumor cells showed a significantly higher (P>0.05) proliferation rate, 8.8% relative to 3.0%, respectively. The malignant cells that were reactive and non-reactive for BP1 were morphologically similar (Table 2).
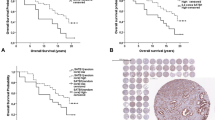
The BP1 immunoreactivity was 69% among a sampling of 123 individual cases of prostatic adenocarcinoma within the tissue microarray platforms. Positive immunoreactivity was strong and diffuse in the foci of prostatic carcinoma in the tissue microarray and the results were nearly identical with the extent of immunoreactivity identified in the tissue sections from prostatectomy samples. The cases were evaluated in duplicate, and the intra- and interobserver concordance was very good based on kappa-statistics of greater than 0.7. Significant BP1 immunoreactivity did not correlate with Gleason score, though the percentage of Gleason scores 8, 9, and 10 were under 12% of the total cases sampled in the tissue microarray and less than 14% of cases from the prostatectomy specimens (Table 3). BP1 immunoreactivity (2+) did not correlate with biochemical serum PSA recurrence, defined as consecutive elevations of PSA following post-resection PSA nadir, using the clinical data that accompanied the microarray platforms (Table 4).
Discussion
This study demonstrates that the frequency and intensity of BP1 expression, as measured by immunohistochemical reactivity, increases within PIN and prostatic carcinoma relative to benign acinar glands and that the extent and intensity of the immunoreactivity progresses through the carcinogenic stages of preneoplasia and invasive cancer. Compared to normal acinar components, the neoplastic glands consistently showed the highest number of BP1 positive cells and the highest intensity of BP1 immunoreactivity. There is little or no BP1 immunoreactivity in normal acinar glands from non-neoplastic prostatic resections. Benign acinar glands show modestly increased but scattered immunoreactivity in acinar glands adjacent to carcinoma, whereas malignant glands demonstrated diffuse and strong immunoreactivity in 70% of prostatic carcinomas. The overall expression of BP1 in prostatic adenocarcinomas and its enhanced expression through neoplastic progression resemble the BP1 expression pattern previously identified in breast cancers.4, 5 Analogous to breast cancers, BP1 immunoreactivity was not present in normal prostatic tissue, but demonstrated diffuse and strong immunoreactivity within neoplastic cells of PIN and invasive adenocarcinoma.
In the selected examples where high-grade PIN was discovered adjacent to carcinoma, the expression was concordantly strong in both preneoplastic and invasive carcinoma. PIN has been accepted as a morphologic pattern in the sequence of prostatic carcinogenesis that demonstrates cytopathologic, biochemical, and molecular features that are concordant with invasive carcinoma and differ markedly from benign and hyperplastic acinar glands.10 Molecular profiling has shown that PIN and invasive carcinoma share similar expression signatures that include protein synthetic pathways, induction of transcription factors, and oncogene activation.11, 12, 13 The transition from PIN to invasive carcinoma is marked by gene alterations that control cell adhesion, proteinase activity, and motility of the invasive phenotype.13 Utilizing molecular approaches or immunohistochemical assays, the development of PIN from normal glands or those undergoing proliferative inflammatory atrophy are characterized by an increase in alpha-methyl acylCo-A racemase (AMACR) and the transition to invasive carcinoma is marked by loss of PTEN and p27.14 Our findings that tissue sections containing both PIN and invasive carcinoma show identical significant BP1 immunoreactivity conforms to the cited research that BP1 acts proximally in the carcinogenic process of breast cancer. The multifocality of PIN and prostate carcinoma suggests that alteration of BP1 expression may be a necessary component of prostate carcinogenesis among a majority of prostate cancers.
We have also found BP1 enhanced expression in neoplasia other than prostate and breast cancer. BP1 is expressed in 63% of the bone marrows containing acute myeloid leukemia; 81% of pediatric and 47% of adult patients with acute myeloid leukemia, as well as in 32% of pediatric T-cell acute lymphocytic leukemia, patients demonstrate BP1 expression in their hematopoietic cancers.15 In contrast to leukemias and lymphomas, BP1 mRNA expression is barely if at all detectable in normal myeloid elements of the bone marrow or in PHA-stimulated T cells.15
The study provides supporting evidence that BP1 immunoreactivity is identified in both tissue sections from radical prostatectomy specimens as well as from tissue microarray platforms. The interpretive concordance of the BP1 staining patterns among pathologists with respect to malignant, pre-invasive, or benign foci was very good according to kappa-statistics. The diagnostic agreement of immunoreactivity of BP1 expression in cancers between the tissue blocks from surgical resections and the tissue microarray was very good. The ability of tissue microarray to provide thorough and enhanced multiple sample molecular profiling of diagnostic or predictive markers has been demonstrated,16, 17, 18, 19, 20 and mathematical and statistical analysis has shown justification and applicability to the methods.16, 17, 21 Our tissue microarray platform had the additional benefit of carrying abundant clinical data so that correlations could be assessed with the BP1 immunohistochemical reactivity. Though BP1 immunoreactivity is expressed in the majority of adenocarcinomas of the prostate, it currently lacks association with the Gleason score or degree of differentiation. This result needs to be further investigated, because the relative number of Gleason scores of 8 or above (diffuse Gleason pattern 4 or any Gleason pattern 5) was less than 12% of all the tissue microarray microsections. Similarly, the tissue sections had less than 14% Gleason scores equal to or greater than 8, and Gleason scores of or greater than 7 were only 28% of the total cases. Our total numbers were, as yet, insufficient to achieve statistical significance. Studies with larger sets of higher Gleason scores will be necessary to resolve whether BP1 expression correlates with higher grade tumors. Using the clinical data from the tissue microarray, BP1 immunoreactivity did not correlate with PSA biochemical recurrence. This clinical finding also needs to be confirmed with a larger dataset, though it may indicate that BP1 acts more proximally in the carcinogenic sequence and that other pathologic and biologic factors may predict tumor dissemination and recurrence. What is clear from our results is that tumor cells with BP1 expression tend to show a greater proliferative activity than those lacking BP1 immunoreactivity. The proliferation marker Ki-67 (MIB-1), as an indicator of cell-cycle activity, demonstrates nearly three times the expression (8.8 vs 3.0%) when the tumor cells co-express BP1 relative to tumor cells lacking BP1 immunoexpression. Previous results indicate that BP1 may have anti-apoptotic regulatory activity in both leukemia15 and breast cancer, where it regulates the bcl-2 gene.22 These published findings, as well as the current MIB-1 results, showing that BP1 expression correlates with increased cell proliferation markers, suggest that BP1 may have a role as a promoter and/or enhancer of cellular survival and proliferation with resultant tumor growth.
The specific role(s) of BP1 in tumor development and progression has not been clearly defined and delineated. Current and published work suggests a role for BP1 in carcinogenesis and progression. Based on previous work4, 5 and the present results in prostate cancer, it appears that BP1 is involved in the carcinogenic sequence as it appears to be expressed in 46% of ductal carcinomas in situ of the breast and all the examples of PIN identified in prostate cancer sections. Moreover, BP1 appears to be involved in cellular proliferation and in altering or blocking the pathway of programmed cell death (apoptosis). The process of tumor growth may facilitate the formation and expansion of biologically more aggressive cell clones. An example of this feature may be appreciated in that expression of BP1 in the leukemia cell line K562 substantially increased their clonogenicity, and abrogation of BP1 expression in K562 cells causes apoptosis (PE Berg, unpublished data). Both prostate and breast cancer cells expressing BP1 have a significantly higher cell proliferation index, compared to cells of identical morphology but lacking BP1 expression. Together, our current and past findings suggest that BP1 may be an important regulatory factor in the oncogenic pathway of several malignancies, including prostate cancer.
References
Drago JR . The role of new modalities in the early detection and diagnosis of prostate cancer. CA Cancer J Clin 1989;39:326–336.
Epstein JI, Srigley J, Grignon D . Recommendations for the reporting of prostate carcinoma. Hum Pathol 2007;38:1305–1309.
Chase MB, Fu S, Haga SB, et al. BP1, a homeodomain-containing isoform of DLX4, represses the beta-globin gene. Mol Cell Biol 2002;22:2505–2514.
Fu SW, Schwartz A, Stevenson H, et al. Correlation of expression of BP1, a homeobox gene, with estrogen receptor status in breast cancer. Breast Cancer Res 2003;5:82–87.
Man YG, Fu SW, Schwartz AE, et al. Expression of BP1, a homeobox gene, correlates with progression and invasion of mammary ductal carcinoma. Breast Cancer Res Treat 2005;90:241–247.
Schmitt FC . Multistep progression from an oestrogen-dependent growth towards an autonomous growth in breast carcinogenesis. Eur J Cancer 1995;31A:2049–2052.
Man YG, Martinez A, Avis IM, et al. Phenotypically different respiratory epithelial cells with hnRNP A2/B1 over-expression display similar genetic alterations. Am J Respir Cell Mol Biol 2000;23:636–645.
Sheikh MS, Garcia M, Pujol P, et al. Why are estrogen receptor negative breast cancers more aggressive than the estrogen receptor positive breast cancers? Invasion Metastasis 1995;14:329–336.
Kaptain S, Tan LK, Chen B . Her-2/neu and breast cancer. Diagn Mol Pathol 2001;10:139–151.
Bostwick DG, Qian J . High grade prostatic intraepithelial neoplasia. Mod Pathol 2004;17:360–379.
Ashida S, Nakagawa H, Katagiri T, et al. Molecular features of the transition from PIN to prostate cancer: genome-wide gene expression profiles of prostate cancers and PIN. Cancer Res 2004;64:5963–5972.
Konishi N, Shimada K, Ishida E . Molecular pathology of prostate cancer. Pathol Int 2005;55:531–539.
Tomlins SA, Mehra R, Rhodes DR, et al. Integrative molecular concept modeling of prostate cancer progression. Nat Genet 2007;39:41–51.
Gonzalgo ML, Isaacs WB . Molecular pathways to prostate cancer. J Urol 2003;170:2444–2452.
Haga SB, Fu S, Karp JE, et al. BP1, a new homeobox gene, is frequently expressed in acute leukemias. Leukemia 2000;14:1867–1875.
Camp RL, Chung GG, Rimm DL . Automated subcellular localization and quantitation of protein expression in tissue microarrays. Nat Med 2002;8:1323–1327.
Camp RL, Charette LA, Rimm DL . Validation of tissue microarray technology in breast carcinoma. Lab Invest 2000;80:1943–1949.
Rubin MA, Dunn R, Strawderman M, et al. Tissue microarray sampling strategy for prostate cancer biomarker analysis. Am J Surg Pathol 2002;26:312–319.
Zhang D, Salto-Tellez M, Putti TC, et al. Reliability of tissue microarrays in detecting protein expression and gene amplification in breast caner. Mod Pathol 2003;16:79–84.
Packeisen J, Korsching E, Herbst H, et al. Demystified tissue microarray technology. Mod Pathol 2003;56:198–204.
Liu X, Minin V, Huang Y . Statistical methods for analyzing tissue microarray data. J Biopharm Stat 2004;14:671–685.
Stevenson HS, Fu S, Pinzone JJ, et al. BP1 transcriptionally activates bcl-2 and inhibits TNFα-induced cell death in MCF7 breast cancer cells. Breast Cancer Res 2007;9:R60.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Disclosure/conflict of interest
The authors have no potential conflict of interest to declare.
Rights and permissions
About this article
Cite this article
Schwartz, A., Man, YG., Rezaei, M. et al. BP1, a homeoprotein, is significantly expressed in prostate adenocarcinoma and is concordant with prostatic intraepithelial neoplasia. Mod Pathol 22, 1–6 (2009). https://doi.org/10.1038/modpathol.2008.168
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/modpathol.2008.168
Keywords
This article is cited by
-
Management of Borderline Ovarian Tumors: Series of Case Report and Review of the Literature
Indian Journal of Surgery (2021)
-
NKL homeobox genes in leukemia
Leukemia (2012)
-
Homeodomain protein DLX4 counteracts key transcriptional control mechanisms of the TGF-β cytostatic program and blocks the antiproliferative effect of TGF-β
Oncogene (2011)
-
DNA methylation of HOXD3 as a marker of prostate cancer progression
Laboratory Investigation (2010)